

Chemistry- Atomic Structure Unit Test Study Guide (Ch. 4 & 5)
Presentation
•
Chemistry
•
10th Grade
•
Easy
Lila Alvarez
Used 7+ times
FREE Resource
16 Slides • 33 Questions
1
Atomic Structure Unit Test Study Guide (Ch. 4 & 5)

Test September 29, 2023
2
Multiple Choice
smallest particle of an element that retains its identity in a Chemical reaction
Atom
Proton
Neutron
3
In the early 1800s, the english chemist John Dalton performed a number of experiments that eventually led to the acceptance of the idea of atoms. He formulated the first atomic theory since the "death of chemistry" that occurred during the prior 2000 years. Dalton theorized that all matter is made of atoms and that atoms are too small to see, "uncuttable" , and indestructible. He also theorized that all atoms of a given element are exactly alike and atoms of different elements are different.
Dalton’s atomic theory

4
Democritus, a philosopher in ancient Greece, began the search for a description of matter. He questioned whether matter could be divided into smaller and smaller pieces forever until eventually the smallest possible piece would be obtained. He believed that the smallest possible piece of matter was indivisible and he named the smallest piece of matter "atomos", meaning "not to be cut". To Democritus, atoms were small, hard particles that were all made of the same material, but were formed into different shapes and sizes.
Democritus’ idea of the atom.

5
Dalton's Atomic theory
Dalton’s atomic theory.
All elements are composed of tiny indivisible particles called atoms
Atoms of the same element are identical. The atoms of any of element are different from those of any other element.
Atoms of different elements can physically mix together or can chemically combine in simple whole-number ratios to form compounds.
Chemical reactions occur when atoms are separated from each other, joined, or rearranged in a different combination. Atoms of one element, however, are never changed into atoms of another element as a result of a chemical reaction.
6
Explain how scientists observe individual atoms.
Atoms are very small. If you could line up 100,000,000 copper atoms side by side, they would produce a line only 1 cm long!
Despite their small size, individual atoms are observable with instruments such as scanning electron microscopes.
7
Multiple Choice
Atoms are very small. If you could line up 100,000,000 copper atoms side by side, they would produce a line only 1 cm long!
Despite their small size, individual atoms are observable with instruments such as scanning electron microscopes.
Individual atoms are observable with instruments such as scanning electron microscopes.
Individual atoms are observable with music instruments
Individual atoms are observable with instruments such as scanning printers.
Individual protons are observable with instruments such as scanning electron microscopes.
8
Multiple Choice
negatively charged subatomic particles, mass is 1/1840 the mass of a hydrogen atom, live on the outside “shells” of atoms, symbol e-
Proton
Electron
Neutron
9
Multiple Choice
positively charged subatomic particles, mass 1840 times that of an electron, live in the nucleus of atoms, symbol p+
Electrons
Neutrons
Protons
10
Multiple Choice
subatomic particle with no charge, mass equal to proton, live in nucleus of atom, symbol n0
Protons
Neutrons
Electrons
11
Multiple Choice
tiny central core of an atom and is composed of protons and neutrons.
Protons
Nucleus
Electrons
12
In the nuclear atom, the protons and neutrons are located in the positively charged nucleus. The electrons are distributed around the nucleus and occupy almost all the volume of the atom.
They exist in...
Three kinds of subatomic particles are electrons, protons, and neutrons.
Three Subatomic Particles:
List and describe the three subatomic particles. Include where each particle exists in an atom, their charges, and their mass.
13
Multiple Choice
Where are protons and neutrons Located?
are located in the positively charged nucleus.
are located in the negative charged nucleus.
14
Multiple Choice
Where are the electrons located?
The electrons are distributed around the nucleus and occupy almost all the volume of the atom.
The electrons are distributed around the electrons and occupy almost all the volume of the space.
15
Explain the plum pudding model and compare it to Bohr’s atomic model.
This answer choice is referring to Bohr's model of the atom, where electrons occupy fixed energy orbits around the nucleus. So as we discussed before, the plum pudding model is different from the hard-sphere model of the atom because the plum pudding model included negatively charged particles known as electrons.

16
Multiple Choice
is the number of protons in the nucleus of an atom of that element.
atomic number
mass number
atomic mass
17
Multiple Choice
The total number of protons and neutrons in an atom is called the.....
Mass quantity
atomic number
Mass number
18
Multiple Choice
are atoms that have the same number of protons but different numbers of neutrons.
Isotope
atomic number
atomic mass
19
Multiple Choice
______ is defined as 1/12th of the mass of a carbon-12 atom.
atomic mass unit (amu)
atomic mass
mass quanitty
20
Multiple Choice
a weighted average mass of the atoms in a naturally occurring sample of the element.
atomic quantity
atomic mass
21
Explain why each element differs?
Isotopes
Because isotopes of an element have different numbers of neutrons, they also have different mass numbers.
Despite these differences, isotopes are chemically alike because they have identical numbers of protons and electrons, which are the subatomic particles responsible for chemical behavior.
What makes one element different from another?
Elements are different because they contain different numbers of protons.
An element’s atomic number is the number of protons in the nucleus of an atom of that element.
Since all hydrogen atoms have one proton, the atomic number of hydrogen is 1.
Remember that atoms are electrically neutral. Thus, the number of electrons must equal the number of protons.
22


23
Multiple Select
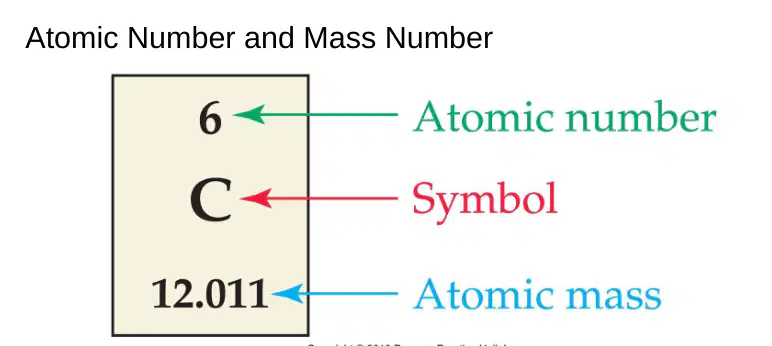
How many protons, electrons, and neutrons are in each atom?
(Carbon (C) (atomic #6) > mass = C: 12.011
Protons = 6
Electrons = 6
Neutrons = 6
Protons = 12
24

25
Fill in the Blanks
26
Multiple Choice
The fixed energies an electron can have are called ...
Energy Levels
quantum
Energy
27
Multiple Choice
A .... of energy is the amount of energy required to move an electron from one energy level to another energy level.
atomic mass
quantum
quantum mechanical model
28
Multiple Choice
The ...................................................determines the allowed energies an electron can have and how likely it is to find the electron in various locations around the nucleus of an atom.
quantum mechanical model
guantaum level
atomic mass
29
Multiple Choice
....is the probability of finding an electron at various locations around the nucleus.
atomic unit
atomic mass
atomic orbital
30
In 1913, Niels Bohr developed a new atomic model
Bohr proposed that an electron is found only in specific circular paths, or orbits, around the nucleus.

31
Explain how energy is affected by the energy level.
The amount of energy an electron gains or loses in an atom is not always the same. Like the rungs of the strange ladder, the energy levels are not equally spaced.
The higher level are closer together. The higher the energy level occupied by an electron, the less energy it takes to move from that energy level to the next higher energy level.
32
How do scientists use the electron cloud to determine the location of electrons?
In the quantum mechanical model of the atom, the probability of finding an electron within a certain volume of space surrounding the nucleus can be represented as a fuzzy cloudlike region. Where the cloud is more dense, the probability of finding an electron is high.

33
Atomic Orbitals - S, P, D, F
Different atomic orbitals are denoted with letters. S orbitals are spherical and p orbitals are dumbbell-shaped.

34
Multiple Choice
The ways in which electrons are arranged in various orbitals around the nuclei of atoms are called ______
electron configurations
electron connection
35

36
Fill in the Blanks
37
Fill in the Blanks
38
Fill in the Blanks
39
Multiple Choice
What's the max orbitals of S ?
2 max
6 max
1 max
3 max
40
Multiple Choice
What's the max orbitals of P ?
7 max
6 max
6.5 max
4 max
41
Multiple Choice
What's the max orbitals of D ?
10 max
9 max
11 max
5 max
42
Multiple Choice
What's the max orbitals of F ?
10 max
13 max
7 max
14 max
43
Multiple Choice
height of a wave; distance from midline to top of wave
amplitude
height
length
distance
44
Multiple Choice
distance from crest to crest;
measured in nanometers
(1 meter = 1 x 109 nm)
Wavelength (λ)
measuring tape
height/distance
45
Multiple Choice
number of waves per second;
measured in Hertz (1 Hz = 1/sec)
Accuracy
Frequency (ν)
46
Multiple Choice
Electrons in atoms will absorb and emit energy in the form of ____________ (waves/photons).
electromagnetic radiation
electromagnetic ration
electromagnetic distance
47
Multiple Choice
a band of colors, as seen in a rainbow, produced by separation of the components of light by their different degrees of refraction according to wavelength.
distance
spectrum
Saturn
48
Multiple Choice
Each spectral line in an atomic emission spectrum of an element corresponds exactly to one wavelength of light emitted by the electrons of that element.
*no two elements have the same ___________.
atomic emission spectrum
spectrum
addition postulate
49
Describe what happens to atoms when they gain or lose energy.
When an electric current is passed through a gaseous element, the electrons of the atoms are energized. This energy causes them to emit light.
When atoms absorb energy, their electrons move to higher energy levels. These electrons lose energy by emitting light when they return to lower energy levels.
Atomic Structure Unit Test Study Guide (Ch. 4 & 5)

Test September 29, 2023
Show answer
Auto Play
Slide 1 / 49
SLIDE
Similar Resources on Wayground

41 questions
Y10T2W6 Mid-Term 2 Revision- Stoichiometry
Presentation
•
10th Grade

44 questions
Periodic Trends
Presentation
•
10th Grade

45 questions
Lab Safety
Presentation
•
10th Grade

42 questions
All About Venn Diagrams
Presentation
•
10th Grade

43 questions
Sulphur - full topic (review + 16/11/2020 lesson)
Presentation
•
10th Grade

44 questions
AP Chemistry Periodic Trends
Presentation
•
10th Grade

46 questions
Y10CT1W11L1 Group 7: The Halogens
Presentation
•
10th Grade

46 questions
Parts of an Equation & Reaction Types
Presentation
•
10th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Interpreting Solubility Curves
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
10th Grade

43 questions
Acids and Bases Review
Quiz
•
10th - 11th Grade

20 questions
Thermochemistry Review
Quiz
•
10th Grade

25 questions
Unit 4 (Mid) Rumble
Quiz
•
10th Grade