

Phy Y11 C22 C23 IGCSE Atom Model Radioactivity Nuclear Reaction
Presentation
•
Physics
•
9th - 12th Grade
•
Medium
Standards-aligned
Kewin Aljoe
Used 7+ times
FREE Resource
41 Slides • 105 Questions
1
Atom Model
radioactivity
Nuclear Decay


2
3
J.J. Thomson (1897 / England)
Known For: electrons were smaller particles of an atom and were negatively charged.
Experiment:
Cathode Ray Tube Experiment
Visual Representation


4
5
Ernest Rutherford (1911 / England)
Known For: Protons (positive charged) and atoms were mostly empty space
Experiment:
Gold Foil Experiment
Visual Representation



6
Vocabulary
Alpha particles (a)
are composite particles consisting of two protons and two neutrons tightly bound together
7
8
Multiple Select
Rutherford's experiment providing evidence that:
a very small nucleus surrounded by mostly empty space
a nucleus containing most of the mass of the atom
a nucleus that is positively charged
a very small nucleus surrounded by electrons
9
Niels Bohr (1913 / England)
Known For: Energy levels of electrons
Electrons can jump across energy levels
Experiment:
Emissions Spectrum (color of light)
Visual Representation
"Planetary model"


10
11
Erwin Schrödinger (1926 / Austria)
A.K.A.: Modern Model of the Atom
Known For: Quantum mechanics
electrons can only exist in specified energy states
Experiment:
mathematical equations
Visual Representation
"Electron Cloud model"

12
James Chadwick (1932 / England)
Known For: Discovered neutrons
neutral particles in the nucleus of an atom
Experiment:
mathematical equations (atom was too heavy with just protons)
Visual Representation
"Quantum model"


13
| | | | |
|---|---|---|---|---|
| | | | |
| | | | |
| | | | |
Summary of what is known about the current model of the atom:
Subatomic Particles | Symbol | Relative charge | Relative mass (amu) | Actual mass |
|---|---|---|---|---|
electrons | e | -1 | 1/1840 | 9.11 X 10 |
protons | p | +1 | 1 | 1.67 X 10 |
neutrons | o | 0 | 1 | 1.67 X 10 |
14
Nuclide notation is a way of showing the atomic number and mass of an atom.
We can use the nuclide notation to work out the number of protons neutrons and electrons.
N
7
14
Mass number
Atomic Number
15

16
Explanation Slide...
The proton number, also known as the atomic number, indicates the number of protons in a nucleus. For this nuclide, the correct proton number is 10, which corresponds to the element neon.
17
Multiple Choice

What is the proton number of a nucleus of this nuclide?
10
12
22
32
18
Explanation Slide...
A neutral atom of radium-226 has 88 electrons, which equals its atomic number. The atomic number indicates the number of protons and, in a neutral atom, the number of electrons is equal to the number of protons.
19
Multiple Choice

How many electrons orbit the nucleus of a neutral atom of radium-226?
0
88
138
226
20
Multiple Choice
Positive ion formed by
gaining proton (s)
losing proton (s)
gaining electron (s)
losing electron (s)
21
Multiple Choice
Negaitive ion formed by
gaining proton(s)
losing proton(s)
gaining electron(s)
losing electron(s)
22
Multiple Select
nucleon consist of
proton
electron
neutron
nucleus
23
Multiple Choice

The diagram shows nuclide notation for Uranium. What is the number of electron in the Uranium?
92
146
238
330
24
Multiple Choice

The diagram shows nuclide notation for Uranium. What is the number of neutron in the Uranium?
92
146
238
330
25
Multiple Choice
the relative charge on a nucleus equal to number of
proton
electron
neutron
nucleon
26
Multiple Choice
the relative mass of a nucleus equal to number of
proton
electron
neutron
nucleon
27
Multiple Choice
"Nuclide of an element with same proton number but different mass number".
This is definition for
nucleon
nuclide
isotope
mass
28

29
Nuclear fission
Nuclear fission is the splitting of a large atomic nucleus into smaller nuclei.
In a nuclear reactor , a neutron is absorbed into a nucleus (typically uranium-235@PLUTONIUM).
This causes the nucleus to become uranium-236, which is violently unstable.
The entire nucleus splits into two large fragments called 'daughter nuclei '. (Krypton and Barium)

30

31
FUSION
What we see as light and feel as warmth is the result of a fusion reaction in the core of our Sun: hydrogen nuclei collide, fuse into heavier helium atoms and release tremendous amounts of energy in the process.

32
DIFFERENT NUCLEAR FISSION AND FUSSION.
Nuclear fission and fusion involve the disintegration and combination of the elemental nucleus.
In the case of nuclear fission, an atom divides into two or smaller or lighter atoms.
Nuclear fusion occurs when two or more atoms join or fuse together to form a large or heavy atom.
33
Multiple Choice
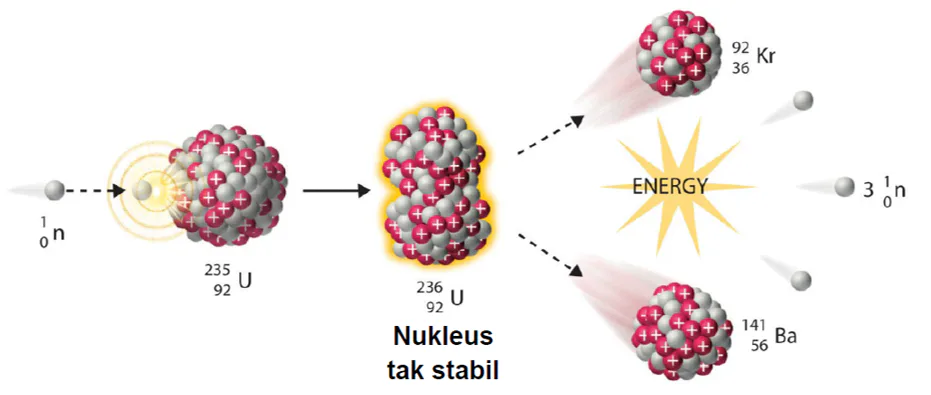
The diagram shows process of
radiation
nuclear fission
nuclear fusion
nuclear energy
34
Multiple Choice
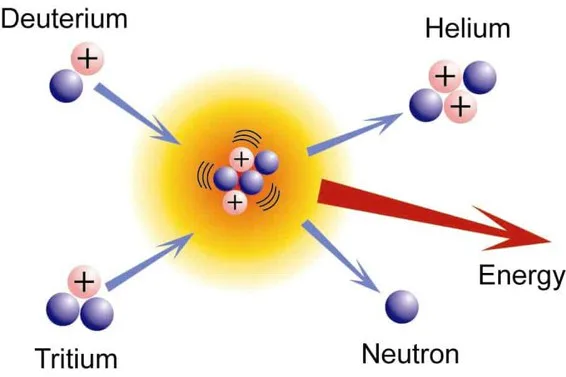
The diagram shows process of
radiation
nuclear fission
nuclear fusion
nuclear energy
35
Multiple Choice
Nuclear ______ splits into neutron and then into smaller nuclei
Fission
Fusion
36
Multiple Choice
nuclear _______ is where two small nuclei collide.
fission
fusion
37
Multiple Choice
nuclear _______ has a much greater release of energy
fission
fusion
38
Multiple Choice
A hydrogen bomb relies on nuclear ______
fission
fusion
39
Multiple Choice
Nuclear ______ emitted alpha particles
fission
fusion
40
Multiple Choice
Which come first nuclear fission or Nuclear Fusion
fission
fusion
41
Multiple Choice
nuclear _____ have nuclei that are not stable and undergo subsequent radioactive decays.
fission
fusion
42
Multiple Choice
nuclear ______ can only occur with conditions that have extreme high pressures and temperatures.
fission
fusion
43
Multiple Choice
The sunshine we enjoy every day comes from nuclear _____
fission
fusion
44
Multiple Choice
Nuclear ______ process uses large atoms, such as Polonium?
fission
fusion
45
Multiple Choice
46
Multiple Choice
47
Multiple Choice
48
Multiple Choice
49
Multiple Choice
50
Multiple Choice
51
Multiple Choice
52
53
54

55
Multiple Choice
Alpha , Beta, Gamma
Beta , Gamma , Alpha
Gamma, Beta, Alpha
Gamma, Alpha, Beta
56

57
58
Multiple Choice
alpha
beta
gamma
59
Multiple Choice
alpha
beta
gamma
60
Multiple Choice

Alpha radiation
Beta radiation
Gamma radiation
61
Multiple Choice

Alpha radiation
Beta radiation
gamma radiation
62
Multiple Choice
This type of electromagnetic wave has the most energy and therefore can be very dangerous.
Microwaves
Radio Waves
X Rays
Gamma Rays
63
64
65
Multiple Choice

What does Z represent?
Atomic Mass (protons + neutrons)
Atomic Number (protons)
Charge (protons - electrons)
Element Symbol
66
Multiple Choice

What does X represent?
Atomic Mass (protons + neutrons)
Atomic Number (protons)
Charge (protons - electrons)
Element Symbol
67
Multiple Choice

What does A represent?
Atomic Mass (protons + neutrons)
Atomic Number (protons)
Charge (protons - electrons)
Element Symbol
68
Multiple Choice

What type of radiation is this?
Alpha
Beta
Gamma
69
Multiple Choice

What type of radiation is this?
Alpha
Beta
Gamma
70
Multiple Choice

What type of radiation is this?
Alpha
Beta
Gamma
71
72
Multiple Choice
Which particle has a negative charge
Alpha
Electron
Proton
Neutron
73
Multiple Choice
When nuclei decay, massive amounts of __________ is released.
energy
electrons
protons
neutrons
74

75
Multiple Choice
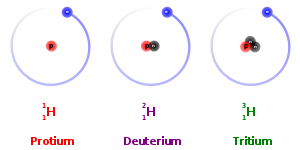
76
Multiple Choice

77
Multiple Choice

What is the mass number of Uranium
92
238.029
340.029
None of these
78
79
Multiple Choice
Helium and Hydrogen atoms
Hydrogen atoms
Hydrogen and Lithium atoms
Hydrogen and Carbon atoms
80
81
Multiple Choice
Underwater
All around us
In the radioactive waste
On the sun
82
83
Multiple Choice
Fusion
Half-life
Fission
Fusion or fission
84
Multiple Choice
85
Multiple Choice
86
Multiple Choice

What is the function of the cooling towers of the nuclear reactor?
to create steam
to spin the turbine
to limit the heat produced by the uranium
to turn steam back into water
87
Multiple Select
Choose THREE benefits of using nuclear power
no CO2 emitted to the atmosphere to cause global warming
no fossil fuels are burned
there is no waste with nuclear power
it provides a large amount of electricity
the waste stays radioactive for thousands of years
88
Multiple Choice
Nuclear Power uses ____________ to create energy
coal
natural gas
uranium
cooling towers
89
90
Multiple Choice

Look at the diagram of an atom of a common element. What is the atomic number?
3
4
7
10
91
Explanation Slide...
Atomic number = proton number
92
Multiple Choice

Look at the diagram of an atom of a common element. What is the mass number?
3
4
7
10
93
94
Multiple Choice
Two isotopes of the same element have
the same number of protons but different numbers of neutrons
the same number of protons but different numbers of electrons
the same number of neutrons but different numbers of protons
the same number of electrons but different numbers of protons
95
96
Multiple Choice
A radioactive isotope is a substance that
will eventually gain electrons through bonding
will become stable by cooling down
is unstable and will 'decay' by losing particles
is not found naturally on the Earth
97
Multiple Choice

Which of the above best describes the words 'contamination' and irradiation'?
A
B
C
D
98
Multiple Choice
Which type(s) of radiation is the most ionising?
alpha
beta
gamma
alpha and beta
beta and gamma
99
Multiple Choice
Which type(s) of radiation is a fast moving electron?
alpha
beta
gamma
alpha and beta
beta and gamma
100
Multiple Choice
Which type(s) of radiation can pass through paper?
alpha
beta
gamma
alpha and beta
beta and gamma
101
Multiple Choice

When Uranium decays it emits an alpha particle, forming an isotope of Thorium. Which of the above decay equations is correct?
A
B
C
D
102
103
Multiple Choice
What change takes place to an atom during beta decay?
A proton changes into a neutron
A proton changes into an electron
An electron changes into a neutron
A neutron changes into a proton
104
Multiple Choice

In the above decay equation, what are the missing numbers x and y?
A
B
C
D
105
Multiple Choice
The half-life of a substance is
the amount of substance left after it has decayed
the time it takes for half of the isotope to decay
the time it takes for half the mass to have radiated away
the mass of half of the isotope after decaying
106
107
Multiple Choice
The activity of an isotope is
the number of particles/rays emitted per second
the same as the half-life in seconds
the amount of radiation emitted over the lifespan of the isotope
the amount of heat generated per second
108
109
110
Multiple Choice

Which of the above graphs shows how the activity of a sample varies with time?
A
B
C
D
111
Multiple Choice
A sample of 16 g of radioactive substance X has a half-life of 2 hours. What percentage of the sample is left after 6 hours?
none
12.5 %
25 %
33.3 %
112
Multiple Choice

Which of the following half-lives would be most suitable for a medical tracer used in a patient?
3 minutes
3 hours
3 days
3 years
113
Multiple Choice
What form of radiation should a medical tracer emit?
alpha
beta
gamma
any of these
114
Multiple Choice
Nuclear fusion occurs in Hydrogen nuclei in the Sun. A hydrogen nuclei consists of a single proton. Fusion requires very high temperatures and high speeds of nuclei to make them fuse together because...
the nuclei bounce off each other at low speeds
protons repel each other as they are positively charged
the protons are too massive to fuse easily
the protons decay easily at low temperatures
115
Multiple Choice
The unit of activity is...
Becquerel
Seconds
Metres
Sievert
116
Multiple Choice

What is the half-life of this radioactive substance?
5700 years
11400 years
17100 years
22800 years
117
Multiple Choice
The most ionising form of nuclear radiation is...
alpha
beta
gamma
infrared
118
Multiple Choice

A radioactive source emits radiation that can pass through a sheet of paper but not through thick aluminium. What does this show about the radiation?
It is alpha-particles.
It is beta-particles.
It is gamma-rays.
It is a mixture of alpha-particles and gamma-rays
119
Multiple Choice

Which line in the table describes the nature of an alpha-particle and a gamma-ray?
A
B
C
D
120
Multiple Choice

The equation shows the decay of the nuclide X:
What are the values of P and Q ?
A
B
C
D
121
Multiple Choice

An unstable nucleus has 145 neutrons and 92 protons. It emits a β-particle.
How many neutrons and protons does the nucleus have after emitting the β-particle?
A
B
C
D
122
Multiple Choice

A Geiger counter detects radiation from radioactive sources.
A radioactive source is inside a thick aluminium container as shown:
Which type of radiation from this source is being detected?
α-particles
β-particles
γ-rays
radio waves
123
Multiple Choice

Which line in the table describes the nature of an α-particle and of a γ-ray?
A
B
C
D
124
Multiple Choice
A radioactive isotope has a half-life of 2 minutes. What can be deduced from this statement?
After 2 minutes, half of the isotope remains.
After 1 minute, half of the isotope remains.
After 4 minutes, half of the isotope remains.
After 4 minutes, none of the isotope remains.
125
Multiple Choice
A detector is used to measure the count-rate near a radioactive source. The reading is 4000
counts per minute. After 30 minutes the count-rate has fallen to 500 counts per minute.
What is the half-life of the radioactive source? You may ignore the effects of background
radiation.
3 minutes
5 minutes
6 minutes
10 minutes
126
Multiple Choice
A freshly made sample of radioactive material gives a count rate of 8000 counts per minute. After
20 days, it gives a count rate of 500 counts per minute.
What is the half-life of the material?
4.0 days
5.0 days
20 days
80 days
127
Multiple Choice

A radioactive source emits radiation that can pass through a sheet of paper but not through thick aluminium. What does this show about the radiation?
It is alpha-particles.
It is beta-particles.
It is gamma-rays.
It is a mixture of alpha-particles and gamma-rays
128
Multiple Choice

Which line in the table describes the nature of an alpha-particle and a gamma-ray?
A
B
C
D
129
Multiple Choice

A radioactive nucleus R decays with the emission of a β-particle as shown:
Which equation is correct?
x = p
y = q
p = x – 1
q = y – 1
130
Multiple Choice

How many nucleons are in a nucleus of ?
19
20
39
58
131
Multiple Choice

The equation shows the decay of the nuclide X:
What are the values of P and Q ?
A
B
C
D
132
Multiple Choice
Which type of radiation has the greatest ionising effect?
α-particles
β-particles
γ-rays
all have the same ionising effect
133
Multiple Choice

An unstable nucleus has 145 neutrons and 92 protons. It emits a β-particle.
How many neutrons and protons does the nucleus have after emitting the β-particle?
A
B
C
D
134
Multiple Choice
Which particles are found in the nucleus of an atom?
neutrons and protons only
neutrons only
protons and electrons only
protons, electrons and neutrons
135
Multiple Choice

A Geiger counter detects radiation from radioactive sources.
A radioactive source is inside a thick aluminium container as shown:
Which type of radiation from this source is being detected?
α-particles
β-particles
γ-rays
radio waves
136
Multiple Choice

Which line in the table describes the nature of an α-particle and of a γ-ray?
A
B
C
D
137
Multiple Choice
A radioactive isotope has a half-life of 2 minutes. What can be deduced from this statement?
After 2 minutes, half of the isotope remains.
After 1 minute, half of the isotope remains.
After 4 minutes, half of the isotope remains.
After 4 minutes, none of the isotope remains.
138
Multiple Choice
A detector is used to measure the count-rate near a radioactive source. The reading is 4000
counts per minute. After 30 minutes the count-rate has fallen to 500 counts per minute.
What is the half-life of the radioactive source? You may ignore the effects of background
radiation.
3 minutes
5 minutes
6 minutes
10 minutes
139
Multiple Choice
A freshly made sample of radioactive material gives a count rate of 8000 counts per minute. After
20 days, it gives a count rate of 500 counts per minute.
What is the half-life of the material?
4.0 days
5.0 days
20 days
80 days
140
Multiple Choice
Between 1909 and 1911, Geiger and Marsden carried out experiments in which alpha particles
were fired at metal foil. Most of the alpha particles passed through the foil with small deflections,
but some were deflected through a large angle.
These results suggest that
atoms contain clouds of electrons through which some alpha particles cannot pass.
atoms contain neutrons that alpha particles bounce off.
atoms have positive and negative charges spread throughout their volume.
atoms have positive charges concentrated in a small volume.
141
Multiple Choice

A narrow beam of alpha-particles is fired at a thin piece of gold foil.
Which is the final direction of the largest number of alpha-particles?
A
B
C
D
142
Multiple Choice
The half-life of a radioisotope is 2400 years. The activity of a sample is 720 counts /s.
How long will it take for the activity to fall to 90 counts /s?
300 years
2400 years
7200 years
19 200 years
143
Multiple Choice
What occurs in the decay of a radioactive nucleus?
The nucleus absorbs another nucleus.
The nucleus absorbs at least one form of radiation.
The nucleus always splits into two equal fragments.
The nucleus emits at least one form of radiation.
144
Multiple Choice

A student investigates a radioactive source that emits only alpha-particles. Without any source
nearby, the detector shows a low reading. The source and thick cardboard are placed near the detector, as shown:
What is the reading on the detector now, and why?
A
B
C
D
145
Multiple Choice

In nuclear ……1……, hydrogen nuclei ……2…… to form helium nuclei, releasing energy.
Which words correctly complete gaps 1 and 2?
A
B
C
D
146
Multiple Choice

Ra decays with a half-life of 1600 s.
Rn decays with a half-life of 52 s.
Po decays with a half-life of 9.1 s.
Pb decays with a half-life of 10.6 h.
The changing count rate for one of these radioactive nuclides is shown in the graph.
From the half-life shown by the graph, which was the decaying radioactive nuclide?
Ra
Rn
Po
Pb
Atom Model
radioactivity
Nuclear Decay


Show answer
Auto Play
Slide 1 / 146
SLIDE
Similar Resources on Wayground

142 questions
untitled
Presentation
•
KG - University

139 questions
Evolution
Presentation
•
9th - 12th Grade

137 questions
Sp2- Scrimmage Review 1
Presentation
•
9th Grade - University

135 questions
#13 ~ CH. 12 ~ WWII ~ Gallery Walk U.S. History
Presentation
•
9th Grade - University

133 questions
STAAR Enrichment & Prep
Presentation
•
9th - 12th Grade

143 questions
Intro to Word
Presentation
•
9th - 12th Grade

144 questions
A2.2 Cell Structure
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

25 questions
The Ultimate College Knowledge Quiz
Quiz
•
8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Math Review
Quiz
•
6th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade
Discover more resources for Physics

25 questions
Electromagnetism
Quiz
•
9th - 12th Grade

10 questions
Exit Check 8.7: The Big Bang
Quiz
•
9th Grade

25 questions
Final Review Units 5-6
Quiz
•
9th Grade

60 questions
Final Review Units 7-8
Quiz
•
9th Grade

10 questions
Exit Check 8.6: Life Cycle of Stars
Quiz
•
9th Grade

45 questions
Spring 2026 Final Exam
Quiz
•
9th - 12th Grade