

Heat Energy
Presentation
•
Chemistry
•
9th - 12th Grade
•
Hard
James Gonzalez
FREE Resource
10 Slides • 98 Questions
1
Multiple Choice

2
Multiple Choice
3
Multiple Choice
4
Multiple Choice
5
Multiple Choice

6
Multiple Choice
7
Multiple Choice
8
Multiple Choice
9
Multiple Choice
10
Multiple Choice
Determine the heat energy needed to heat the water in the pot to boiling.
11
Multiple Choice
The temperature of a sample of water increases from 20°C to 46°C as it absorbs 5650 joules of heat. What is the mass of the sample? (Specific heat of water is 4.18 J/g°C.)
51.98 g
65.3 g
51.99 g
12
Multiple Choice

A 4.50 g coin of copper absorbed 254 Joules of heat. What was the change in temperature of the copper? The specific heat of copper is .385 J/goC.
210 oC
146.61 oC
244.8 oC
13
Multiple Choice

Which will heat up faster?
copper
granite
iron
basalt
14
Multiple Choice

Which will heat up slower?
water
lead
granite
iron
15
Multiple Choice
16
Multiple Choice
Which of the following best explains why the sand at the beach is hotter than the water?
Sand has a higher specific heat than water.
Sand has a lower specific heat than water.
There is more water than sand at the beach.
There is more sand than water at the beach.
17
Multiple Choice
The amount of energy required to raise the temperature 1ºC for every gram is called____?
Thermal Energy
Specific Heat
Temperature
Kinetic Energy
18
Multiple Choice

19
Multiple Choice

20
Multiple Choice
21
Multiple Choice

The diagrams shown represents two solids and the temperature of each. What occurs when the two solids are placed in contact with each other?
Heat energy flows from solid A to solid B.
Heat energy flows from solid A to solid B. Solid A increases in temperature.
Heat energy flows from solid B to solid A. Solid B decreases in temperature.
Heat energy flows from solid B to solid A.
22
Drag and Drop
60°C
70°C
80°C
23
Multiple Choice
Which term represents a form of energy?
heat
degree
kilocalorie
temperature
24
Multiple Choice
Which form of energy is converted to thermal energy when propane burns in air?
electromagnetic
nuclear
electrical
chemical
25
Multiple Choice
26
Multiple Choice

27
Multiple Choice

28
Multiple Choice
29
Multiple Choice
30
Multiple Choice
31
Multiple Choice
C
32
Multiple Choice
33
Multiple Choice
34
Multiple Choice

According to the ESRT, which material would require the most heat energy to increase the temperature of 1 gram of the material one Celsius degree?
Ice
Water
Basalt
Granite
35
Multiple Choice

Which material would require the greatest amount of heat energy to raise its temperature from 5oC to 10oC? [Refer to the Earth Science Reference Tables.]
Iron
Lead
Granite
Dry Air
36
Multiple Choice

If the same amount of heat is added to each sample, the smallest change in temperature will occur in the
Iron
Dry Air
Granite
Water
37
Multiple Choice

What would heat up the fastest if left out in the sunlight on a clear, summer day?
Liquid Water
Dry Air
Lead
Granite
38
Multiple Choice
Why does water heat heat up and cool down VERY SLOWLY?
High Specific Heat
Low Specific Heat
It's solid
It's always cold
39
Multiple Choice
40
Multiple Choice
41
Multiple Choice
Heat ALWAYS moves from
Low to High Temperature
High to Low Temperature
It doesn't move
High to Low Pressure
42
Multiple Choice
Specific Heat is....
The speed of the particles in a substance
The temperature of the substance
How much energy it takes to heat something up
Heat
43
Multiple Choice
When heat is absorbed by a substance, what happens to the temperature of the substance?
it increases
it stays the same
it decreases
it depends on the substance
44
Multiple Choice
When heat is released by a substance, what happens to its temperature?
it increases
it stays the same
it decreases
it depends on the substance
45
Multiple Choice
46
Multiple Choice
47
Multiple Choice
48
Multiple Choice
49
Multiple Choice
50
Multiple Choice
51
Multiple Choice
52
Multiple Choice
The unit for temperature is
K or °C
J
J/gC
g or kg
53
Multiple Choice
What letter do we use to represent mass?
Q
m
c
ΔT
54
Multiple Choice
What does ΔT represent?
tf −ti
ti −tf
t - celsius
celsius - t
55
Multiple Choice
When is your answer negative?
When the sample is cooled or loses heat energy.
When the sample heats up or gains heat energy.
When the sample absorbs heat energy.
56
Multiple Choice
57
Multiple Choice

58
Multiple Choice
59
Multiple Choice

60
Multiple Choice

61
Multiple Choice

62
Multiple Choice

63
Multiple Choice

64
Multiple Choice
65
Multiple Choice

66
Multiple Choice

67
Multiple Choice

68
Multiple Choice
A material was cooled from 100ºC to 40ºC. What is the temperature change? (Remember: Change in temperature = T final - T initial )
60ºC
40ºC
-60ºC
-40ºC
69
Multiple Choice

70
Multiple Choice

71
Multiple Choice

Observe the diagram. Which statement is true about the kinetic energy of the molecules in the containers?
The molecules are moving faster in Diagram A
The molecules are moving slower in Diagram A
The molecules are moving at the same rate in both diagrams
72
Multiple Choice
73
Multiple Choice

74
Multiple Choice

75
Multiple Choice

Thermal energy
Temperature
Heat
Convection
76
Multiple Choice
Identify the variables in this problem.
Find the specific heat of a substance that releases 7852 J when 532g cools from 80°C to 70°C.
q = 10°C, m = 532g
q = 7852J, ΔT = 10°C
q = 10°C, m = 532g, ΔT = 80°C
77
The cool thing is, this is the only formula you need!
q = mcΔT
78
Multiple Choice
79
Multiple Choice
80
Multiple Choice
81
Multiple Choice
82
Multiple Choice
83
Multiple Choice
84
Multiple Choice
85
Multiple Choice
Is this reaction endothermic or exothermic?
86
Multiple Choice
Is this reaction endothermic or exothermic?
87
Multiple Choice
88
Multiple Choice
→
89
Multiple Choice

Which state of matter does this picture show?
Solid
Liquid
Gas
90
Multiple Choice
When particles are heated they,
expand and move apart
expand and move together
contract and move apart
contract and move together
91
Multiple Choice
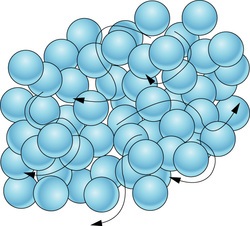
What state of matter does this picture show?
Solid
Liquid
Gas
92
Multiple Choice
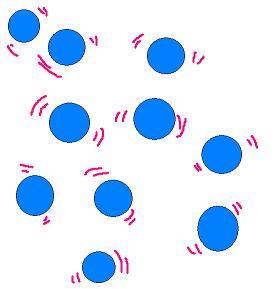
Which state of matter does this picture show?
Solid
Liquid
Gas
93
Multiple Choice

What is happening in this picture of water boiling?
Molecules are speeding up
Molecules are slowing down
94
Multiple Choice

What is happening in this picture of water boiling?
Molecules are speeding up
Molecules are slowing down
95
Multiple Choice
96

97

98
Multiple Choice
Increasing the mass of an object will increase its temperature
True
False
99
Multiple Select
Which of the following will increase the thermal energy of an object. (May be more than one answer)
increasing the temperature
increasing the mass
reducing the mass
reducing the temperature
100

101

102

103

104

105
Match
Specific Heat Capacity
Temperature
Thermal Energy
Convection
Conduction
the amount of energy it takes to raise the temperature of a substance
the average amount of kinetic energy in particles
the total amount of kinetic energy in particles
the movement of fluids (gases and liquids) due to temperature differences
the transfer of thermal energy through collisions of particles when objects touch
the amount of energy it takes to raise the temperature of a substance
the average amount of kinetic energy in particles
the total amount of kinetic energy in particles
the movement of fluids (gases and liquids) due to temperature differences
the transfer of thermal energy through collisions of particles when objects touch
106

107
Fill in the Blanks
Type answer...
108
How much heat is needed to raise a 0.30 kg piece of aluminum from 30℃ to 150℃?
Formula: Q = mcΔT
Solving Specific Heat Problems
Some text here about the topic of discussion

Show answer
Auto Play
Slide 1 / 108
MULTIPLE CHOICE
Similar Resources on Wayground

100 questions
Kinetics A2
Presentation
•
9th - 12th Grade

96 questions
WW1
Presentation
•
9th - 12th Grade

105 questions
History of Floral Design
Presentation
•
9th - 12th Grade

103 questions
Int Science SEM 1 FINAL REVIEW
Presentation
•
9th - 12th Grade

106 questions
GRAMMAR - PASIVE VOICE (ADVANCED)
Presentation
•
10th Grade - University

105 questions
Lesson 2: Conventional and 21st Century Genres
Presentation
•
11th Grade

109 questions
Analisis Data
Presentation
•
KG - University

102 questions
Virginia Geology & Dating Review
Presentation
•
11th Grade - University
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Chemistry

25 questions
Waygrounds 5/20 PS.8.1.2
Quiz
•
9th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

20 questions
Naming & Writing Chemical Formulas
Quiz
•
10th Grade

15 questions
Types of chemical reactions
Quiz
•
9th - 12th Grade

32 questions
Acids/Bases, Electrochem, Orgo
Quiz
•
9th - 12th Grade