

ATOMIC GRADE 7 REVIEW
Presentation
•
Science
•
•
Practice Problem
•
Hard
Noble Teacher
FREE Resource
36 Slides • 19 Questions
1

2

3

4

5

6

7

8

9

10

11

12

13

14

15

16
Multiple Choice
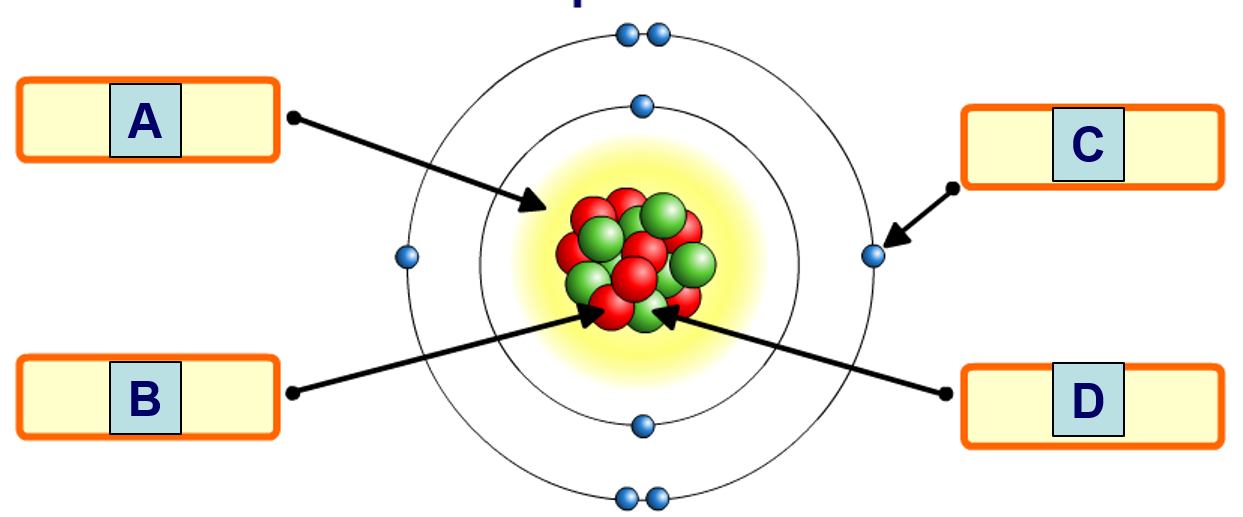
Which of the following is the nucleus?
A
B
C
D
17
Multiple Choice

What particle is B?
electron
proton
nucleus
neutron
18
Fill in the Blanks

Type answer...
19
Multiple Choice

One particle in an atom is electron, which one is it?
A
B
C
D
20
Atomic Mass and Mass Number
Module 3 - Physical Science

21
The Periodic Table of Elements
The Periodic Table is an organizational tool used by scientists to place elements into different categories.
Elements are the building blocks of matter, each having different chemical and physical properties.
Today, we will be looking at what atoms are composed of, and how we describe those atoms.

22
Review
The atomic number is the number of protons in the element.
The atomic mass is the number of protons and neutrons in one atom of that element.
All elements are electrically neutral, meaning they have ZERO NET CHARGE. This means that the number of protons (positive charges) are equal to the number of neutrons (negative charges)

23
Review
The element Helium (He) has an Atomic Number of 2. This means that all Helium atoms have exactly two protons.
Since the atom is electrically neutral, that means the atom also has exactly two electrons.
How do we determine how many neutrons there are in an atom?

24
Review
Atomic Mass = Protons + Neutrons
We round the atomic mass to the nearest whole number, 4
Since we already determined that Helium has two protons, that means: 4=2+Neutrons
There must be 2 Neutrons in one atom of Helium

25
Let's Practice Together!
What is the Atomic Number?
What is the Atomic Mass?
How many protons are in a Lithium Atom?
How many electrons are in a Lithium Atom?
What is the rounded Atomic Mass?
How many neutrons are in a Lithium Atom?

26
Let's Practice Together!
What is the Atomic Number? 3
What is the Atomic Mass? 6.941
How many protons are in a Lithium Atom? 3
How many electrons are in a Lithium Atom? 3
What is the rounded Atomic Mass? 7
How many neutrons are in a Lithium Atom? 7-3=4

27
Multiple Choice

What is the Atomic Number for Beryllium (Be)?
4
9.012
9
5
28
Multiple Choice

What is the Atomic Mass for Beryllium (Be)?
4
9.012
9
5
29
Multiple Choice

How many Protons are in one atom of Beryllium (Be)?
4
9.012
9
5
30
Multiple Choice

How many Electrons are in one atom of Beryllium (Be)?
4
9.012
9
5
31
Multiple Choice

What is the Rounded Mass of Beryllium (Be)?
4
9.012
9
5
32
Multiple Choice

How many neutrons are in one atom of Beryllium (Be)?
4
9.012
9
5
33
Elements, Compounds and Mixtures
U1L1

34
Elements
There are more than 100 different different elements
An element is a type of atom with the same number of protons
The term element is also used to describe a substance that is made of the same type of atom
Elements are pure chemical substances.

35
Compound
Elements can chemically combine to form new, pure chemical substances.
A chemical substance that is made of two or more different elements that chemically combine together is called a compound
The force between compounds is STRONGER than between mixtures


36
Mixture
A compound is NOT the same as a mixture
A mixture is a physical combination of two or more different chemical substances.
A compound is a chemical combination of two or more different chemical substances (specifically, elements)
Unlike an element and compound, a mixture is NOT a pure chemical substance


37

38
Multiple Choice

This is an example of a(n)...
Element
Compound
Mixture
Atom
39
Multiple Choice

This is an example of a(n)...
Element
Compound
Mixture
Molecule
40
Multiple Choice

A(n) _____ is a type of atom with the same number of protons
Element
Compound
Mixture
Molecule
41
Multiple Choice

A chemical substance that is made of two or more DIFFERENT elements that chemically combine together
Element
Compound
Mixture
Molecule
42

All of the known elements of the world can be found on the periodic table.
In different combinations, they are the building blocks of all matter!
Elements
43

Compounds are two or more DIFFERENT elements chemcially bonded together. This means that they cannot be separated easily.
Compounds
44

A mixture is a material made up of two or more different substances which are not chemically bonded. This means that the substances keep their original properties.
Mixtures
45

General properties of a mixture:
● The components of a mixture can be easily separated
● The components each keep their original properties
Characteristics of Mixtures
46
Multiple Select
Which of the following are examples of Mixtures?
The atmosphere
(N + O + Ar + H2O...)
Carbon Dioxide (CO2)
Oxygen
(O2)
Lemonade
(sugar, lemon juice, water)
Anything on the Periodic Table
47
Multiple Select
Which of the following are examples of ELEMENTS? Choose all that are correct.
Oxygen (O)
Water
(H2O)
Flourine
(F)
Lemonade
Sulfur Dioxide (SO2)
48
Multiple Select
Which of the following are examples of compounds? Check all that apply.
Salt
(NaCl)
Salt Water
(NaCl and H2O)
Sugar
(C6H12O6)
Carbon Dioxide
(CO2)
49
Chemical Compounds
By David Stanford
50
Chemical Compounds
The element names are represented by a single uppercase letter or an uppercase letter followed by a lowercase letter.
Example: Ne (neon), C (carbon), Pb(lead)
Some text here about the topic of discussion
51
Chemical Compounds
Subscripts are the numbers written on the lower right side of the element symbol, this tells you the number of atoms of that element.
Example: O2 (two atoms of oxygen)
Some text here about the topic of discussion
52
Chemical Compounds
If an element symbol has no subscript there is only one atom of that element in that molecule.
Example: O2 (two atoms of oxygen) O (one atom of oxygen)
H2O (2 atoms hydrogen and 1 atom oxygen)
Some text here about the topic of discussion
53
Chemical Compounds
If an element symbol has no subscript there is only one atom of that element in that molecule.
Example: O2 (two atoms of oxygen) O (one atom of oxygen)
H2O (2 atoms hydrogen and 1 atom oxygen)
Some text here about the topic of discussion
54
Multiple Choice
How many atoms of oxygen are in H2O?
1
2
3
4
55
Multiple Choice
How many atoms of hydrogen are in H2O?
1
2
3
4

Show answer
Auto Play
Slide 1 / 55
SLIDE
Similar Resources on Wayground

51 questions
Solar Radiation
Presentation
•
6th Grade

51 questions
Chemistry 1-4: Electrons in Atoms
Presentation
•
10th - 12th Grade

48 questions
Magnets
Presentation
•
8th Grade

51 questions
DESIGNING THE EXPERIMENT
Presentation
•
7th Grade

53 questions
Minerals
Presentation
•
9th - 12th Grade

47 questions
Seasons on Earth
Presentation
•
6th - 8th Grade

46 questions
Calculating Net Forces GuidedLesson
Presentation
•
6th - 8th Grade

49 questions
12.2 Fossils and Geological time
Presentation
•
9th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade