

IGCSE CIE Chemistry - Rates of reaction
Presentation
•
Chemistry
•
10th Grade
•
Practice Problem
•
Medium
MAD VLAD
Used 7+ times
FREE Resource
16 Slides • 16 Questions
1
Reaction rates
And Limiting factors
2

3

4
Replace this text with your body text.
Duplicate this text as many times as you would like.
Subheader text
Replace this text with your body text.
Duplicate this text as many times as you would like.
Subheader text
Exo-Endo thermic

5
Activation Energy
and Collision Theory
Activation Energy - minimum amount of energy colliding particles must have to react.
Collision theory -
particles must collide, with surface energy, at the proper orientation.
Higher number of collision, higher the reaction rate.

6

7
Multiple Choice
true
false
8
Multiple Choice
The energy needed for a reaction to occur
The minimum amount of energy needed for a reaction to potentially occur
The energy added by a catalyst
The energy possessed by the products
9
Multiple Choice
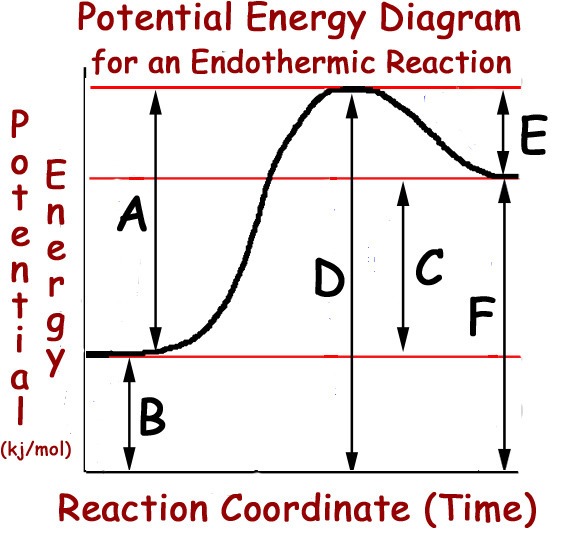
A
B
C
D
10
Multiple Choice
What is collision theory?
Molecules must collide in the correct orientation with enough energy to bond.
Molecules need enough energy to collide and react.
Atoms constantly collide and react.
The minimum energy needed for atoms to react
11
Multiple Choice
True
False
12
Multiple Choice
What is the rate of reaction?
How much energy is needed for a reaction to occur.
The energy required to break a bond.
The time it takes for a reaction to occur.
Collision Theory
13
14

15

16

17
Multiple Choice
What is the purpose of a catalyst?
Helps to slow down a reaction
Raises the activation energy
Lowers the activation energy
Is consumed by the reaction
18
Multiple Choice
What factors effect rate of reaction?
Temperature, Concentration, Pressure and Energy
Surface Area, Concentration, Energy and Pressure
Temperature, Pressure, Concentration and Surface area
Pressure, Surface area, Density and Energy
19
Multiple Choice
Increasing the temperature of your solution will.......
Not affect the rate of reaction.
Speed up the rate of reaction
Slow down the rate of reaction
20
Multiple Choice
Slow collision frequency
Allow less effective collision between the particles
Cause particles to lose speed
Increase collision between the particles thus increasing the rate.
21
Multiple Choice
concentration
surface area
temperature
reactants
22
Multiple Choice
catalyst
product
reactant
solute
23
Multiple Choice
it increases the amount of reactants
it lowers the activation energy
it increases the energy of particle collisions
it increases the frequency of particle collisions because there are more collision sites
24
Rates of Reaction
The rate of reaction is simply the speed at which a reaction takes place.
The units of the rate of reaction: g s-1 or cm3 / dm3 s-1, which can then be converted into mol dm3 s-1.
25
The word equation for the reaction of calcium carbonate with hydrochloric acid

26
The table shows results obtaining using this apparatus

27
The graph of the results is plotted
it is easier to see the pattern that they make.

28
29
Reaction Rates
How fast a reaction goes

30
Multiple Choice
Why does a higher temperature increase the rate of a reaction?
it increases both the frequency and energy of particle collisions
it only increases the frequency of particle collisions
it only increases the energy of particle collisions
it reduces the activation energy of the reaction
31
Multiple Choice
Grinding a seltzer tablet into powder increases the rate of reaction due to increased
concentration
surface area
temperature
reactants
32
Multiple Choice

The following graph shows two different reaction pathways for the same overall reaction at the same temperature. Which pathway is slower and why?
Red, because the activation energy is larger
Blue, because the activation energy is lower
both reaction progress at the same rate
Reaction rates
And Limiting factors
Show answer
Auto Play
Slide 1 / 32
SLIDE
Similar Resources on Wayground

30 questions
Know Your Lab Equipment
Presentation
•
10th Grade

22 questions
Nomenclature Review
Presentation
•
10th Grade

26 questions
Endo/Exothermic
Presentation
•
10th Grade

23 questions
Cell Cycle
Presentation
•
10th Grade

24 questions
Ions
Presentation
•
10th Grade

24 questions
Acids and Bases
Presentation
•
10th Grade

21 questions
Moles and Molar Mass
Presentation
•
10th Grade

25 questions
Leadership Styles
Presentation
•
10th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Interpreting Solubility Curves
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
10th Grade

43 questions
Acids and Bases Review
Quiz
•
10th - 11th Grade

20 questions
Thermochemistry Review
Quiz
•
10th Grade

25 questions
Unit 4 (Mid) Rumble
Quiz
•
10th Grade