

Batteries
Presentation
•
Chemistry
•
9th - 12th Grade
•
Medium
+1
Standards-aligned
Susan Schroer
Used 10+ times
FREE Resource
11 Slides • 12 Questions
1
Where it all started.
The invention of the battery as we know it is credited to the Italian scientist Alessandro Volta, who put together the first battery to prove a point to another Italian scientist, Luigi Galvani. In 1780, Galvani had shown that the legs of frogs hanging on iron or brass hooks would twitch when touched with a probe of some other type of metal. He believed that this was caused by electricity from within the frogs’ tissues, and called it ‘animal electricity’.
2
3

Volta, believed that the electric current came from the two different types of metal. He stacked of layers of silver and zinc interspersed with layers of cloth or paper soaked in saltwater, and found that an electric current did in fact flow through a wire applied to both ends of the pile.
Alessandro Volta's Battery
4
a cathode (+)
an anode (-)
an electrolyte
The cathode (positive end) and anode (negative end) are hooked up to an electrical circuit
Batteries have 3 parts...

5
Multiple Choice
What two portions of a battery react to produce electrons?
Anode and Cathode
Cathode and Electrolyte
Anode and Electrolyte
Cathode and Electrons
6
an energy source
which provides the "push"; volts
the load
which converts electrical energy to another form of energy
the wires
which connect the source and load together
the switch
which opens and closes the circuit
Circuits have 4 parts...

7
2 Types of Batteries:
Dry Cell - traditional batteries; uses a paste electrolyte with enough moisture to allow current to flow
Wet Cell - electric battery device consisting of 2 or more electrochemical cells that convert chemical energy into electrical energy using a liquid electrolyte
Batteries and Electrical Circuits

8

the electrolyte is not really dry; it is a paste
these are your standard AA, C, and D type batteries
the (+) terminal is made of carbon
Dry Cell
9

the electrolyte is a liquid
example: car battery
the electrolyte here is sulfuric acid
Wet Cell
10
Multiple Choice
Which of the following choices represent the three components of a battery?
Anode, electrons, and wires
protons, electrons, and neutrons
Anode, cathode, and electrolyte
AC, DC, and Electrons
11
Schematic Diagrams are pictures that we can use to show circuits in a simple way.
they use symbols like the ones in the top right image to represent the parts of a circuit
Circuit Diagrams Practice


12
Multiple Choice
How many chemical reactions take place in a typical battery?
1
2
3
4
13

the circuit's pathway is broken
meaning the switch is open or a part of the circuit is disconnected and electrons cannot flow to the load.
has "breaks" in the circuit
Open Circuit (no light)
the circuit path is complete, meaning that the switch is closed and electrons are allowed to flow
each part of the circuit is connected
Closed Circuit (light)
Lightbulb
14
Schematic Diagrams are pictures that we can use to show circuits in a simple way.
they use symbols like the ones in the top right image to represent the parts of a circuit
Circuit Diagrams Practice


15
Multiple Choice
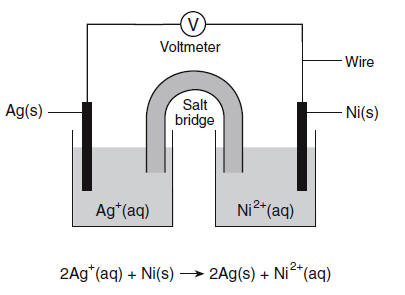
Ag+ + e- →Ag
Ag → Ag+ + e-
Ni2+ + 2e- → Ni
Ni → Ni2+ + 2e-
16
Multiple Choice

Magnesium atoms lose electrons
Mass of the iron electrode decreases.
Electrons flow from the iron half-cell to the magnesium half-cell.
Negative ion flow from magnesium half-cell to iron half-cell
17
Multiple Choice
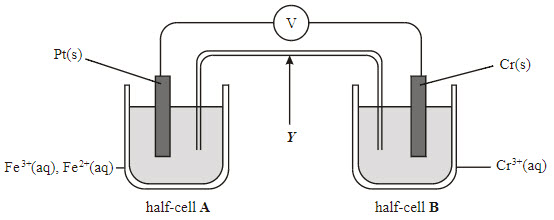
To complete the circuit
To allow flow of electrons between both cells
To allow flow of ions
To connect both half cells
18
Multiple Choice
anode
cathode
salt bridge
voltmeter
19
Multiple Choice
anode
cathode
20
Multiple Choice
Electrons flow from the cathode to the anode
Electrons flow from left to right
Electrons flow from anode to cathode
Electrons flow from right to left
21
Multiple Choice
In a rechargeable battery that is half used, what would be the conditions in the battery?
All of the electrons would be in the anode
Some of the electrons would be in the cathode and anode
The electrons would be found in the electrolyte
All of the electrons would be in the cathode.
22
Multiple Choice
What is the main difference between AC current and DC current?
the electrons in AC current flow completely through the circuit
the electrons in an AC circuit stay on one stationary place
the electrons in AC move back and forth, but not through the complete circuit
electrons are consumed by the circuit in AC but not DC
23
Multiple Choice
In a rechargeable battery, what happens when a dead battery is plugged into a charger and electrical outlet?
The electrons move back to the cathode
The chemical reactions in the battery reverse and recharge the battery
The electrons react with the lithium ions
The electrons react with the electrolyte
Where it all started.
The invention of the battery as we know it is credited to the Italian scientist Alessandro Volta, who put together the first battery to prove a point to another Italian scientist, Luigi Galvani. In 1780, Galvani had shown that the legs of frogs hanging on iron or brass hooks would twitch when touched with a probe of some other type of metal. He believed that this was caused by electricity from within the frogs’ tissues, and called it ‘animal electricity’.
Show answer
Auto Play
Slide 1 / 23
SLIDE
Similar Resources on Wayground

14 questions
Electron Math
Presentation
•
9th - 12th Grade

20 questions
Ionic Bonding
Presentation
•
9th - 12th Grade

18 questions
Quantitative vs Qualitative observations
Presentation
•
9th - 12th Grade

17 questions
Nuclear Decay
Presentation
•
9th - 12th Grade

17 questions
Gas Laws - Dalton's Partial Pressure
Presentation
•
9th - 12th Grade

21 questions
Chemical Reactions Intro
Presentation
•
9th - 12th Grade

21 questions
Elements, Compounds, Mixtures
Presentation
•
9th - 12th Grade

16 questions
Scientific Method
Presentation
•
9th - 11th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Chemistry

25 questions
Waygrounds 5/20 PS.8.1.2
Quiz
•
9th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

20 questions
Naming & Writing Chemical Formulas
Quiz
•
10th Grade

15 questions
Types of chemical reactions
Quiz
•
9th - 12th Grade

32 questions
Acids/Bases, Electrochem, Orgo
Quiz
•
9th - 12th Grade
