

periodic table review
Presentation
•
•
Practice Problem
•
Medium
Mr. Jackson
Used 5+ times
FREE Resource
20 Slides • 30 Questions
1
Intro to Atoms & The Periodic Table
2
An Element
The Simplest form of a pure substance

3
The Atom
The smallest particle of an element that retains the chemical properties of an element.

4
The atomic mass unit (amu) is used to mass atoms and equals 1.67 x 10-27 Kg

5
Atom Composition
The atom is composed of three subatomic particles
Particles which are smaller than the atom

6

Proton
Charge: +1
Mass: 1 amu
Location: In Nucleus
7

Electron
Charge: -1
Mass: 1/1836 amu ~ 0 amu
Location: Orbits nucleus in electron cloud
8
Neutron
Charge: 0 (neutral)
Mass: 1 amu
Location: In nucleus

9

10
Multiple Choice

What is the charge of a proton
+1
0
-1
11
Multiple Choice

What is the charge of a Neutron?
+1
-1
0
12
Multiple Choice

Where is the electron located?
The nucleus
The orbitals of the atom
Inside a neutron
13
The Periodic Table
The periodic table tells us important information about elements and atoms that make up elements.
14
Atomic Number
The elements are numbered with whole numbers in order.

15
Atomic Number & Protons
Atomic number always equals the number of protons in an atom.
This means that the protons are also used as the ID of an element
16
Multiple Choice

1
3
4
7
17
Multiple Choice

14
7
15
18
18
Atomic Mass
The other number found on the periodic table is the atomic mass
The mass of an atom of the element

19
Subatomic Particles & Atomic Mass
Because the mass of an electron is so small
We can assume that the atomic mass is equal to the number of protons and neutrons.
20
Mass Number
The mass number is the atomic mass rounded to the nearest whole number
21
Multiple Choice

What is the atomic number of Cu?
63.546
29
2
34
22
Multiple Choice

What is the mass number of Neon?
20.180
20
10
23
Match

A
B
C
D
E
Proton
Neutron
Electron
Shell
Nucleus
Proton
Neutron
Electron
Shell
Nucleus
24
25
Multiple Choice
Periods are the _______ on the periodic table.
rows
columns
26
Multiple Choice
The period number tells you the number of _______ that atom has.
energy levels
valence electrons
protons
neutrons
27
Multiple Choice

Based on the image below, this element should be in period
1
2
3
4
28
Multiple Choice
Groups are the _________ on the periodic table.
rows
columns
29
Multiple Choice
The group number tells you the number of ________ that atom has.
valence electrons
energy levels
electron shells
protons
30
Multiple Choice

Based on the image below, this element should be in group
2
4
6
8
31
Multiple Choice

By looking at this picture, you can tell that these elements are both in group
1 (1 valence electron)
2 (2 valence electrons)
3 (3 valence electrons)
4 (4 valence electrons)
32
Summary
# of protons = Atomic number
# of electrons = # of protons = Atomic number
# of neutrons = mass # - Atomic #
* Mass #- rounded atomic mass (whole #)
33
Multiple Choice
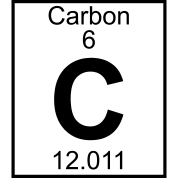
atomic mass
atomic number
element name
chemical symbol
34
Multiple Choice
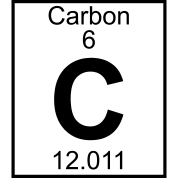
atomic mass
atomic number
element name
chemical symbol
35
Multiple Choice
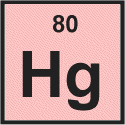
Helium
Rubidium
Hydrogen
Mercury
36
Multiple Choice

Lithium
Oxygen
Hydrogen
Francium
37
Multiple Choice

The fluorine atom has an atomic number of
19
9
38
Multiple Choice

What is the mass number and atomic number ?
Mass number = 7, atomic number = 14
Mass number = 14, atomic number = 7
39


40
Labelling
Match the following subatomic particles to their correct place in the atom.
41
Match
Match the following subatomic particles to their correct charge.
Proton
Neutron
Electron
Positive
Neutral
Negative
Positive
Neutral
Negative
42

43

Ionic vs. Covalent Bonding

44
Multiple Choice
A bond between a metal and a nonmetal is called a(n)
covalent bond
ionic bond
metallic bond
transfer bond
45
Multiple Choice
A bond between a nonmetal and a nonmetal is called a(n)
covalent bond
ionic bond
metallic bond
transfer bond
46
Multiple Choice
Calcium Oxide, CaO
Covalent Bond
Ionic Bond
47
Multiple Choice

The transfer of Valence electrons from a metal to a non metal is known as
Covalent Bonding
Metallic bonding
Molecular bonding
Ionic Bonding
48
Multiple Choice

Share electrons
transfer electrons
None
conduct electricity
49
Multiple Choice
Identify the following compound as ionic or covalent: CF4
ionic
covalent
50
Multiple Choice
Identify the following compound as ionic or covalent: MgO
ionic
covalent
Intro to Atoms & The Periodic Table
Show answer
Auto Play
Slide 1 / 50
SLIDE
Similar Resources on Wayground

45 questions
Elements of Poetry
Presentation
•
6th - 9th Grade

46 questions
OST Grade 5 Science Year in Review
Presentation
•
5th Grade

46 questions
Matrices; 11.2
Presentation
•
9th - 12th Grade

46 questions
Exploring Angle Pairs
Presentation
•
10th Grade

45 questions
Commonly Confused Words
Presentation
•
10th - 11th Grade

45 questions
Synthetic Materials Tutorial
Presentation
•
9th - 12th Grade

45 questions
Inherited and Aquired behavior
Presentation
•
5th Grade

45 questions
Gregor Mendel
Presentation
•
7th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for

6 questions
FOREST Generosity
Presentation
•
KG

21 questions
Star Wars
Quiz
•
KG - 12th Grade

16 questions
3D Shapes
Quiz
•
KG - 1st Grade

10 questions
Word Knowledge and Skills #3
Quiz
•
KG - 8th Grade

24 questions
5th Grade Math EOG Review
Quiz
•
KG - University

73 questions
NWEA MATH Practice 161-170
Quiz
•
KG - 3rd Grade

13 questions
Time
Quiz
•
KG - 2nd Grade

12 questions
Alliteration Practice
Quiz
•
KG - 5th Grade