

Phy Science Final Exam Review
Presentation
•
Science
•
9th - 12th Grade
•
Hard
John Oglesby
Used 3+ times
FREE Resource
51 Slides • 66 Questions
1
Physical Science
Final Exam Review
2
What is on the Final?
Periodic Table and Atomic Structure 14 questions x 2 pts = 28 pts
Periodic Table Organization
Atomic Structure
Protons/Neutrons/Electrons
Atoms/Ions/Isotopes
Chemical Reactions 4 questions x 2 pts = 8 pts
Writing and Balancing Equations
Moles & Molar Conversions 2 questions x 4 pts = 8 pts
Moles to grams, gr to moles
Energy 4 questions x 5 pts = 20 pts
Newtons Laws 4 Questions x 2 pts = 8 pts
Essay (Chem Eqns & Reaction types) 2 questions x 5 pts = 10 pts
Last Question Easy 18 pts
3
Periodic Table & Atomic Structure
14 questions x 2 pts each = 28 pts
PT Organization
Atomic Structure - Atomic Number, Atomic Mass,
Atoms / Ions / Isotopes
4

5

6
Multiple Choice
What is a group
Up and down row on the table
Up and down column on the table
Side to side row on table
Side to side column on the table
7
Multiple Choice
What is a period
Up and down row on the table
Up and down column on the table
Side to side row on table
Side to side column on the table
8
Multiple Choice
What group is carbon in?
1
2
13
14
15
9
Multiple Choice
Are the alkline earth metals more or less reactive than the alkali metals
More
Less
10

11

12
Multiple Choice
Which element is a Halogen?
Argon
Iodine
Calcium
Sodium
13
Multiple Choice
Which element is a Noble Gas?
Neon
Chlorine
Sodium
Phosphorus
14
Multiple Choice
Group 1
Alkali Metals
Alkaline Earth Metals
Rare Earth Metals
Noble Gases
Halogens
15
Multiple Choice
Group 17
Alkali Metals
Alkaline Earth Metals
Rare Earth Metals
Noble Gases
Halogens
16
Multiple Choice
Alkali Metals have how many valence electrons
1
2
6
8
17
Multiple Choice
Halogens have how many valence electrons
1
2
7
8
18
Multiple Choice
Noble Gases have how many valence electrons
1
2
6
8
19
Valence electrons:
The electrons in an atom’s outermost orbitals; determine the chemical properties of an element.
-Each group of representative elements (1,2, 13-18) have an assigned amount of valence electrons.

20
Fill in the Blanks
Type answer...
21
Fill in the Blanks
Type answer...
22
Multiple Choice
What charge of ion will Chlorine form?
1-
1+
7-
2-
23
Multiple Choice
What charge of ion will Sodium form?
1-
1+
7-
2-
24
Day 1
What are the sub-atomic particles?
Where do they live?
What is their relative mass?
What is their charge?
What is Atomic Number?
What is Mass Number?
What is an Isotope
What is an Ion
25


26


27


28


29


30


31
Multiple Choice
The negatively charge (-) particle of an atom.
proton
electron
neutron
32
Multiple Choice
The no charge (n0) particles of an atom.
Proton
Electron
Neutron
33
Multiple Choice
The positively charge (+) particles of an atom
Proton
Electron
Neutron
34
Multiple Choice
Protons and neutrons are found inside nucleus
True
False
35
Multiple Choice
nucleus
proton
isotope
molecule
36


The number of Protons DEFINES the element.
Helium has 2 PROTONS. Helium will never have 3 protons. If it did...it would be LITHIUM!
37
Multiple Choice
The atomic number of Hydrogen (H) is
1
2
3
4
38
Multiple Choice
The atomic number of Nitrogen (N) is
7
6
8
9
39
Multiple Choice
The atomic number of Cobalt (Co) is
27
26
25
24
40
Fill in the Blanks
Type answer...
41
An ATOM is Neutral. That means it has no charge.
So...It has to have the same number of electrons as it has protons!
P +1
e -1
Net Charge is 0
42


So atoms will have the same number of ELECTRONS as they do PROTONS!
43
Multiple Choice
How many electrons does a neutral atom of Nitrogen have?
14
7
8
6
44
Multiple Choice
What is the charge of a proton, and what is its location in an atom?
Neutral charge, located in nucleus.
Positive charge, located in nucleus.
Neutral charge, located in the orbitals/cloud.
Positive charge, located in the orbitals/cloud.
45
Multiple Choice
WHAT IS THE ATOMIC NUMBER OF OXYGEN?
16
8
15.9999
9
46
What is an Isotope?
47

Isotopes have the same number of protons (so they are the same element)
But different numbers of Electrons
48
Isotope Symbols
As you can see to the right there are certain ways elements and isotopes are represented as symbols. There is the element symbol (the biggest part). Then the upper number will the the mass number (number of protons + number of neutrons) and the lower number is just the atomic number (number of protons).

49


50
Multiple Choice
You find out that an atom has 5 protons, 6 neutrons, and 5 electrons. What is this atom's atomic number?
5
6
11
1
51
Average Atomic Mass
DIFFERENT than Mass Number
Average of all of the isotopes of an atom found in nature.

52
Multiple Choice
What is the atomic number of Nitrogen
7
14
28
5
53
Multiple Choice
What is the atomic number of carbon
18
12
5
6
54
Multiple Choice
What is the atomic mass of carbon
6 g/mole
12 g/mole
18 g/mole
23 g/mole
55
Chemical Reactions
4 Questions x 2 Pts each = 8 Pts
Writing Chemical Equations
Balancing Chemical Equations
Types of Chemical Reactions
56
Writing Chemical Equations
8E: Write and balance chemical equations using the law of conservation of mass.
57
58
DIATOMIC ELEMENTS


59
Multiple Select
Which of the following elements are considered diatomic? Check all.
Phosphorous
Nitrogen
Iodine
Carbon
60
Drag and Drop
Nitrogen reacts with Hydrogen to form Ammonia (NH3).
61
Drag and Drop
62
Drag and Drop

63
Drag and Drop

64
Multiple Choice
Which is the correct chemical equation for the following: Barium Chloride combined with sodium sulfide yields barium sulfide and sodium chloride
BaCl + NaS → BaS+ NaCl
BaCl2 + Na2S → BaS+ NaCl
BaS+ NaCl → BaCl + NaS
BaCl2 + Na2S → BaS+ NaCl2
65

66
2H2 + O2 ---> 2H2O
This equation is BALANCED on both sides.
Notice that there are 4 Hydrogens and 2 Oxygens on each side.
The full-size numbers to the LEFT of the elements/compounds are called COEFFICIENTS and are the ONLY VALUES YOU CAN CHANGE.
The small numbers to the right of the elements are called SUBSCRIPTS and MUST STAY THE SAME in the equation.
67

68

69
In this example, we want the number of Mg (Magnesium) on the LEFT of the arrow to be the same on the RIGHT of the arrow.
We also want the number of O (Oxygen) to be the same number on both the LEFT and RIGHT of the arrow.
70
Once we count up the Mg and O on both sides, we can see what needs to change.
We CANNOT TAKE ANYTHING AWAY.
We can ONLY ADD using coefficients.
You'll see this done in the next slide...
71

72

73
Multiple Choice
Mg + __HCl ---> MgCl2 + H2
74
Multiple Choice
Is the following equation balanced?
Al + O2 ---> 2Al2O3
YES
NO
75
Multiple Choice
What coefficients are needed to balance the equation?
___Al + ___ O2 ---> 2Al2O3
3,2
3,4
4 , 3
4,2
76
Multiple Choice
What coefficients are needed to balance the following chemical equation?
__H2O → __H2 + __O2
1, 1, 1
2, 2, 2
2, 2, 1
2, 2, 3
77
Multiple Choice
What coefficients are needed to balance the following chemical equation?
__Zn + __HCl → __ZnCl2 + __H2
1,1,1,1
1,2,1,1
2,1,2,2
2,1,2,1
78
Multiple Choice
4Fe + 3O2 --> 2Fe2O3
79
Types of Chemical Reactions
Access Types of Chemical Reactions Notes from Google Drive!

80

81

82

83

84

85
Multiple Choice
86
Multiple Choice
Al(OH)3 → Al2O3 + H2O
87
Multiple Choice

What type of reaction involves 2 substances combining to form 1 new compound?
Decomposition Reaction
Single Displacement Reaction
Double Displacement Reaction
Synthesis Reaction
88
Multiple Choice
2Na +S → Na2S
Double Replacement
Decomposition
Combustion
Synthesis
89
Multiple Choice

90
Multiple Choice

Identify the type of chemical reaction
Synthesis
Decomposition
Single replacement
Double replacement
Combustion
91
Multiple Choice

Identify the type of chemical reaction
Synthesis
Decomposition
Single replacement
Double replacement
Combustion
92

93
Multiple Choice
What reaction has the following general formula:
A + CD --> C + AD
Combination
Decomposition
Single Displacement
Double Displacement
Combustion
94
Multiple Choice
What type of reaction is the following:
3CuS +2Al −> Al2S3+3Cu
Combination
Decomposition
Single Displacement
Double Displacement
Combustion
95
Multiple Choice
96
Energy
GPE & KE
97

98

99
Math Response
A 16-kg barbell is now 0.5 m above the floor. What is it's Gravitational Potential Energy in Joules?
100
Multiple Choice
What kind of energy would you find in an object high above the ground?
Gravitational Potential
Kinetic
Thermal
Chemical
101
Multiple Choice
102
Multiple Choice
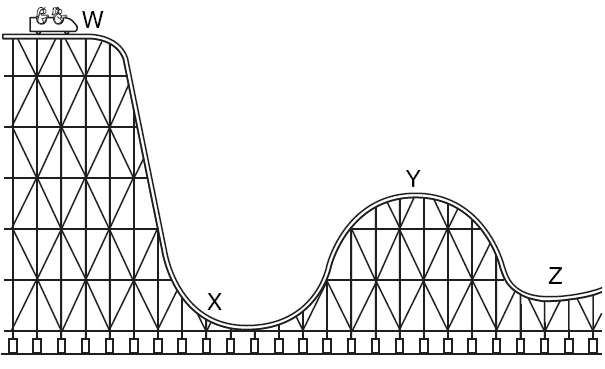
103

104
Math Response
Calculate the gravitationals PE of a 2.5 kg monkey hanging from a branch 7 m above the jungle floor
GPE = 2.5 x 9.8 x 7m = ___J
105

106
Math Response
Use the formula KE = 0.5 x mass x velocity x velocity
how much KE in a 0.15 kg baseball the is thrown at 35 m/s
KE = 0.5 x 0.15kg x 35 m/s x 35 m/s
107

108

What is the kinetic enrgy of a 44 kg cheetah running at a speed of 31 m/s
Use Formula KE = 0.5 x m x v x v
Calculate Kinetic energy
109
Forces LT5 - Newton's 2nd Law Calculations
Physical Science 9A

110

111
Open Ended
A baseball accelerates downward at 9.8 m/s2. With negligible air resistance and a net force of 1.4 N, what is the baseball’s mass?
F=ma112
Multiple Choice
An object accelerates 8 m/s2 when a force of 40 newtons is applied to it. What is the mass of the object?
4 kg
20 kg
0.2 kg
5 kg
113
Multiple Choice
When force is 20 N, mass is 5 kg, what is acceleration?
F = ma
25 m/s2
100 m/s2
5 m/s2
4 m/s2
114
Multiple Choice
An object of mass 10 kg is accelerated upward at 2 m/s2. What force is required?
20 N
5 N
2 N
0 N
115
Multiple Choice
Which object has greater inertia?
116
Multiple Choice
What is inertia?
Inertia is an object’s resistance to change in motion.
Inertia is anything that makes a moving object slow down or stop.
117
Multiple Choice
What will happen to an object if it is put into motion in space?
It will stop eventually due to friction.
It will continue to move until it is acted on by an unbalanced force.
It will speed up over time.
It will slow down over time.
Physical Science
Final Exam Review
Show answer
Auto Play
Slide 1 / 117
SLIDE
Similar Resources on Wayground

108 questions
Heat, Energy, and Temperature
Presentation
•
9th - 12th Grade

111 questions
English IV End of Semester Review
Presentation
•
9th - 12th Grade

106 questions
Ecology Vocabulary Review
Presentation
•
9th - 12th Grade

106 questions
Punnett Square and Blood Types
Presentation
•
9th - 12th Grade

106 questions
Blood Type Genetic Problems Lessons and Quizzes
Presentation
•
9th - 12th Grade

108 questions
SPANISH 1 STUDY GUIDE 2021
Presentation
•
9th - 12th Grade

108 questions
Temperature and Heat
Presentation
•
9th - 12th Grade

107 questions
FSA Review - Foundation #2: Angles, Parallel & Transversals
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

25 questions
The Ultimate College Knowledge Quiz
Quiz
•
8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Math Review
Quiz
•
6th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade
Discover more resources for Science

20 questions
MCAS Biology Review
Quiz
•
9th - 10th Grade

21 questions
Explore DNA Processes and Protein Formation
Quiz
•
9th - 12th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

50 questions
living environment regents review
Quiz
•
9th Grade

17 questions
Unit 9 - Chemical and Nuclear Reactions
Quiz
•
9th - 12th Grade

50 questions
chem final review
Quiz
•
9th Grade

10 questions
Exploring Ecological Levels: Organism to Ecosystem
Interactive video
•
6th - 10th Grade

233 questions
BioPhysics B - Final Exam
Quiz
•
9th - 12th Grade