

1st Quarter 2023 Review
Presentation
•
Biology
•
9th - 12th Grade
•
Easy
Anonymous Anonymous
Used 3+ times
FREE Resource
14 Slides • 36 Questions
1

2

3


4

5

6

7

8

9
10
Multiple Choice
11
Multiple Choice
12
Multiple Choice
13
Multiple Choice

14
Multiple Choice
15
Multiple Choice
16
Multiple Choice
17
Multiple Choice
18
Multiple Choice
19

20
21
22
Multiple Choice

23
Multiple Choice
24
Multiple Choice
hormones
25
Multiple Choice
26
Multiple Choice
27
Multiple Choice
28
Multiple Choice
29
Multiple Choice
30
Multiple Choice
31
Multiple Choice

32


33
34
Multiple Choice
35
Multiple Choice
36
Multiple Choice
37
Multiple Choice
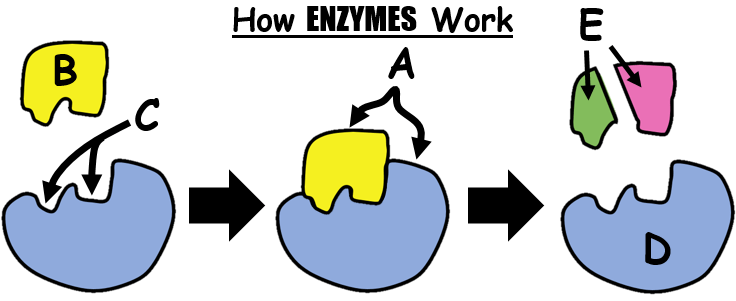
38
Multiple Choice
39
Multiple Choice
40
Multiple Choice
41
Multiple Choice
42
Multiple Choice
What percentage of energy is passed on to the next trophic level?
5%
10%
50%
90%
43
Multiple Choice

44
Multiple Choice
45
Multiple Choice
46
Multiple Choice
47
Multiple Choice

When a population grows past the ecosystem's carrying capacity, what happens to the population?
The population continues to grow
The population will go extinct due to limited resources
The population starts to die off to return to carrying capacity
The population grows, then finds a new carrying capacity
48
Multiple Choice
A population is...
All of the living and nonliving things in an area
Multiple species that live in one area
A group of one species in one area
None of these
49
Multiple Choice

50
Multiple Choice


Show answer
Auto Play
Slide 1 / 50
SLIDE
Similar Resources on Wayground

44 questions
Powers Mendelian Genetics Presentation
Presentation
•
9th - 12th Grade

41 questions
Cell Organelles
Presentation
•
9th - 12th Grade

46 questions
Meiosis/Codon Chart
Presentation
•
9th - 12th Grade

43 questions
Waves Lesson 1
Presentation
•
9th - 12th Grade

46 questions
CATEGORY 1 STAAR REVIEW
Presentation
•
9th - 12th Grade

44 questions
9 Week Test Review: Unit 1
Presentation
•
9th - 12th Grade

46 questions
AR Verb Conjugations
Presentation
•
9th - 12th Grade

44 questions
Pinnepeds
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

20 questions
"What is the question asking??" Grades 3-5
Quiz
•
1st - 5th Grade

20 questions
“What is the question asking??” Grades 6-8
Quiz
•
6th - 8th Grade

10 questions
Fire Safety Quiz
Quiz
•
12th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

34 questions
STAAR Review 6th - 8th grade Reading Part 1
Quiz
•
6th - 8th Grade

20 questions
“What is the question asking??” English I-II
Quiz
•
9th - 12th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

47 questions
8th Grade Reading STAAR Ultimate Review!
Quiz
•
8th Grade
Discover more resources for Biology

24 questions
BIOLOGY STAAR REVIEW
Quiz
•
9th Grade

30 questions
Biology STAAR Review #3
Quiz
•
9th Grade

16 questions
AP Biology: Unit 1 Review (CED)
Quiz
•
9th - 12th Grade

50 questions
Biology EOC Review
Quiz
•
9th Grade

25 questions
photosynthesis and cellular respiration
Quiz
•
9th Grade

20 questions
AP Biology: Unit 3 Review (CED)
Quiz
•
9th - 12th Grade

20 questions
Enzymes
Quiz
•
9th Grade

20 questions
Food Chains and Food Webs
Quiz
•
7th - 12th Grade