

L1.1a: Phases of Matter Quizizz
Presentation
•
Chemistry
•
9th - 12th Grade
•
Medium
+8
Standards-aligned
Dorvera Owens
Used 23+ times
FREE Resource
12 Slides • 27 Questions
1

L1.1a: Phases of Matter Summary
2
Open Ended
When you apply heat energy to a substance, where does the energy go? Think about the law of conservation of energy. Find a picture to support your answer.
3

Review and summarize the lesson on states and phases of matter.
Objective
4

5
Multiple Choice
Anything that has mass and takes up space is called a(n)...
Mass
Matter
Volume
Atom
6
Multiple Choice
The smallest part of an element is called a(n)...
Matter
Weight
Atom
Mass
7

8
Multiple Choice
Energy cannot be created nor destroyed. Just _________
heated
transferred
degraded
manifested
9
Multiple Choice
If energy is NOT created or destroyed, what is happening to it?
energy is transferred between kinetic energy types
energy is transferred between kinetic and potential energies
the big bang has to create more energy
it is being lost and gained by the universe
10

11
Multiple Choice
Liquid to Gas
melting
vaporization
condensation
sublimation
12
Multiple Choice
Gas to liquid
vaporization
melting
condensation
sublimation
13
Multiple Choice
Solid to liquid
freezing
melting
sublimation
condensation
14
Multiple Choice
15
Multiple Choice

Frost on this window pane is:
deposition
sublimation
condenstaion
evaporation
16
Multiple Choice
What best describes the motion of molecules in a liquid?
flowing past each other
moving freely in all directions
17
Multiple Choice
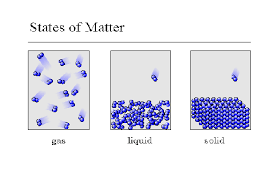
18
Multiple Choice

19

20
Multiple Choice
Can you make the particles of any substance stop moving?
Yes, by increasing the temperature
No, particles will always be in motion no matter the temperature
Yes theoretically, by lowering the temperature extremely.
I need more information
21
22
23
Match
Match the following
100 degrees C
0 degrees C
0 K
32 degrees F
273 K
boiling point of water
freezing point of water at Celcius
absolute zero on Kelvin scale
freezing point of water at Fahrenheit
freezing point of water at Kelvin
boiling point of water
freezing point of water at Celcius
absolute zero on Kelvin scale
freezing point of water at Fahrenheit
freezing point of water at Kelvin
24
Multiple Choice
What happens if you add energy to particles using a heater?
no change other than all atoms speed up
more atoms would condense
more atoms would vaporize due to boiling
more atoms would slow down and turn to a gas
25

26
Multiple Choice

What makes this snowperson melt?
heat causing particles to slow down
heat causing particles to speed up
light making particles stop
air making particles disappear
27
Multiple Select

Answer the following questions about states of matter
3 States of Matter
Which of the following statements are true about states of matter and kinetic energy
Fewer particles means there is more kinetic energy
As temperature increases so does kinetic energy
As temperature decreases particles move closer together
As the number of particles increases so does kinetic energy
28
Multiple Choice
Thermal energy always moves:
From a high temperature object to a lower temperature object.
From a lower temperature object to a higher temperature object.
From an object with lower kinetic energy to an object with higher kinetic energy.
From an object of higher mass to an object of lower mass.
29
Multiple Choice
____________ is the measure of the
average kinetic energy of the particles in a material.
Element
Compound
Atom
Temperature
30
Multiple Choice
Sequence these states of matter from lowest energy level to highest energy.
Solid Liquid Gas
Gas Liquid Solid
Liquid Solid Gas
31

32
Multiple Choice

Use the following phase diagram of water to answer the following questions.
Phase Diagram of Water
What state is water in at a temperature of 50°C and pressure of 100 atm?
Solid
Liquid
Gas
Plasma
33
Multiple Choice

Use the following phase diagram of water to answer the following questions.
Phase Diagram of Water
At 100°C and a pressure below 1.0 atm, water exists in which phase of matter?
Solid
Liquid
Gas
Plasma
34
Multiple Choice

Use the following phase diagram of water to answer the following questions.
Phase Diagram of Water
What state is water in at a temperature of -50°C and pressure of 1 atm?
Solid
Liquid
Gas
Plasma
35

36
Multiple Choice
Substances that are made up of ONLY one type of atom (element) or compound.
Heterogeneous mixture
Pure substance
Suspension
Homogeneous mixture
37

38
Multiple Choice
Chalk is a mineral found in rocks. It contains carbon, oxygen and calcium atoms. Chalk is a ___________________ .
element
compound
39
Open Ended
What is one new thing you learned in today's lesson? Give an example and a picture to support your answer.

L1.1a: Phases of Matter Summary
Show answer
Auto Play
Slide 1 / 39
SLIDE
Similar Resources on Wayground

33 questions
Polyatomic Ions
Presentation
•
10th - 12th Grade

34 questions
Chemistry: Unit 1 Module 5 Dimensional Analysis
Presentation
•
10th - 12th Grade

34 questions
HW: Balancing Equations
Presentation
•
9th - 12th Grade

33 questions
Atoms and Atomic Structure
Presentation
•
8th - 11th Grade

32 questions
Phase Changes & Phase Diagrams
Presentation
•
9th - 12th Grade

33 questions
Sectionalism
Presentation
•
9th - 12th Grade

35 questions
Electron Configuration
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Interpreting Solubility Curves
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
10th Grade

43 questions
Acids and Bases Review
Quiz
•
10th - 11th Grade

20 questions
Thermochemistry Review
Quiz
•
10th Grade

25 questions
Unit 4 (Mid) Rumble
Quiz
•
10th Grade