

Thermochemistry
Presentation
•
Chemistry
•
10th Grade
•
Medium
Richard Leachman
Used 3+ times
FREE Resource
87 Slides • 16 Questions
1


UNIT 11 THERMODYNAMICS
1
2

ENERGY
• Energy is the measure of the ability to
cause change to occur (work)
• The property of an object that enables
it to do work
Units of energy:
Joule (J) = newton x meter
J = N x m
4
3

Types of Energy
Energy appears in many forms. There are five
main forms of energy:
Mechanical (Kinetic and Potential)
Chemical
Electromagnetic
Heat (Thermal)
Nuclear
5
4

Kinetic Energy
• Kinetic energy is energy of motion.
• Kinetic energy depends on both mass
and velocity.
• The faster the object moves- the more
kinetic energy
6
5

•As the temperature of a sample
of matter is increased, what
happens to the average kinetic
energy of the particles in the
sample?
•As the temperature of a sample
of matter is increased, the
average kinetic energy of the
particles in the sample increase.
7
6

Potential Energy
• The amount of energy that is
stored
• 3 types of potential energy
• Elastic
• Ex. Pulling a rubber band back and
holding
• Chemical
• Ex. Burning a match
• Gravitational
• A bolder resting on top of a hill
• Objects at high positions have greater
gravitational potential energy then
objects in lower positions
8
7

Chemical Energy
• Chemical energy is the energy stored in the bonds of
atoms and molecules.
• This a form of potential energy until the bonds are
broken.
• Fossil fuels and biomass store chemical energy.
Examples:
• Digesting food…bonds are
• broken to release energy for
• your body to store and use.
• • Sports… your body uses energy
• stored in your muscles obtained
• from food.
• • Fire–a chemical change.
9
8

Electromagnetic Energy
• a form of energy that is reflected or emitted
from objects in the form of electrical and
magnetic waves that can travel through space
• Moving electric charges
Examples:
• Power lines carry electricity
• Electric motors are driven by electromagnetic
energy
• Light is this form of energy (X-rays, radio
• waves, laser light etc.)
11
9

Thermal Energy
• The internal energy or thermal energy of a
substance is determined by the movement of the
molecules and the potential energy of the
arrangement of molecules.
• Temperature is the measure of the
average kinetic energy of the
molecules.
• Heat energy is the energy transferred
from a warmer substance to a
colder one by the collisions of
molecules.
12
10

Units of Thermal Energy
• The unit for all energy is the joule.
• However, sometimes the calorie is used for heat.
• The calorie is defined as the amount of heat needed to
raise 1 g of a substance 1 degree Celsius.
• A Calorie (food calorie, with a capital C) is 1000 cal
1 cal = 4.18 joules or 1kcal = 4180 J
To convert calories to
joules multiply the calories
by
4.18.
To convert joules to
calories
divide by 4.18.
13
11

•What happens to the energy
produced by burning gasoline in
a car engine?
15
12

•What happens to the potential
energy when gasoline is burned
in an automobile engine?
16
13

Nuclear Energy
• When the nucleus of an atom splits,
nuclear energy is released.
• Nuclear energy is the most concentrated
form of energy.
• Fission/fusion
21
14
Multiple Choice
The biggest heat source for our planet is the ______________________.
Earth
fire
Moon
Sun
15
Multiple Select

What happens to water if it gains heat?Choose 3 correct options.
It boils.
It freezes.
It changes its state.
It turns to steam.
It changes its property.
16

Law of Conservation of
Energy and Heat Transfer
Chem.11B Understand the law of
conservation of energy and the
processes of heat transfer.
22
17

Law of Conservation of
Energy
• Conservation of energy (1st
law of thermodynamics) is
one of several conservation
laws.
• It states that the total inflow
of energy into a system must
equal the total outflow of
energy from the system
• In other words, energy can
be converted from one form
to another, but it cannot be
created or destroyed.
23
18

Heat Energy
• The law of conservation of energy is
also true of heat energy.
• If a substance gets hotter
something else must get colder.
heatlost = heatgained
24
19

•If heat is produced by a
chemical system, what will
happen to its surroundings?
20

•When energy is changed from
one form to another, can all of
the energy still be accounted for?
26
21
Multiple Choice

Rahma got an ice cream from her mom. The ice cream melts under the sun because the ice cream ___________.
gains heat from the sun
loses heat to the sun
produce too much heat
22

Relating Temperature to
Energy Transfer as Heat
•Heat, q, is energy that transfers from
one object to another because of a
temperature difference.
•The transfer of energy always takes
place from a substance at a high
temperature to a substance at a lower
temperature.
27
23

• Example : You are holding a hot water bottle
what will happen:
• The warmer object (hot water bottle)
will transfer energy to the cooler
object (your hand).
•When energy is transferred as heat,
the temperature of the water falls
while the temperature of your skin
rises.
•The great the difference in
temperature of the two object, the
more energy that will be transferred.
•This explains why hot things always
cool down.
28
24

• As an object becomes hotter, what
happens to the rate of heat transfer
from the object to a cooler body with
which it is in contact?
• As an object becomes hotter,the rate
of heat transfer from the object to a
cooler body with which it is in contact
becomes faster.
29
25

•What happens to boiling
water if more heat is added?
30
26

• How does the amount of heat
absorbed by a melting solid
compare to the amount of heat lost
by a solidifying liquid?
33
27
Multiple Choice
Heat transfer is also called...
Thermal Energy
Conduction
Convection
Radiation
28
Multiple Choice
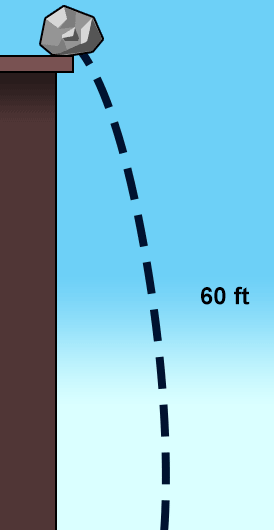
The rock in the picture is pushed over the edge and it begins to fall. Which type of energy conversion is taking place?
mechanical to heat
sound to mechanical
potential to kinetic
kinetic to potential
29
Multiple Choice
Maria’s father started a fire in the fireplace. He crumpled some paper, lit a match, and soon the logs in the fireplace were burning. In this case, the stored chemical energy in the logs was changed into
electrical energy.
mechanical motion.
heat and light energy.
electrical and heat energy.
30

Let’s Look @ Temperature
• The internal energy or thermal energy of a substance is
determined by the movement of the molecules and the
potential energy of the arrangement of molecules.
• Temperature measures the average kinetic energy of
the particles in a sample of matter
(Kinetic Energy = ½ mv2).
• The greater the kinetic energy (the faster the molecules
are moving), the higher the temperature, and the hotter
it feels. When the kinetic energy decreases (molecules
slow down), the temperature decreases.
• A substance can change in temperature due to heat
transfer.
34
31

Measuring Temperature
• Thermometers are device that is
used to measure kinetic energy
not temperature.
• Thermometers rely on a simple
physical property of all
substances
• MOST OBJECTS EXPAND WHEN THEIR
TEMPERATURE INCREASES
• Thermometers use liquids substance
like mercury and colored alcohol
that expand as their temperatures
increase and contract as
temperature decreases
35
32

Temperature Scales
• Fahrenheit Scale
• Most familiar to you from your friendly weather reports
• Units called DEGREES FAHRENHEIT [ °F]
• Water freezes at 32 °F and Boils at 212 °F
• Celsius Scale
• Widely used in science and other countries
• Units called DEGREES CELSIUS [°C]
• Celsius scale is based the values of 0 °C to freezing point of
water and a value of 100 °C to boiling point of water (at
standard pressure)
• Kelvin Scale
• Based on absolute zero the temperature at which an objects
energy is minimal
• Units called KELVIN [K]
• On the Kelvin scale zero Kelvin is absolute zero
36
33

Methods of Energy Transfer
• Energy transfer as heat from a hot object can
occur in 3 ways
Conduction
Convection
Radiation
• Heat transfer will stop when thermal equilibrium
is reached, that is the rate at which energy flows
out of a substance equals the rate that energy
flows into the substance.
37
34

Conduction
• The transfer of energy as heat between
particles as they collide with a substance or
between 2 objects in contact
• Energy transfer through solids
• Example: Heating marshmallows with a metal
rod, as the marshmallow cook, the wire you
are holding is getting hotter.
38
35

Convection
• The transfer of energy by the
movement of fluid with
different temperature
• During convection, energy is
carried away by a heated gas or
liquids that expand and rises
above cooler, denser gas or
liquid
• Energy transfer through gases
and liquids (both fluids)
• The cycle of a heated fluid that
rises and then cools and fall is
called convection current
39
36

Radiation
• The transfer of energy by electromagnetic
waves
• Energy transfer that does not need any
material to transfer to, it travels in waves
• Example: You stand near the heat of the fire
and feel the heat, energy is transferred as
eat from the fire in this case in the form of
electromagnetic waves
• Radiation differs from conduction and
convection in that it does not involve the
movement of matter
40
37

Review Main Points
• The law of conservation of energy: energy
cannot be created or destroyed. It can only
be transferred from one form to another.
• Heat is the transfer of energy from the
particles of one object to those of another
object due to temperature difference
between the two objects.
• Also remember that, transfer of energy
always takes place from a substance at a
higher temperature to a substance at a lower
temperature
• Three methods of energy transfer:
conduction, convection and radiation
41
38
Multiple Choice
When heat energy moves in a solid it is called....
Conduction
Convection
Radiation
39
Multiple Choice
Walking without shoes on a hot day is an example of...
Conduction
Convection
Radiation
40
Multiple Choice
When heat energy moves in a liquids or gases it is called...
Conduction
Convection
Radiation
41
Multiple Choice
Boiling water is an example of...
Conduction
Convection
Radiation
42
Multiple Choice

This picture is an example of...
Conduction
Convection
Radiation
43
Multiple Choice

This picture is an example of...
Conduction
Convection
Radiation
44

THERMOCHEMICAL
EQUATIONS
42
C.11.C use thermochemical equations to
calculate energy changes that occur in chemical
reactions and classify reactions as exothermic
or endothermic
45

• Endothermic and
Exothermic Processes
• Essentially all chemical
reactions and changes in
physical state involve
either:
a) release of heat, or
b) absorption of heat
43
46

• In studying heat changes, think of
defining these two parts:
•the system - the part of the universe
on which you focus your attention
•the surroundings - includes
everything else in the universe
• Heat - represented by “q”, is energy that
transfers from one object to another,
because of a temperature difference
between them.
•only changes can be detected!
44
47

•Heat flowing into a system from
it’s surroundings:
•defined as positive
•q has a positive value
•called endothermic
•system gains heat (gets
warmer) as the surroundings
cool down
45
48

•Heat flowing out of a system
into it’s surroundings:
•defined as negative
•q has a negative value
•called exothermic
•system loses heat (gets
cooler) as the surroundings
heat up
46
49

Potential energy graph: a closer look
d
Activated complex /
transition state
A + B
Products
(Potential energy stored in
chemical bonds)
Path of reaction
c
DH
a
e
C + D
b
f
A + B C + D + Energy
Reactants
50

•
What are the general equation
forms of Endothermic and
Exothermic reactions?
Exothermic: A + B -> C + D + heat
Endothermic: A + B + heat -> C + D
48
51
Multiple Choice

What type of reaction is this?
Endothermic
Exothermic
Allergic
52
Multiple Choice

What type of reaction is this?
Endothermic
Exothermic
Allergic
53

Thermochemical Equations
• A Thermochemical Equation is a balanced
stoichiometric chemical equation that includes the
enthalpy change, ΔH.
• Enthalpy (H) is the transfer of energy in a
reaction (for chemical reactions it is in the form
of heat) and ΔH is the change in enthalpy.
•By definition, ΔH = Hproducts –
Hreactants
•Hproducts < Hreactants, ΔH is negative
•Hproducts > Hreactants, ΔH is positive
49
54

Thermochemical Equations
•In working with thermochemical
equations you will find the following
rules helpful.
•When a thermochemical equation is
multiplied by a factor, the value of
H for the new equation is obtained
by multiplying the value of H by the
same factor.
•When a chemical equation is
reversed, the sign of H is reversed.
51
55

Writing Thermochemical Equations
• Thermochemical equations show the
exchange of heat in a chemical reaction.
•For example, Burning one mole of wax
releases 20,000 kJ of heat energy.
•This could be written as:
−C40H82 + 60.5 O2 → 40 CO2 + 41
H2O + 20,000 kJ
•Instead we usually write:
−C40H82 + 60.5 O2 → 40 CO2 + 41
H2O ΔH = -20,000 kJ
52
56

53
▫ Reacting 2 moles of solid sodium with 2
moles of water to produce 2 mole of aqueous
sodium hydroxide and 1 mole of hydrogen
gas will release 367 kJ of energy
▫ 2Na (s) + 2 H2O (l) → 2 NaOH (aq) + H2 (g) +
367 kJ or
▫ 2Na (s) + 2 H2O (l) → 2 NaOH (aq) + H2 (g)
∆H=- 367 kJ
Write the following thermochemical equations
showing ∆H.
57

▫ 184.6 kJ of energy is needed to
produce 1 mole of hydrogen gas and 1
mole of chlorine gas from 2 moles of
hydrogen chloride gas.
▫ 2 HCl (g) + 184.6 kJ → H2 (g) + Cl2 (g)
or
▫ 2 HCl (g) → H2 (g) + Cl2 (g) ∆H= +
184.6 kJ
54
58

• The Standard Heat of Formation is
defined as the change in enthalpy
(temperature) when one mole of a
substance in the standard state (1
atm or 101 kPa of pressure and
25°C) is formed from its pure
elements under the same
conditions. = DHf
55
Thermochemical equations using
Standard Heat of Formations
59

• What are the standard conditions of
temperature and pressure for a
thermochemical equation?
• The standard conditions of temperature
and pressure for a thermochemical
equation are 25° C and 101kPa.
• What is the amount of heat released by
the complete burning of 1 mole of a
substance known as?
• Heat of Combustion
56
60

C2H2(g) + 2 H2(g) → C2H6(g)
• Information about the substances
involved in the reaction represented
above is summarized in the following
tables.
Substance DH°f
(kJ/mol)
C2H2(g) 226.7
C2H6(g) -84.7
57
61

Thermochemical equations using
Standard Heat of Formations
Write the equation for the heat of
formation of C2H6(g)
Substance DH°f
(kJ/mol)
C2H2(g) 226.7
C2H6(g) -84.7
1st: Using our balanced chemical
equation, we see how many moles of each
compound we have.
C2H2(g) + 2 H2(g) → C2H6(g) [(H2) does not have a
DH°f ]
1 mol of C2H2(g) and 1 mol C2H6(g)
58
62

2nd: We plug in the ∆H°f for
each of our compounds,
remembering that
∆H° = [∆H°f products] – [∆H°f
reactants]
∆H° = [C2H6(g)] – [C2H2(g)] =
3rd: We solve for ∆H°
∆H° = [-84.7] – [226.7] = -
331.4 kJ/mol
59
63

Practice Problems
• Solve for the ΔHrx and
write the following
thermochemical
equations.
1. What is the ΔHrx for the
process used to make lime
(CaO)?
•CaCO3(s) → CaO(s) + CO2(g)
60
Substance
DH°f
(kJ/mol)
CaCO3(s)
-1207.6
CaO(s)
-634.9
C 4H10 (g)
-30.0
H2O (g)
-241.82
CO2 (g)
-393.5
2. What is the ΔHrx for the combustion of C4H10(g)?
2 C4H10 (g) + 13 O2 (g) → 10 H2O (g) + 8 CO2 (g)
64

Practice Problems
• Solve for the ΔHrx and write the
following thermochemical equations.
• 1. What is the ΔHrx for the process
used to make lime (CaO)?
• CaCO3(s) → CaO(s) + CO2(g)
61
Substance
DH°f
(kJ/mol)
CaCO3(s)
-1207.6
CaO(s)
-634.9
C 4H10 (g)
-30.0
H2O (g)
-241.82
CO2 (g)
-393.5
ΔHrx = [ΔH°f (CaO) + ΔH°f (CO2)] – [ΔH°f
(CaCO3)]
ΔHrx = [(-634.9)+(-393.5)] – [(-1207.6)]
ΔHrx = [ -1028.4] – [-1207.6] = +179.2 kJ
CaCO3(s) → CaO(s) + CO2(g) ΔHrx =
179.2 kJ/mol
65

Thermochemical & Endothermic/
Exothermic equations
• In the previous slides, we saw how ΔH° could
be both positive or negative.
• Depending on the sign of ΔH°, the reaction
can either be exothermic or endothermic.
• Exothermic reactions release heat from the
system to the surroundings so the
temperature will rise.
•ΔH° will be negative because the reaction loses
heat.
•ΔH° can be written into the chemical equation
as a product.
63
66

•Endothermic reactions absorb
heat from the surroundings into
the system so the temperature
will decrease.
•ΔH° will be positive because the
reaction absorbs heat.
•ΔH° can be written into the
chemical equation as a reactant.
64
67

Classify the following as endothermic
or exothermic
• Ice melting
• 2 C4H10(g) + 13 O2(g) → 10 H2O(g) + 8 CO2(g) ΔHrx
= -5506.2 kJ/mol
• 2 HCl (g) + 184.6 kJ → H2 (g) + Cl2 (g)
• Water vapor condensing
65
68

Exothermic vs. Endothermic
EXOTHERMIC
§ A change in a chemical
energy where
energy/heat EXITS the
chemical system
§ Results in a decrease
in chemical potential
energy
§ ΔH is negative
ENDOTHERMIC
§ A change in chemical
energy where
energy/heat ENTERS
the chemical system
§ Results in an increase
in chemical potential
energy
§ ΔH is positive
66
69

• In an exothermic reaction, the energy
stored in the chemical bonds of the
reactants is______.
• In an exothermic reaction, the energy
stored in the chemical bonds of the
reactants is greater than the energy
stored in the bonds of the products.
• A process that absorbs heat is a(n)
____________ process.
• Endothermic
67
70

•If you were to touch the flask in
which an endothermic reaction
were occuring, what would the
flask feel like?
68
71

•Is the vaporization of a liquid an
exothermic process or
endothermic process?
•If the heat involved in a
chemical reaction has a negative
sign, what happens to the
surroundings?
69
72

SPECIFIC HEAT: THE
EQUATION
70
C.11.D perform calculations involving heat,
mass, temperature change, and specific heat
73

Temperature and Energy
•We relate energy and temperature
by discussing a substance’s heat
capacity.
• Heat Capacity = heat required to
raise temp. of an object by 1oC
•Depends on both the object’s
mass and its chemical
composition
•
71
74

•Specific Heat Capacity
•A physical property of matter
• “Specific Heat Capacity (abbreviated “C”)
- the amount of heat it takes to raise the
temperature of 1 gram of the substance
by 1 oC
•often called simply “Specific Heat”
• Depends on both the object’s mass and
its chemical composition
72
75

Example
• If you have ever touched the
metal on a car and the fabric on
the car seat on a hot day, you
have experienced the affect of
specific heat. The metal seems
much hotter than the fabric seat
even if after receiving the same
amount of energy from the sun.
This is caused by the difference
in the specific heat of each of
the materials. The metal has a
lower specific heat and gives up
its thermal energy at a much
higher rate than does the fabric
which has a much higher
specific heat.
73
76

High Specific Heat and Water
• Water has a very high specific heat
compared to other matter; therefore
ocean water stays about the same
temperature throughout day and night
despite the differences in temperature
between night and day. That also
explains why water is used in car
radiators to cool the engine.
• Low specific heat = less energy required to
change the temperature
• High specific heat = more energy required to
change the temperature
74
77

Practice
Which would get hotter if left in the
sun?
• Penny vs. Water
• Keys vs. soccer ball
• Plastic recycling bin vs. metal trash
can
75
78

Specific Heat Capacity
• Temperature change of a substance
depends on three things:
• Mass, m
• Amount of energy added, Q
• Specific Heat, Cp
76
Final
temperature
Initial
temperature
Temperature
change
79

Specific Heats
of Common
Substances
77
Note the
tremendous
difference
in Specific
Heat.
Water’s
value is
VERY
HIGH.
80

• If you want to cool a hot drink, it is best
to use a spoon with a relatively
________ specific heat.
• What 3 things does the amount of heat
transferred from an object depend on?
• The 3 things the amount of heat
transferred from an object depend on
are the specific heat of the object, the
initial temperature of the object, and the
mass of the object.
78
81

Using Q = m x Cp x (Tf – Ti)
The following problems will show you how to solve
for different variables in our equation.
• How much energy does it take to
raise the temperature of 50 g of
aluminum (cp = 0.9025 J/gC0) by 10
0C?
79
Q = (50g) (0.9025 J/gC0) (100C)
Q = (m) (cp) (Tf - Ti)
Q = 451.25 Joules
82

Using Q = m x Cp x (Tf – Ti)
• If we add 30 J of heat to lead (cp =
0.1276J/gC0) with a mass of 10 g,
how much will its temperature
increase?
80
Q = (m) (cp) (Tf - Ti)
30J = (10g) (0.1276 J/gC0) (x)
30J = (1.276 J/0C) (x)
23.50C = x = temperature increase
83

• What is the amount of heat required to
raise the temperature of 200.0 g of
aluminum by 10° C?
• (specific heat of aluminum = 0.21
cal/g x °C)
• Q = (m) (cp) (Tf - Ti)
• Q = 200.00 g x 0.21 cal/g x °C x
10 °C
• Q = 420 cal
81
84

• What is the specific heat of a
substance if 1560 cal is required to
raise the temperature of a 312 g
sample by 15°C?
• Q = (m) (cp) (Tf - Ti)
• Cp = Q
(m) (Tf - Ti)
C = 1560 cal
C = 0.33 cal/g x °C
(312 g) (15°C)
82
85
Multiple Choice
Calculate A 50g sample is heated with 500 joules of energy and has a 10*C temperature change. Calculate it’s specific heat.
1 j/g*C
250000 j/g*C
500 j/g*C
10 j/g*C
86
Multiple Choice
4180 j of heat energy are added to change the temperature of water by 10*C. How many grams of water are being heated? cwater=4.18 j/g*C
4.18 j/g*C
10000g
174724g
100g
87

• By what quantity must the heat
capacity of an object be divided to
obtain the specific heat of that
material.
• Q = (m) (cp) (Tf - Ti)
• Its mass
• The heat capacity of an object
depends in part on its _______.
• Its mass
85
88

• Two objects are sitting next to each
other in direct sunlight. Object A gets
hotter than Object B. Compare the
specific heats of the two.
• Object A has a lower specific heat than
object B.
• How does the specific heat of a 100 g
sample of Iron, compare to a 10 g
sample of Iron?
• They have the same specific heat
because they are the same metal.
86
89

• When 45 g of an alloy is dropped
into 100.0 g of water at 25°C, the
final temperature is 37°C. What is
the specific heat of the alloy?
• Q = (m) (cp) (Tf - Ti)
• You use the formula ΔQ = ΔT*mc.
The heat gained by the water equals
the negative of the heat lost by the
alloy: ΔQ(water) = -ΔQ(alloy); so
ΔT(w)*m(w)*c(w) = -ΔT(a)*m(a)*c(a)
87
90

• ΔT(w)*m(w)*c(w) = -ΔT(a)*m(a)*c(a)
What we want is c(alloy):
C(a) = -[ΔT(w)*m(w)*c(w)] / [ΔT(a)*m(a)]
The specific heat of water is 4.184
J/g·°C, and there is 100 g of water.
• There is 45 g of alloy.
The ΔT(w) = Tf(w) - Ti(w) = 37.0°C -
25.0°C = 12.0°C
Let's plug it in:
c(a) = -[ΔT(w)*m(w)*c(w)] /
[ΔT(a)*m(a)]
88
91

• The ΔT(a) = Tf(a) - Ti(a) = 37.0°C -
100.0°C = -63.0°C
Let's plug it in:
c(a) = -[ΔT(w)*m(w)*c(w)] /
[ΔT(a)*m(a)]
c(alloy) = -[(12.0°C)*(100.0 g)*(4.184
J/g·°C)] / [(-63.0°C)*(45.0 g)] = 1.77
J/g·°C (note three sig figs).
89
92

• The specific heat capacity of silver is
0.24 J/g x °C. How many joules of
energy are needed to warm 0.500 g
of silver from 25.0°C to 27.5°C?
• Q = (m) (cp) (Tf - Ti)
• Q = .500 g x .24 x 2.5
• q=mcdeltaT q=(4.37)(0.24g)(25.0
degrees C-27.5 degrees C)= -2.62 J
91
93

Calorimetry
C.11.E use calorimetry to calculate the heat
of a chemical process
92
94

Calorimetry
• Calorimetry is the science of measuring
the heat of chemical reactions or
physical changes.
• Calorimetry is also known as a laboratory
procedure that measures the amount of heat
transferred to the surroundings by a reaction.
•Calorimetry can be calculated when heat of
combustion is given and the mass of the
substance is known or,
•During a calorimetry procedure, the heat
released during a chemical or physical change
is transferred to another substance, such as
water, which undergoes a temperature change.
93
95

The Mole and Energy Transfer
• Molar heat of fusion is the amount
of energy needed to change one
mole of a substance from the solid
phase to the liquid phase at
constant temperature and pressure.
• Heat of combustion is the amount
of heat released by the complete
burning of 1 mole of substance.
94
96

• Heat changes can occur when a
substance melts, solidifies, dissolves, or
when a substance vaporizes.
• During a phase change (solid to liquid,
liquid to vapor and back, the
temperature remains constant.
• To calculate the amount of heat
absorbed as a substance melts the
information needed is the mass of the
substance, the specific heat of the
substance, and the change in
temperature.
95
97

• When do heat changes occur? (4 of
them)
• When a substance dissolves, melts,
solidified or vaporizes.
• What information is needed to
calculate the amount of heat
absorbed as a substance melts? (3
things)
• The mass, specific heat, and change
in temperature of the substance.
97
98

Calorimetry Calculations
• Example 1: Propane is a commonly
used fuel. 1 mol of C3H8 releases
2,220 kJ of heat during combustion.
The molar mass of C3H8 is 44.1 g/mol.
How much heat is released if a
firework contains 67.8 g of C3H8?
98
¡ 2nduse the heat of combustion of propane to calculate
energy (heat) released
÷ 1.53 mol C3H8 x 2,220 kJ = 3413.06 kJ =>3410 kJ released
1 mol
¡ 1stconvert the grams of C3H8 to moles of C3H8.
÷ 67.8 g C3H8 x 1 mol C3H8 = 1.53 mol C3H8
44.1 g C3H8
99

Calorimetry Calculations
• The temperature change, fuel mass,
and water volume data from a
calorimetry procedure can be used
to determine how much heat is
transferred during a combustion
reaction.
• The amount of energy transferred from a
substance during combustion depends on the
identity and mass of the substance.
• The equation can be seen as q1 = - q2. One will
be losing energy, the other will be gaining
energy.
99
100

Calorimetry Calculations
• Example 2: 175 grams of hot aluminum (100.°C) is
dropped into an insulated cup that contains 40.0 mL
of ice cold water (0.0°C). Follow the example above
to determine the final temperature, x.
100
¡ 1stset up expressions for energy released and energy
absorbed.
÷ Q = - (175 g) (0.900 J/g●◦C) (x -100 ◦C) for silver and Q = (40.0 g) (4.184
J/g●◦C) (x -0.0 ◦C) for cold water
¡ 2ndput expressions together.
÷ - (175 g) (0.900 J/g●◦C) (x -100 ◦C) = (40.0 g) (4.184 J/g●◦C) (x -0.0 ◦C)
¡ 3rdsolve for x.
÷ - 157.5 (x – 100) = 167.4 (x - 0.0)
÷ - 157.5 x + 1575 = 167.4 x
÷ 1575 = 324.9 x => x = 48.5◦C
101

• On what principle does calorimetry
depend?
• Law of Conservation of Energy
• What do you need to do to
determine the heat change for a
reaction in an aqueous solution?
• You can mix the reactants in a
calorimeter and measure the
temperature change.
101
102

• What is the amount of heat needed to
melt one more of a solid called?
• Molar heat of fusion.
• When do heat changes occur? List 4.
• Heat changes occur when a substance
dissolves, a substance melts, a
substance solidified, a substance
vaporizes.
102
103

• Hess’s Law of Heat Summation
• This law makes it possible to
calculate DH for complicated
chemical reactions.
• If you add two or more
thermochemical equations to give a
final equation, then you can also
add the heats of reaction to give the
final heat of reaction.
103


UNIT 11 THERMODYNAMICS
1
Show answer
Auto Play
Slide 1 / 103
SLIDE
Similar Resources on Wayground

100 questions
C1: States of Matter
Presentation
•
9th Grade

99 questions
Continuity and Unity of Life
Presentation
•
10th - 12th Grade

100 questions
汉语(1)
Presentation
•
KG

102 questions
S2 L2 Vocab Intro
Presentation
•
10th Grade

99 questions
self guided periodic table intro
Presentation
•
11th Grade

97 questions
APHG Unit 3 Review Lesson
Presentation
•
9th Grade

104 questions
Angiosperms
Presentation
•
11th Grade

101 questions
CRM 1.2 Text 1 Master Lesson
Presentation
•
10th Grade
Popular Resources on Wayground

20 questions
"What is the question asking??" Grades 3-5
Quiz
•
1st - 5th Grade

20 questions
“What is the question asking??” Grades 6-8
Quiz
•
6th - 8th Grade

10 questions
Fire Safety Quiz
Quiz
•
12th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

34 questions
STAAR Review 6th - 8th grade Reading Part 1
Quiz
•
6th - 8th Grade

20 questions
“What is the question asking??” English I-II
Quiz
•
9th - 12th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

47 questions
8th Grade Reading STAAR Ultimate Review!
Quiz
•
8th Grade
Discover more resources for Chemistry

15 questions
Balancing and Reaction Types
Quiz
•
10th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

16 questions
STAAR Review Quizziz 5
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

5 questions
DOL REC: pH and pOH Calculations
Quiz
•
10th - 11th Grade

20 questions
Naming & Writing Chemical Formulas
Quiz
•
10th Grade

5 questions
DOL PreAP: pH and pOH Calculations
Quiz
•
10th Grade

13 questions
IP PREAP: pH and pOH Calculations
Quiz
•
10th - 11th Grade