

Average Atomic Mass
Presentation
•
Science, Chemistry
•
11th Grade
•
Medium
+3
Standards-aligned
Mr. Poyser
Used 11+ times
FREE Resource
2 Slides • 49 Questions
1
Multiple Choice
The properties of elements are periodic functions of their
mass numbers
atomic numbers
atomic masses
atomic radii
2
Multiple Choice
If an element has two isotopes with masses 10 amu and 12 amu, and their abundances are 25% and 75% respectively, what is the average atomic mass of the element?
13 amu
10.5 amu
11.5 amu
9 amu
3
Multiple Choice
If an element has two isotopes with masses 5 amu and 7 amu, and their abundances are 40% and 60% respectively, what is the average atomic mass of the element?
5.6
6.8
7.5
6.2
4
Multiple Choice
If an element has two isotopes with masses 8 amu and 10 amu, and their abundances are 60% and 40% respectively, what is the average atomic mass of the element?
8.8 amu
10.4 amu
9.6 amu
9.2 amu
5
Multiple Choice
If 75.0% of the isotopes of an element have a mass of 35.0 amu and 25.0% of the isotopes have a mass of 37.0 amu, what is the atomic mass of the element?
35.0 amu
35.5 amu
37.0 amu
36.0 amu
6
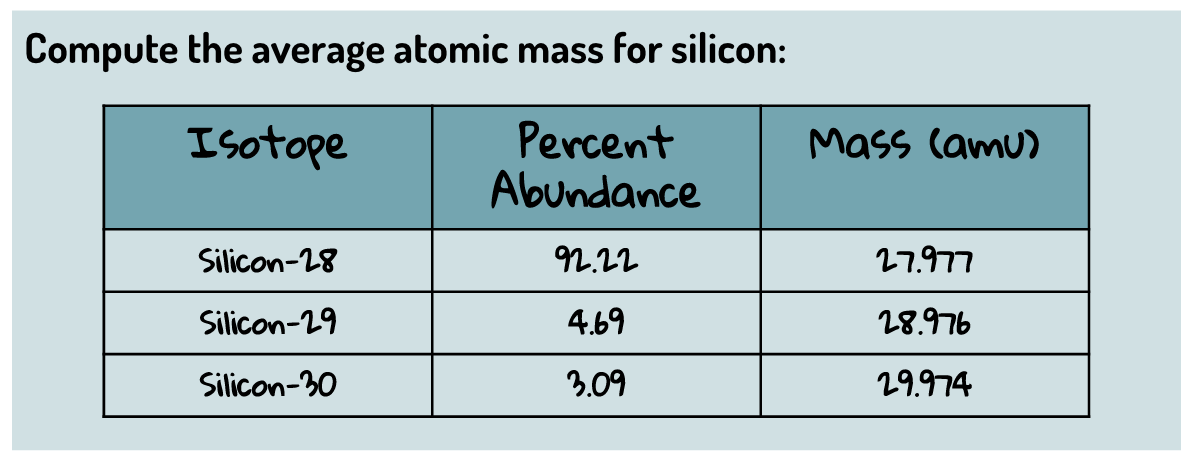
Multiple Choice
How many protons does this element have?
7
Multiple Choice
What is the average mass of this element?
8
Multiple Choice
9
Multiple Choice
10
Multiple Choice
11
Multiple Choice
12
Multiple Choice
I. 7735X II. 7733X III. 8137X IV. 8135 X
13
Multiple Choice

14
Multiple Choice

15
Multiple Choice
An element has three isotopes. Given the abundances and relative masses, calculate the average atomic mass and determine (from the periodic table) which element it is.
Abundances | Relative masses
0.005% | 234.04 amu
0.720% | 235.04 amu
99.275% | 238.05 amu
Uranium (#92, Atomic Mass: 238.03 amu)
Fluorine (#9, Atomic Mass: 19.00 amu)
Mercury (#89, Atomic Mass: 200.59 amu)
Polonium (#84 209 amu)
16
Multiple Choice
Element Z has 2 natural isotopes. One isotope has a mass of 15.0 amu and a relative abundance of 30%. The other isotope has a mass of 16.0 amu and a relative abundance of 70%. Estimate the average atomic mass for this one element.
15.0 amu
16.0 amu
15.7 amu
16.9 amu
17
Multiple Choice
Chromium has four naturally occurring isotopes (chromium-50, chromium-52, chromium-53, and chromium-54). Which isotope would you predict is the most abundant? (Hint: You will need to look at the average atomic mass of chromium on the periodic table to make your decision.)
chromium-50
chromium-52
chromium-53
chromium-54
18
Multiple Choice

Which has more protons?
neither
carbon - 12
carbon - 13
19
Multiple Choice

Which has more electrons?
neither
carbon - 12
carbon - 13
20
Multiple Choice

Which has more neutrons?
neither
carbon - 12
carbon - 13
21
Fill in the Blanks

Type answer...
22
Fill in the Blanks

Type answer...
23
Ions
Ion means charged atom
To be charged it needs more or less electrons than protons
more electrons = NEGATIVE
less electrons = POSITIVE

24
Ion Notation
Same as isotope
includes charge in upper right
+ means LESS ELECTRONS than PROTONS
(-) means MORE ELECTRONS than PROTONS

25
Multiple Choice

How many protons?
36
17
18
19
26
Multiple Choice

How many neutrons?
36
17
18
19
27
Multiple Choice

How many electrons?
36
17
18
19
28
Multiple Choice

How many protons?
9
4
5
2
29
Multiple Choice

How many neutrons?
9
4
5
2
30
Multiple Choice

How many electrons?
9
4
5
2
31
Multiple Choice
If a proton is added or removed from an atom, what happens?
It becomes a different element.
It becomes a different isotope.
Nothing happens.
32
Multiple Choice

How many neutrons are in 1 atom of Carbon-13?
6
13
7
20
33
Multiple Choice
Which is the correct symbol for Fluorine-18?
34
Multiple Choice

35
Multiple Choice
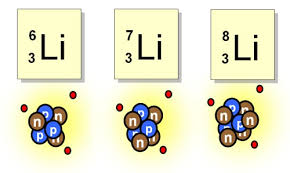
36
Multiple Choice
37
Multiple Choice
What is the average mass of this element?
38
Multiple Choice

39
Multiple Choice
The atomic mass of an element is the ___.
average of the mass number and the atomic number for the element
weighted average of the masses of the isotopes of the element
total mass of the isotopes of the element
total number of subatomic particles in the nucleus
40
Multiple Choice
41
Multiple Choice
42
Multiple Choice
The average atomic mass on the periodic table is found from...
calculating the weighted average mass of all the isotopes of that element
Calculating different types of elements together
calculations the average of all elements in that period together.
making a guess about how much all the isotopes might wiegh together.
43
Multiple Choice
44
Multiple Choice

45
Multiple Choice

46
Multiple Choice
The vertical (up and down) columns in the Periodic Table are called
groups
towers
periods
atomic numbers
47
Multiple Choice
Write the hyphen notation for the isotope that has 39 protons and 50 neutrons.
Yttrium-11
Tin-39
Tin-89
Yttrium-89
48
Multiple Choice
49
Multiple Choice
A Bromine ion gains 1 electron, which of the following is the correct symbol for a Bromine ion?
Br-1
Br+1
Br+7
Br-7
50
Multiple Choice

51
Multiple Choice
The properties of elements are periodic functions of their
mass numbers
atomic numbers
atomic masses
atomic radii
Show answer
Auto Play
Slide 1 / 51
MULTIPLE CHOICE
Similar Resources on Wayground

47 questions
9.10 Unit 9 Review Part 1
Presentation
•
11th Grade

45 questions
Commonly Confused Words
Presentation
•
10th - 11th Grade

48 questions
Weathering and Erosion Deposition Landforms
Presentation
•
11th Grade

48 questions
Types of Rock Weathering
Presentation
•
11th Grade

48 questions
Weathering and Erosion Deposition
Presentation
•
11th Grade

45 questions
Unit 1 - Sustainability Review
Presentation
•
11th Grade

47 questions
The Periodic Table
Presentation
•
10th - 12th Grade

45 questions
IB Bio 1.4 Notes - Membrane Transport
Presentation
•
11th Grade
Popular Resources on Wayground

15 questions
Grade 3 Simulation Assessment 1
Quiz
•
3rd Grade

22 questions
HCS Grade 4 Simulation Assessment_1 2526sy
Quiz
•
4th Grade

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

20 questions
Math Review
Quiz
•
3rd Grade
Discover more resources for Science

20 questions
Earth Day Trivia
Quiz
•
9th - 12th Grade

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

20 questions
Earth Day
Quiz
•
3rd - 12th Grade

20 questions
Cladograms and Phylogenetic Trees
Quiz
•
9th - 12th Grade

31 questions
STAAR Strand 1 TEKS(11,12)
Quiz
•
9th - 12th Grade

16 questions
Taxonomy and Classification
Quiz
•
6th - 12th Grade

50 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

37 questions
Hydrosphere CSA wayground review
Quiz
•
9th - 12th Grade