

Separation Techniques
Presentation
•
Chemistry
•
•
Practice Problem
•
Medium
Manisha Dogra
Used 7+ times
FREE Resource
0 Slides • 29 Questions
1
Multiple Choice
What is the method suitable to obtain pure water from seawater.
Simple distillation
Fractional distillation
Filtration
Evaporation to dryness
2
Multiple Choice
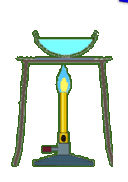
3
Multiple Choice
4
Multiple Choice
5
Multiple Choice
6
Multiple Choice
True or false: Magnets can be used to separate iron filings from sand
True
False
7
Multiple Choice

Name the separation technique
Filtration
Evaporation
Distillation
Separation
8
Multiple Choice
Filtration can be used to separate...
an insoluble solid from a solution
a soluble solid from a solution
a dissolved solid from a solution
9
Multiple Choice
The change of a liquid to a gas to remove solvent and collect solute is called
Decantation
Filtration
Evaporation
Centrifuge
10
Multiple Choice
Decanting is used to separate...
an insoluble solid from a solution.
a soluble solid from a solution.
11
Multiple Choice
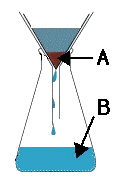
12
Multiple Choice
A substance will expand when it is...
heated
cooled
13
Multiple Choice

What method is shown in the picture?
Filtration
Evaporation
Chromatography
Sieving
14
Multiple Choice
Sand is...
soluble (can dissolve)
insoluble (can't dissolve)
15
Multiple Choice
A gas looses energy and turns to a liquid. What is this called?
melting
freezing
condensation
evaporation
16
Multiple Choice
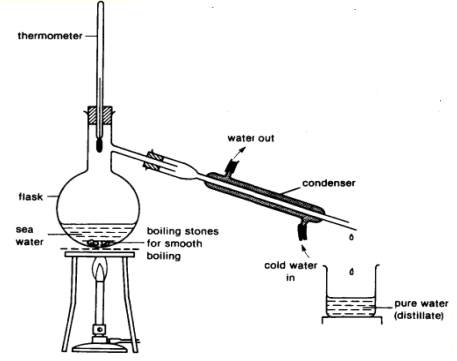
What is the name of this separation technique?
Simple distillation
Fractional distillation
Boiling
Evaporation
17
Multiple Choice
Which of the following statements tells us that a sample of liquid is pure water?
It is colourless.
It leaves no residue when filtered.
It boils at 100oC.
It is odourless.
18
Multiple Choice
What is the physical property on which fractional distillation depends?
different boiling points
different melting points
different solubilities
different densities
19
Multiple Choice
For paper chromatography to work,
the dyes must have different boiling points.
the dyes must have different solubilities in the solvent.
the starting line on the chromatogram must be drawn using pen.
The starting line must be below the level of the solvent.
20
Multiple Choice
Which separation technique is used to obtain salt from seawater?
Simple distillation
Filtration
Chromatography
Evaporation
21
Multiple Choice
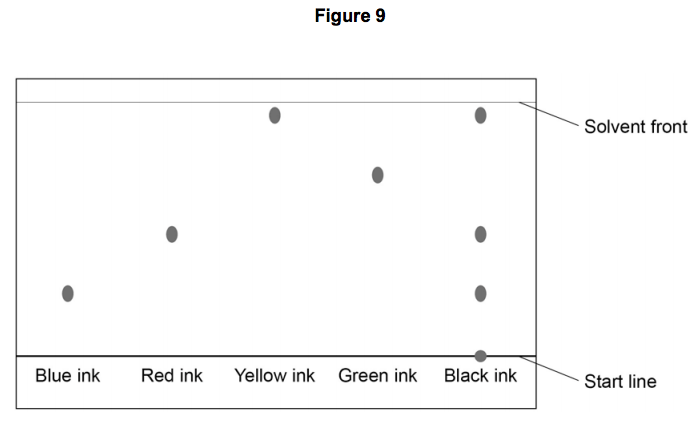
Which ink is a mixture?
Blue
Red
Yellow
Black
22
Multiple Choice

Dyes in water soluble markers may be separated by means of
Simple distillation
Crystallisation
Chromatography
Filtration
23
Multiple Choice

The image shows a mixture of concrete nails and saw dusts. What technique is best to use to separate the nails from the saw dusts?
sieving
chromatography
decantation
magnetic separation
24
Multiple Choice
Which tool could you use to separate large and small particles?
A sieve
A distillation flask
Chromatography
Evaporating basin
25
Multiple Choice
Solutions are made up of...?
Solutes and insolubles
Solubles and solutes
Solutes and solvents
Solubles and solvents
26
Multiple Choice
Which part of a filtration is the "filtrate"?
The filter paper
The solid material left in the funnel
The liquid that has gone through
The conical flask
27
Multiple Select

What is mixture?
A mixture is a physical combination of two or more substances.
A mixture is a chemical combination of only one substance.
In it the substances retain their individual properties and can be separated by physical means.
In it the substances do not retain their individual properties and can be separated by chemical means.
28
Multiple Select

What are homogenous mixtures?
In homogenous mixtures the components are evenly distributed.
In homogenous mixtures the components are not evenly distributed.
They are distinguishable from each other.
They are not distinguishable from each other.
29
Multiple Choice

In a heterogeneous mixture, the components are not uniformly distributed throughout and they remain distinct.
True
False
What is the method suitable to obtain pure water from seawater.
Simple distillation
Fractional distillation
Filtration
Evaporation to dryness
Show answer
Auto Play
Slide 1 / 29
MULTIPLE CHOICE
Similar Resources on Wayground

20 questions
The Structure of An Atom
Presentation
•
11th Grade

21 questions
Mass and The Mole Part 1
Presentation
•
10th - 12th Grade

23 questions
Earth Quake Faults
Presentation
•
7th - 12th Grade

22 questions
Naming compounds
Presentation
•
6th - 7th Grade

21 questions
Ionic Bonding
Presentation
•
8th - 12th Grade

21 questions
Isotopes and Avg. Atomic Mass Notes
Presentation
•
9th - 12th Grade

21 questions
Balancing Chemical Equations
Presentation
•
9th Grade

20 questions
Physical and Chemical Properties
Presentation
•
6th - 8th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

15 questions
Acids and Bases Review
Quiz
•
8th Grade

20 questions
Interpreting Solubility Curves
Quiz
•
10th Grade

10 questions
5.P.2.3 Chemical and Physical Changes
Quiz
•
5th Grade

20 questions
Types of Chemical Reactions
Quiz
•
10th Grade

20 questions
Counting Atoms Practice
Quiz
•
8th Grade