

Gas Laws explained
Presentation
•
Chemistry
•
10th - 12th Grade
•
Easy
Standards-aligned
Shirin Haddadin
Used 9+ times
FREE Resource
5 Slides • 10 Questions
1

2

Include sketches or diagrams to illustrate key concepts and relationships in the gas laws.
3
4
5
6
Multiple Choice
7
Multiple Choice
8
Multiple Choice
9
Multiple Choice
10
Multiple Choice
11
Multiple Choice
12
Multiple Choice
13
Multiple Choice
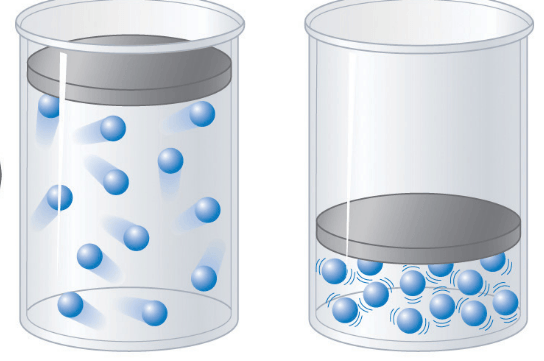
14
Multiple Choice
15
Multiple Choice

Gas particles are in constant motion. They travel in straight lines, and they constantly bounce off the walls of any container that holds them.
What does this constant bombardment of particles against the walls of the container create?
Pressure
Density
Temperature
Magnetism

Show answer
Auto Play
Slide 1 / 15
SLIDE
Similar Resources on Wayground

11 questions
Multistep Stoichiometry
Presentation
•
10th - 12th Grade

11 questions
Bohr Model
Presentation
•
11th - 12th Grade

11 questions
Dalton's Atomic Theory
Presentation
•
10th - 12th Grade

10 questions
Hispanic Heritage Month
Presentation
•
10th - 12th Grade

12 questions
Scientific Method
Presentation
•
9th - 12th Grade

12 questions
Empirical Rule
Presentation
•
10th - 12th Grade

12 questions
Imperfect vs. Preterite
Presentation
•
10th - 12th Grade

10 questions
Poetic Structure
Presentation
•
11th - 12th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Interpreting Solubility Curves
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
10th Grade

43 questions
Acids and Bases Review
Quiz
•
10th - 11th Grade

20 questions
Thermochemistry Review
Quiz
•
10th Grade

25 questions
Unit 4 (Mid) Rumble
Quiz
•
10th Grade