

VSEPR theory
Presentation
•
Chemistry
•
University
•
Medium
Standards-aligned

Luis Bello
Used 14+ times
FREE Resource
16 Slides • 14 Questions
1
Valence Shell Electron Pair Repulsion.
Understanding the secrets behind how electrons are arranged in an atom and their impact on chemical properties.
2
- Welcome to the lecture on VSEPR Theory!
- VSEPR stands for Valence Shell Electron Pair Repulsion theory.
- It is a fundamental concept in chemistry used to predict molecular geometry.
- Understanding VSEPR theory helps us visualize how atoms are arranged in a molecule.
3
Molecular Geometry
- Molecular geometry refers to the three-dimensional arrangement of atoms in a molecule.
- It determines a molecule's shape, which in turn affects its chemical and physical properties.
4
VSEPR Theory - Basic Idea
- VSEPR theory is based on the idea that electron pairs (bonding and non-bonding) around a central atom will arrange themselves in a way to minimize repulsion.
- This leads to specific molecular geometries.
5
Electron Pair Repulsion
- Electrons repel each other due to their negative charges.
- Bonding pairs and lone pairs of electrons will arrange themselves to be as far apart as possible.
6
Electron Pair Arrangement
- Two types of electron pairs: bonding pairs (shared between atoms) and lone pairs (non-bonding pairs).
- The arrangement of these pairs determines molecular geometry.
7
VSEPR Geometries
- There are several basic VSEPR geometries:
1. Linear
2. Trigonal Planar
3. Tetrahedral
4. Trigonal Bipyramidal
5. Octahedral
8
Linear Geometry (AX2)
- Example: BeCl2
- Explanation of the arrangement of bonding and non-bonding pairs.

9
Trigonal Planar Geometry (AX3)
- Example: BF3
- Explanation of the arrangement of bonding and non-bonding pairs.

10
Tetrahedral Geometry (AX4)
- Example: CH4
- Explanation of the arrangement of bonding and non-bonding pairs.

11
Trigonal Bipyramidal Geometry (AX5)
- Example: PCl5
- Explanation of the arrangement of bonding and non-bonding pairs.

12
Octahedral Geometry (AX6)
- Example: SF6
- Explanation of the arrangement of bonding and non-bonding pairs.

13
Bent and Trigonal Pyramidal Geometry (AX6)
- Example: H2O (bent) and NH3 (trigonal pyramidal)
- Explanation of the arrangement of bonding and non-bonding pairs.


14

15

16
Multiple Choice
17
18
Multiple Choice
19
Multiple Choice
20
Multiple Choice
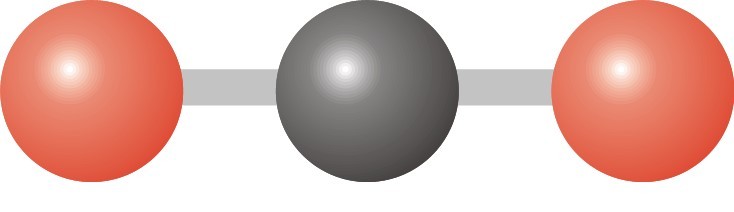
21
Multiple Choice
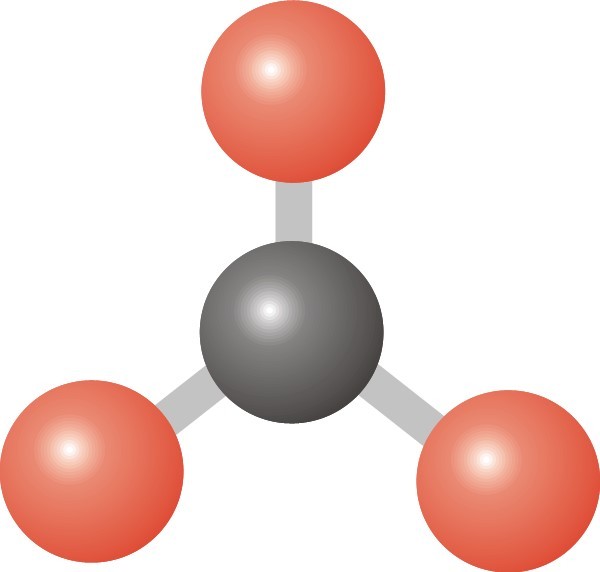
22
Multiple Choice
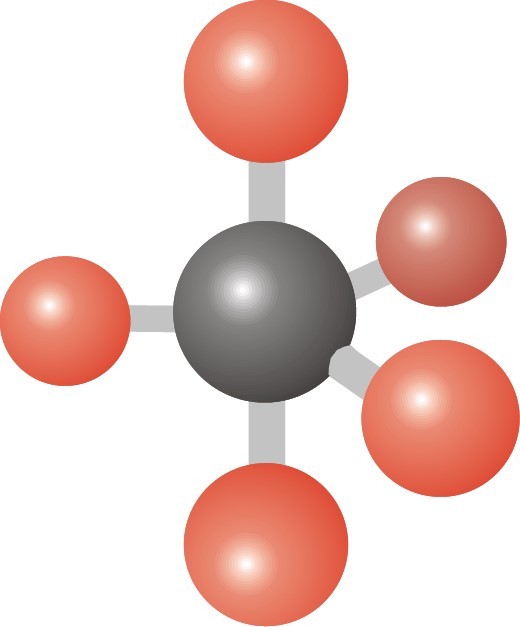
23
Multiple Choice
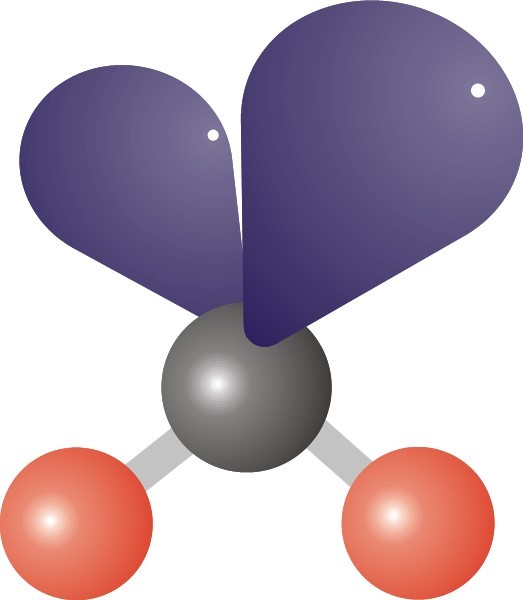
24
Multiple Choice
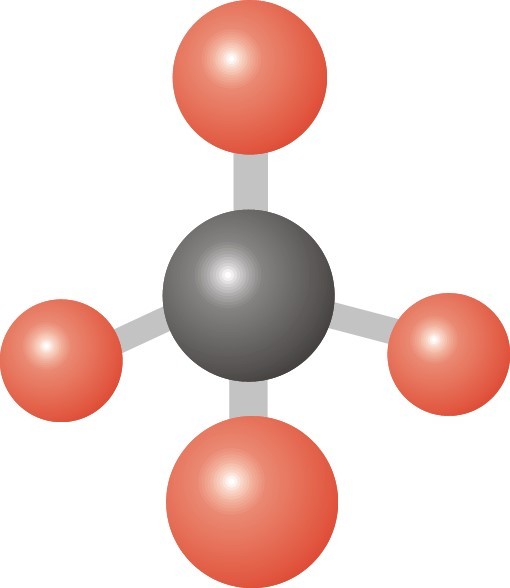
25
Multiple Choice
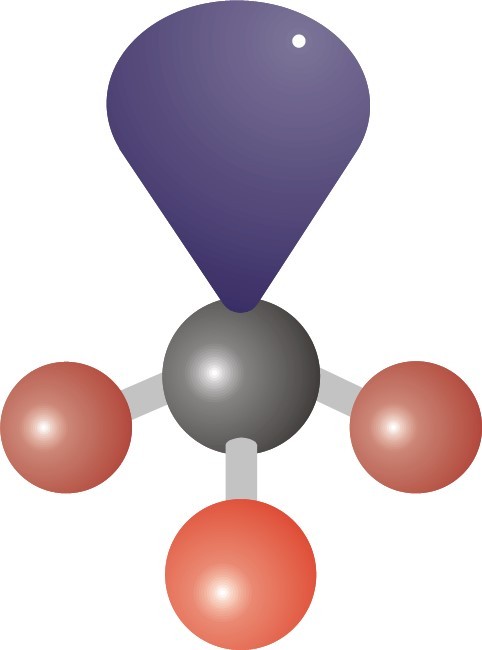
26
Multiple Choice
What molecular geometry would PH3 have?
Trigonal Pyramidal
Trigonal Bipyramidal
Bent
Linear
27
Multiple Choice

28
Multiple Choice
29
Multiple Choice

What shape would this have?
Trigonal planar
Pyramidal
Tetrahedral
Bent
30
Multiple Choice

Valence Shell Electron Pair Repulsion.
Understanding the secrets behind how electrons are arranged in an atom and their impact on chemical properties.
Show answer
Auto Play
Slide 1 / 30
SLIDE
Similar Resources on Wayground

23 questions
Electromagnetism and Right Hand Rules
Presentation
•
12th Grade

22 questions
21st Century Skills -TTS Quetzaltenango
Presentation
•
Professional Development

28 questions
Describing Graphs 2
Presentation
•
University

25 questions
Gases Lesson
Presentation
•
University

26 questions
The Solid State
Presentation
•
University

22 questions
Unidad 2: Metabolismo Celular
Presentation
•
12th Grade

27 questions
Molecular Geometry
Presentation
•
11th - 12th Grade

21 questions
What they carried intro
Presentation
•
12th Grade - University
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Chemistry

20 questions
Guess The App
Quiz
•
KG - Professional Dev...

11 questions
NFL Football logos
Quiz
•
KG - Professional Dev...

19 questions
Minecraft
Quiz
•
6th Grade - Professio...

40 questions
8th Grade Math Review
Quiz
•
8th Grade - University

20 questions
Block Buster Movies
Quiz
•
10th Grade - Professi...

10 questions
Would you rather...
Quiz
•
KG - University

40 questions
Flags of the World
Quiz
•
KG - Professional Dev...

14 questions
Superhero
Quiz
•
1st Grade - University