

Acids and Bases
Presentation
•
Chemistry
•
9th - 12th Grade
•
Medium
Standards-aligned
Karina Marquez
Used 10+ times
FREE Resource
7 Slides • 44 Questions
1
Strong vs. Weak Acids & Bases
Strong acids and bases COMPLETELY disassociate in solution.
Weak acids and bases dissassociate less than 5%

2
Multiple Choice
Acids which dissociate almost completely in aqueous solutions are known as:
Dilute acids
Strong acids
Weak acids
Concentrated acids
3
Multiple Choice
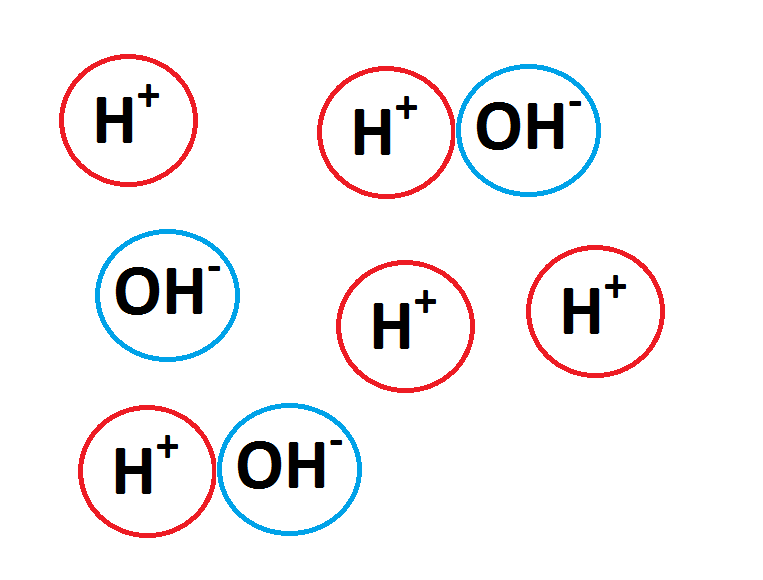
A substance that does not dissociate completely. Ranges from 4-7 on the pH scale.
Strong Acid
Strong Base
Weak Acid
Weak Base
4
Multiple Select
When comparing a strong and weak acid of the same concentration, the strong acid will have...(choose 2 answers)
A higher conductivity
A lower conductivity
A faster rate of reaction
A slower rate of reaction
5
Multiple Select
When comparing a strong and weak acid of the same concentration, the strong acid will have...(choose 2 answers)
A higher pH
A lower pH
a smaller temperature change on neutralisation with a strong base
a bigger temperature change on neutralisation with a strong base
6
Multiple Choice
--hydrochloric acid--
--hydrobromic acid--
--hydroiodic acid--
HClO3, HBrO3, HIO3
HCl, HBr, HI
HClO4, HBrO4, HIO4
HClO, HBrO, HIO
7
Multiple Choice
an acid
a base
an element
a metal
8
Multiple Choice
Dish Soap: pH of 12
Tomato Soup: pH of 4
Baking Soda: pH of 9
Drain Cleaner: pH of 14
9
Multiple Choice
it is a very strong base
it is a weak base
it is a weak acid
it is a strong acid
10
Match
Match each pH with the correct category:
strong acid
strong base
weak acid
weak base
neutral
1.0
13.5
5.5
8.5
7.0
1.0
13.5
5.5
8.5
7.0
11
Drag and Drop
Bases form
12
Electrolytes
Can be 3 things
- Acids (strong and weak)
- Bases ( Strong and weak)
- Salts (metal + nonmetal)
Disassociate in water.
NONelectrolytes do not.

13

14
Multiple Choice
_______ are corrosive substances characterized as having a strong smell, a sour taste, and a _______.
Acids; pH less than 7
Acids; pH greater than 7
Bases; pH greater than 7
Bases; pH less than 7
15

16
Multiple Choice
What is the conjugate acid of HCO31-?
Hint: charge of -1 so lost a proton (+1) so needs to get a proton back
CO3
H2CO3
CO32-
H2CO32-
17
Multiple Choice

Which two species would be identified as Bronsted-Lowry bases?
HF and F-
NH3 and F-
NH3 and NH4+
NH4+ and F-
18
Multiple Choice
What is the conjugate base of HClO4?
H+
Cl-
ClO3-
ClO4-
19
Match
Match the following
H2CO3 (aq) + H2O (l) <--> H3O+ (aq) + HCO3-- (aq)
H2CO3
H2O
H3O+
HCO3--
Bronsted-Lowry
Acid
Bronsted- Lowry
Base
Conjugate Acid
Conjugate Base
Bronsted-Lowry
Acid
Bronsted- Lowry
Base
Conjugate Acid
Conjugate Base
20
Multiple Select
Check all that apply: According to the Arrhenius definition:
An acid contains H and releases H+ ions in solution
An acid contains OH and releases OH- in solution
A base contains OH and releases OH- in solution
A base contains H and releases H+ ions in solution
21

22
Strength vs Concentration
Experience. Chemistry | Lesson 13.2
Two factors contribute to the pH of a solution
Strength-how much the acid ionizes
Stronger acids will have a lower pH than weak acids in the same amounts
Concentration: How many moles there are of the acid
A high concentration will have a low pH
23

24
Multiple Choice
do not break apart into ions (non-electrolyte)
partially break apart into ions (weak electrolyte)
completely break apart into ions (non-electrolyte)
completely break apart into ions (strong electrolyte)
25
Multiple Choice

Which is the stronger acid?
pH 1
pH 4
pH 8
pH 13
26
Multiple Choice
a neutral solution.
a stronger acid.
impossible to tell without testing the pH.
a stronger base
27
Multiple Choice
orange juice
water
vinegar
dishwashing detergent
28
Multiple Choice
shampoo
baking soda
orange juice
water
29
Multiple Choice
is a base
is a neutral substance
could be either an acid or a base
is an acid
30
Multiple Choice
oxide
oxygen
hydrogen
hydroxide
31
Multiple Choice
The lower the pH, the stronger the acid
the higher the pH, the stronger the acid
The lower the pH, the more neutral the acid
The higher the pH, the weaker the base
32
Multiple Choice
they form separate layers
they mix physically but not chemically
they break apart into separate elements
they react chemically to form a salt
33
Multiple Choice
8-14
0-7
7
1
34
Multiple Choice
8-14
0-7
7
1
35
Multiple Choice
8-14
0-7
7
1
36
Multiple Choice
...the strength of an acid.
...the strength of hydrogen ions.
...the concentration of hydrogen ions.
...the concentration of an acid
37
Multiple Choice
Which of the following word pairs correctly completes the sentence below?
_______ are corrosive substances characterized as having a oily feel, a bitter taste, and a _______.
Acids; pH less than 7
Acids; pH greater than 7
Bases; pH greater than 7
Bases; pH less than 7
38
Multiple Choice
Which type of ion does an acid produce when it is dissolved in water?
oxide
oxygen
hydronium
hydroxide
39
Multiple Choice
When an equal strength acid and base are mixed, what kind of chemical reaction occurs?
Neutralization
Combustion
Oxdation
Decomposition
40
Multiple Choice
A substance is found to have the following characteristics:
Very bitter taste
Feels slippery to the touch
Produces OH- ions when dissolved in water
In what category would the substance be classified?
acid
base
enzyme
fatty acid
41
Multiple Choice
HF is...
an acid
a base
a salt
an ionic compound
42
Multiple Choice
HNO2
a base
a salt
an acid
an ionic compound
43
Multiple Choice
base
strong base
acid
weak base
44
Multiple Choice
KOH is ...
an acid
a base
a salt
an ionic compound
45
Multiple Choice
An arrhenius acid
produces hydroxide ions in a solution
is a proton donor
produces hydrogen ions in a solution
is a proton acceptor
46
Multiple Choice
An arrhenius base
produces hydroxide ions in a solution
is a proton donor
produces hydrogen ions in a solution
is a proton acceptor
47
Multiple Choice
A bronsted lowry base
produces hydroxide ions in a solution
is a proton donor
produces hydrogen ions in a solution
is a proton acceptor
48
Multiple Choice
A bronsted lowry acid
produces hydroxide ions in a solution
is a proton donor
produces hydrogen ions in a solution
is a proton acceptor
49
Multiple Choice
Identify the true statement about the following reaction:
F- + H2O --> HF +OH-
H2O is a bronsted lowry base
H2O is not an acid or base according to either model
H2O is a bronsted lowry acid
H2O is an arrhenius acid
50
Multiple Choice
Acid-base conjugate pairs differ by a single
Electron
Proton
Neutron
Oxygen
51
Multiple Choice
Which of the following show an acid and its conjugate base pair (in that order)
H2SO4, SO42-
OH-, H2O
NH4+, H2O
H2CO3, HCO3-
Strong vs. Weak Acids & Bases
Strong acids and bases COMPLETELY disassociate in solution.
Weak acids and bases dissassociate less than 5%

Show answer
Auto Play
Slide 1 / 51
SLIDE
Similar Resources on Wayground

48 questions
Unit 4 Lesson 1 Bohr's Models and Lewis Dot Structures
Presentation
•
10th - 12th Grade

45 questions
Subatomic and Isotopes Review
Presentation
•
9th - 12th Grade

46 questions
Boyle's Law and Avogadro's Law
Presentation
•
9th - 12th Grade

46 questions
Minerals & Mining
Presentation
•
9th Grade - University

46 questions
Anatomical Direction, Planes & Cavities
Presentation
•
8th - 12th Grade

47 questions
Nomenclature of Ionic and Covalent
Presentation
•
9th - 12th Grade

47 questions
Ionic Compound Nomenclature
Presentation
•
9th - 12th Grade

47 questions
Valence electrons
Presentation
•
10th - 12th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Chemistry

25 questions
Waygrounds 5/20 PS.8.1.2
Quiz
•
9th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

20 questions
Naming & Writing Chemical Formulas
Quiz
•
10th Grade

15 questions
Types of chemical reactions
Quiz
•
9th - 12th Grade

32 questions
Acids/Bases, Electrochem, Orgo
Quiz
•
9th - 12th Grade