

TEAS Test Study Guide - Chemistry
Presentation
•
Biology
•
University
•
Practice Problem
•
Easy
SN Goebel
Used 4+ times
FREE Resource
16 Slides • 119 Questions
1
TEAS Test Study Guide - Chemistry
2
Multiple Choice

What is the study of matter, especially its chemical reactions, but also its composition, structures and properties?
Biology
Physics
Chemistry
Geology
3
Multiple Choice

What are the three states of matter?
Solid, liquid, gas
Plasma, gas, solid
Liquid, gas, plasma
Solid, plasma, liquid
4
Multiple Choice

Which of the following is the definition of a chemical reaction?
The physical change of a substance without changing its composition
A process that rearranges the atoms of one or more substances to form one or more different substances
The combining of two or more substances to form a new substance
The breaking down of a substance into simpler substances
5
Multiple Choice

What is the purpose of balancing chemical equations?
To ensure that the total number of atoms on each side of the equation is the same
To make the equation more accurate
To follow a standard procedure in chemistry
To fulfill a requirement in chemical experiments
6
Multiple Choice

What are the smallest units of an element that retain the element's chemical properties?
Cells
Tissues
Organs
Atoms
7
Multiple Choice

What is an element?
A type of atom
A substance that can be broken down into simpler substances by chemical means
A substance that cannot be broken down further into simpler substances by chemical means
A substance that can be broken down into simpler substances by physical means
8
Multiple Choice

Which of the following is NOT a subatomic particle?
Electron
Proton
Protein
Neutron
9
Multiple Choice

Which subatomic particle determines the type of element?
Photon
Electron
Neutron
Proton
10
Multiple Choice

What is the total number of protons in an element called?
Atomic mass
Electron count
Atomic number
Neutron count
11
Multiple Choice

What is the atomic mass of an element?
The total number of atoms in an element.
The total number of neutrons in an element.
The average mass of an atom of an element, taking into account all of its isotopes.
The number of protons in an atom of an element.
12
Multiple Choice

How do you calculate the number of neutrons in an atom?
Add the number of electrons to the atomic number.
Divide the atomic number by the atomic mass.
Subtract the atomic number (# of protons) from the atomic mass.
Multiply the atomic number by the atomic mass.
13
Multiple Choice

What is an isotope?
An isotope is a form of an element that has a different number of protons.
An isotope is a form of an element that has a different number of protons and neutrons.
An isotope is a form of an element that has a different number of electrons.
An isotope is a form of an element that has the same number of protons but a different number of neutrons.
14
Multiple Choice

Where is most of an atom's mass located?
In the nucleus
In the atomic orbitals
In the outer energy shells
In the electron cloud
15
Multiple Choice

Where do the electrons of an atom reside?
Nucleus
Neutrons
Electron shells (orbitals)
Protons
16
Multiple Choice

Which of the following best defines the valence shell?
The nucleus of an atom
The innermost shell of an atom
The region where protons are found
The outermost shell of an atom that contains electrons
17
Multiple Choice

Why is the valence shell important in chemical bonding?
It determines the temperature at which an atom reacts
It determines the color of an atom
It determines the number of neutrons an atom has
It determines the types of chemical bonds an atom can form
18
Multiple Choice

What happens when an atom's valence shell is full?
The atom becomes stable and less reactive
The atom loses all its electrons
The atom becomes highly reactive
The atom becomes larger in size
19
Match

Match the main properties of subatomic particles with their correct descriptions:
Positive charge, located in the nucleus
No charge, located in the nucleus
Negative charge, located in the orbitals
Proton
Neutron
Electron
Proton
Neutron
Electron
20
Multiple Choice

Which of the following statements best describes ions?
Ions are atoms or molecules that have gained or lost neutrons, resulting in a change in their atomic mass.
Ions are atoms or molecules that have gained or lost electrons, resulting in a net positive or negative charge.
Ions are atoms or molecules that have gained or lost electrons, resulting in a change in their atomic structure.
Ions are atoms or molecules that have gained or lost protons, resulting in a change in their atomic number.
21
Multiple Choice

What are compounds?
Substances that cannot be chemically broken down into simpler substances.
Substances that have a fixed composition.
Substances made up of two or more different elements chemically combined.
Substances made up of only one element.
22
Multiple Choice

What is a molecule?
A molecule is a group of atoms bonded together, representing the smallest fundamental unit of a chemical compound that can take part in a chemical reaction.
A molecule is a type of cell found in the human body.
A molecule is a tiny particle that can only be seen through a microscope.
A molecule is a basic unit of energy in the human body.
23



Metals give up electrons, they become positively charged ions; Non-metals gain those electrons and become negatively charged ions. The oppositely charged ions become attracted to each other forming a bond.
Ionic bonds happen between metals and nonmetals
Ionic
Occur when atoms with only partially filled valence shell electrons, come together to share electrons; only nonmetals participate in covalent bonds
Covalent
it is a polar covalent bond. The hydrogen covalent bonds with another electronegative atom (like oxygen or nitrogen). the other molecule becomes partially negative while the hydrogen becomes partially positive. It's the opposite charges that cause the atoms to bond together
Hydrogen
Three Main Types of Bonds
24
Multiple Choice

Which of the following best describes a covalent bond?
Unequal sharing of electrons between two non-metal atoms
Attraction between metal cations and delocalized electrons
Sharing of electrons between two non-metal atoms
Transfer of electrons from a metal to a non-metal
25
Multiple Choice

Which of the following is NOT a characteristic of covalent bonds?
They can form single, double, or triple bonds
They result in a stable molecule
They are strong bonds
They form between a metal and a nonmetal
26
Multiple Choice
Which of the following molecules is an example of a compound held together by covalent bonds?
Calcium carbonate (CaCO3)
Sodium chloride (NaCl)
Water (H2O)
Magnesium oxide (MgO)
27
Multiple Choice

What type of covalent bond occurs when two atoms share more than two pairs of electrons?
Triple bond
Single bond
Ionic bond
Double bond
28
Multiple Choice

How many electrons are shared in a single covalent bond?
3 electrons
4 electrons
1 electron
2 electrons
29
Multiple Choice
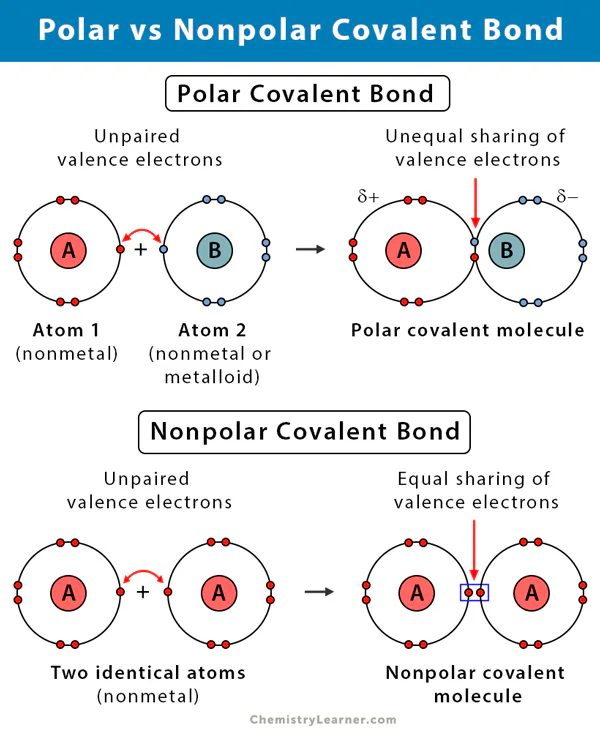
What is the name given to a covalent bond in which electrons are shared equally between atoms?
Ionic bond
Hydrogen bond
Polar covalent bond
Nonpolar covalent bond
30
Multiple Choice

What are polar covalent bonds?
Bonds formed between atoms with different valence electrons, resulting in the formation of ions.
Bonds formed between atoms with opposite charges, resulting in a transfer of electrons.
Bonds formed between atoms with different electronegativities, resulting in an unequal sharing of electrons.
Bonds formed between atoms with similar electronegativities, resulting in an equal sharing of electrons.
31
Multiple Choice

What is electronegativity?
A measure of an atom's ability to attract electrons in a chemical bond.
A measure of an atom's ability to donate electrons.
The measure of an atom's size.
The measure of an atom's atomic mass.
32
Multiple Choice

What are hydrogen bonds?
Physical bonds formed by hydrogen molecules.
Ionic bonds formed between hydrogen and oxygen atoms.
Strong covalent bonds that hold hydrogen atoms together.
Weak electrostatic attractions between a hydrogen atom and an electronegative atom.
33

34
Multiple Choice
Hydrogen bonds occur between a hydrogen atom and which other atom?
Carbon
Sodium
Potassium
Nitrogen, oxygen, or fluorine
35
Multiple Choice

Which property of water helps to moderate temperature changes in the body?
Low heat of fusion
High specific heat
Low boiling point
High vapor pressure
36
Multiple Choice
Which term refers to the amount of energy needed to change the temperature of a substance by 1 degree Celsius (It determines how quickly a substance heats up or cools down)?
Conduction
Radiation
Specific Heat
Latent Heat
37
Multiple Choice

Which property of water makes it an effective solvent?
Viscosity
Transparency
Surface tension
Polarity
38
Multiple Choice

Which property of water allows it to have a high surface tension?
Tension
Adhesion
Capillarity
Cohesion
39
Multiple Choice

Which property of water allows it to stick to the sides of a glass?
Adhesion
Cohesion
Surface tension
Capillary action
40
Multiple Choice

What is the main difference between cohesion and adhesion of water?
Cohesion is the attraction between water molecules and other substances, while adhesion is the attraction between two different water molecules.
Cohesion is the attraction between water molecules, while adhesion is the attraction between water molecules and other substances.
Cohesion is the attraction between water molecules, while adhesion is the attraction between two different water molecules.
Cohesion is the attraction between water molecules and other substances, while adhesion is the attraction between water molecules.
41
Multiple Choice

Which property of water allows it to store large amounts of heat without a significant temperature change?
Surface tension
High heat capacity
Cohesion
Adhesion
42
Multiple Choice

Which of the following best describes the process of capillary action of water?
The movement of water across a semipermeable membrane
The absorption of water by plant roots
The breakdown of water molecules into hydrogen and oxygen
The ability of water to move up through small tubes against gravity
43


44
Multiple Choice

What is the main difference between hydrophilic and hydrophobic substances when placed in water?
There is no difference between hydrophilic and hydrophobic substance reactions in water.
Hydrophilic substances and hydrophobic substances behave the same when placed in water.
Hydrophilic substances dissolve or mix well with water, while hydrophobic substances repel water and do not mix with it.
Hydrophilic substances repel water and do not mix with it, while hydrophobic substances dissolve or mix well with water.
45
Multiple Choice

What are ionic bonds?
A type of bond formed between two metals.
A type of chemical bond formed between two ions with opposite charges.
A type of bond formed between two nonmetals.
A type of bond formed between two atoms with the same charge.
46
Multiple Choice

Which of the following elements commonly form ionic bonds?
Noble gases.
Nonmetal and nonmetal.
Metal and metal.
Metal and nonmetal.
47
Multiple Choice

In an ionic bond, what happens to the electrons?
Electrons are transferred from the metal to the nonmetal.
Electrons are eliminated.
Electrons are shared between the metal and the nonmetal.
Electrons are neutralized.
48
Multiple Choice
What is the overall charge of an ionic compound?
Positive.
It varies depending on the elements involved.
Neutral.
Negative.
49
Multiple Choice

Which type of substances conduct electricity in solution?
Ionic substances
Covalent substances
Polar covalent substances
Metallic substances
50
Multiple Choice

How are ions formed?
Ions are formed by the action of hormones in the endocrine system.
Ions are formed when atoms gain or lose electrons, resulting in a change in their overall charge.
Ions are formed through a process called osmosis.
Ions are formed through the breakdown of proteins in the body.
51
Multiple Choice

What happens to the charge of an atom when it gains an electron?
The charge of the atom remains the same.
The charge of the atom becomes more positive (cation)
The charge of the atom becomes more negative (anion).
The charge of the atom becomes neutral.
52
Multiple Choice

What happens to the charge of an atom when it loses an electron?
Its charge becomes neutral
Its charge remains the same
It becomes negatively charged (anion)
It becomes positively charged (cation)
53
Multiple Choice

Which of the following best describes a salt?
A special type of rock found deep within the Earth.
A type of seasoning used to enhance the taste of food.
A compound formed from the reaction between an acid and a base, consisting of positive and negative ions.
A form of crystallized water.
54
Multiple Choice

What is pH?
The amount of water in a solution
The concentration of hydroxide ions in a solution
A measure of acidity or alkalinity of a solution
The concentration of hydrogen ions in a solution
55
Multiple Choice

What pH level is considered alkaline?
Above 7
7
Between 2 and 12
Below 7
56
Multiple Choice

What pH level is considered acidic?
Above 7
7
Between 2 and 12
Below 7
57
Multiple Choice

What pH level is considered neutral?
Above 7
7
Between 2 and 12
Below 7
58
Multiple Choice

According to the Arrhenius theory, what defines an acid?
Substance that produces hydroxide ions (OH-) in water
Substance that accepts electrons
Substance that releases carbon dioxide (CO2)
Substance that produces hydrogen ions (H+) in water
59
Arrhenius theory states that an acid is a substance that produces hydronium ions when it is dissolved in water.
It also states that a base is a substance that produces hydroxide ions when dissolved in water.
Arrhenius Theory

60
Multiple Choice

According to the Arrhenius theory, what defines a base?
Substance that produces hydrogen ions (H+) in water
Substance that donates electrons
Substance that releases oxygen (O2)
Substance that produces hydroxide ions (OH-) in water
61
Acids are substances that donate a positive hydrogen ion to another substance in a chemical reaction.
A base is the substance that receives that hydrogen ion.
Bronsted-Lowry Acid-Base Theory

62
Multiple Choice

What is the main concept of the Bronsted-Lowry acid-base theory?
Acids accept protons, while bases donate protons.
Acids accept electrons, while bases donate protons.
Acids donate protons, while bases accept protons.
Acids donate electrons, while bases accept protons.
63
An acid is a substance which is capable of accepting a pair of electrons from another substance during the process of bond formation
A base is a substance which can provide a pair of electrons to form a new bond.
Lewis Acid-Base Theory

64
Multiple Choice

What happens in a Lewis acid-base reaction?
The Lewis acid accepts a hydrogen ion from the Lewis base.
The Lewis acid accepts a pair of electrons from the Lewis base.
The Lewis acid donates a hydrogen ion to the Lewis base.
The Lewis acid donates a pair of electrons to the Lewis base.
65
Multiple Choice

Why is pH described as a logarithmic scale?
Each unit change represents a tenfold change in acidity or alkalinity
Each unit change represents a hundredfold change in acidity or alkalinity
Each unit change represents a linear change in acidity or alkalinity
Each unit change represents a twofold change in acidity or alkalinity
66
Multiple Choice
Pure water has the pH of 7. Black coffee has the pH of 5. Black coffee is how many times more acidic than pure water?
1 time more acidic
10 times more acidic
100 times more acidic
1000 times more acidic
67
Take the two pH values and subtract
The answer tells you how many zeros to add to one (1)
Problem:
7-5 = 2
add 2 zeros to 1 - it becomes 100
Black coffee is 100 times more acidic than pure water.
Solution: Pure water has the pH of 7. Black coffee has the pH of 5. Black coffee is how many times more acidic than pure water?
68
Fill in the Blanks
69
Multiple Choice

The body has a natural mechanism to control pH levels. This mechanism is known as:
Alkaline control
Acidic equilibrium
pH regulation
Acid-base balance
70
Multiple Choice
Which of the following is NOT an organ involved in acid-base balance regulation?
Lungs
Kidneys
Liver
Pancreas
71
Multiple Choice
What is the normal pH range for arterial blood in the human body?
6.5-7.0
7.35-7.45
7.55-7.70
8.0-8.5
72
Multiple Choice

What happens to the pH level of blood when carbon dioxide levels increase?
It increases (becomes more alkaline)
It remains constant
It decreases (becomes more acidic)
It has no effect on pH
73
Multiple Choice

Which of the following is a buffer system that helps regulate acid-base balance in the body?
Bicarbonate buffer system
Sulfur buffer system
Nitrogen buffer system
Phosphate buffer system
74
Multiple Choice
A _________ is a region of material that is chemically uniform, physically distinct, and (often) mechanically separable.
phase
acid
base
bond
75
Fill in the Blanks
76
A phase is a set of states of a chemical system that have similar bulk structural properties, over a range of conditions, such as pressure or temperature.
The main states of matter are solids, liquids, gases and plasmas.
Phases of Matter

77
Multiple Choice

Which phase of matter has an indefinite shape and definite volume?
solid
plasma
gas
liquid
78
Multiple Choice

Which phase of matter has a definite shape and volume?
solid
plasma
gas
liquid
79
Multiple Choice

What are the properties of a liquid?
It has no definite volume but definite shape.
It has definite volume and definite shape.
It has no definite volume and no definite shape.
It has definite volume but no definite shape.
80
Multiple Choice

What is the plasma state of matter?
The gaseous state in which matter expands to fill the entire volume of its container.
The solid state in which matter maintains a fixed shape and volume.
The fourth state of matter in which atoms or molecules are highly ionized and form a superheated gas-like state.
The liquid state in which matter takes the shape of its container.
81
Multiple Choice

What is a redox reaction?
A reaction that only involves reduction
A reaction that involves synthesis and decomposition
A chemical reaction that involves both reduction and oxidation
A reaction that only involves oxidation
82
Multiple Choice
Which process occurs during reduction in a redox reaction?
Electrons are gained
Protons are gained
Electrons are lost
Protons are lost
83
An oxidizer will remove electrons from another substance
A reducing agent will gain the electrons lost from the oxidizer
Mnemonic: LEO says GER - Loss of electrons: oxidation; Gain of electrons: reduction
Redox is a concept related to the ability of atoms of various substances to lose or gain electrons

84
Multiple Choice

What is oxidation in a redox reaction?
The process of combining two or more substances
The gain of electrons by a substance
The loss of electrons by a substance
The production of energy in a chemical reaction
85
Multiple Choice

What happens to the atoms during a chemical reaction?
Bonds between atoms break and form, resulting in different substances with different properties
Atoms are destroyed
Atoms are created
Atoms remain unchanged
86
Multiple Choice
What is a chemical equation?
A representation of a physical change in matter.
The process of converting chemical substances into simpler substances.
The process of converting physical substances into chemical substances.
A representation of a chemical reaction using chemical formulas and symbols.
87
Multiple Choice

In a chemical equation, what do the coefficients represent?
In a chemical equation, what do the coefficients represent?
The physical state of each substance.
The temperature at which the reaction occurs.
The number of atoms or molecules of each substance involved in the reaction.
88
Multiple Choice
Which of the following is an example of a chemical equation?
2NaCl
H2O
C6H12O6
2H2 + O2 → 2H2O
89
Multiple Choice
What is the difference between a chemical equation and a chemical formula?
A chemical equation includes numbers, while a chemical formula does not.
A chemical equation represents a chemical reaction, while a chemical formula represents a single compound or element.
A chemical equation shows the structure of a substance, while a chemical formula does not.
A chemical equation involves energy changes, while a chemical formula does not.
90
Multiple Choice

What does it mean to balance a chemical equation?
Balancing equations involves changing the chemical formula of the reactants.
Balancing a chemical equation involves adjusting the coefficients of the reactants and products to have an equal number of atoms on both sides.
Balancing equations refers to equalizing the concentration of the reactants and products.
Balancing equations means adding more reactants to the left side of the equation.
91
Multiple Choice
Which of the following best describes a chemical reaction mechanism?
The process of colliding molecules resulting in the formation of new substances.
The overall description of a chemical reaction, including the reactants and products.
The total mass of reactants being equal to the total mass of products.
The series of steps that shows how reactants are transformed into products.
92
Multiple Choice

Which statement best describes equilibrium in a chemical reaction?
The concentrations of reactants and products remain constant over time.
The concentrations of reactants and products continuously change over time.
The reaction does not reach completion, and reactants and products still exist.
All reactants are consumed, and no products are formed.
93
Multiple Choice

When does dynamic equilibrium occur in a chemical reaction?
When the rate of the forward reaction is faster than the rate of the reverse reaction.
When the rate of the forward reaction is equal to the rate of the reverse reaction.
When the rate of the reverse reaction is faster than the rate of the forward reaction.
When the concentrations of reactants and products are equal.
94
Multiple Choice
Which of the following best describes a catalyst?
A substance that speeds up a chemical reaction without being consumed or permanently changed
A substance that slows down a chemical reaction
A substance that gets consumed in a chemical reaction
A substance that permanently changes during a chemical reaction
95
Multiple Choice

What are catalysts made of?
Protein
Carbohydrates
Lipids
Enzymes
96
Multiple Choice
Which of the following is NOT a characteristic of catalysts?
They can be affected by temperature and pH
They can be reused multiple times
They are consumed during a chemical reaction
They are specific to certain reactions
97
Multiple Choice

What is the periodic table of elements?
It is a list of elements in alphabetical order.
It is a tabular arrangement of chemical elements, organized based on their atomic number, electron configuration, and recurring chemical properties.
It is a chart that shows the elements according to their boiling points.
It is a list of elements sorted by their mass.
98
The elements in the blocks are based on which sub-shell the last electron resides.
It represents the valence shell electron's location in its atomic orbital.
For example, if the last electron goes in the s orbital, then the element is an s block element
The periodic table can be broken down into 4 rectangular blocks

99
Multiple Choice

What do the s, p, d, and f blocks represent on the periodic table?
Different types of elements
Different types of chemical reactions
Different types of atomic masses
Different types of atomic orbitals
100
Multiple Choice

What is the atomic number?
The number of neutrons in an atom
The total number of subatomic particles in an atom
The number of electrons in an atom
The number of protons in an atom
101

The chemical elements on the periodic table are arranged in order of increasing atomic number, which refers to the number of protons of an element.
Atomic Number
102

The periodic table contains only chemical elements
Mixtures, compounds or small atomic particles of elements are not included.
103
Multiple Choice

What is a period on the periodic table?
A period is a group of elements with similar properties.
A period is a horizontal row on the periodic table.
A period is a subatomic particle with a negative charge.
A period is a vertical column on the periodic table.
104
Multiple Choice

What is the purpose of a period on the periodic table?
A period helps organize elements based on their electron configuration and properties.
A period determines the number of protons in an atom.
A period indicates the atomic mass of an element.
A period defines the chemical reactivity of an element.
105
The rows of the periodic table are called periods.
Elements on a row have the same number of electron shells or atomic orbitals
For example, elements on the first row have just one atomic orbital, elements of the second row have 2.
Periods

106
Multiple Choice

In chemistry, what is a group on the periodic table?
A region on the periodic table where metals are located
A type of chemical bond formed between atoms
A horizontal row of elements arranged by increasing atomic number
A vertical column of elements with similar properties
107
Multiple Choice

Elements within a group on the periodic table have similar properties because of their:
Similar atomic size
Similar number of neutrons
Similar number of protons
Same number of valence electrons
108
Multiple Choice
Which of the following statements is true about energy in a chemical reaction?
All of the energy is converted into heat.
No energy is lost or gained.
Some energy is lost as heat.
Energy is created during a chemical reaction.
109
Multiple Choice

Which type of chemical reaction releases energy?
Both exergonic and endergonic
Exergonic
Endergonic
Neither exergonic nor endergonic
110
Multiple Choice

Which type of chemical reaction absorbs energy?
Both exergonic and endergonic
Exergonic
Endergonic
Neither exergonic nor endergonic
111
A reaction is exergonic if the final state is lower on the energy scale than the initial state
A reaction in endergonic if the situation is reversed. The final state is higher on the energy scale than the initial state.
Exergonic vs Endergonic

112
Multiple Choice

Which of the following best describes an endothermic reaction?
A reaction that releases energy in the form of light
A reaction that releases heat to the surroundings
A reaction that absorbs heat from the surroundings
A reaction that occurs spontaneously without any input of energy
113
Multiple Choice

Which of the following best describes an exothermic reaction?
A reaction that absorbs heat from the surroundings
A reaction that releases heat to the surroundings
A reaction that occurs spontaneously without any input of energy
A reaction that requires the presence of oxygen to occur
114
Multiple Choice

What is activation energy?
The energy required to start a chemical reaction
The energy needed to break chemical bonds
The energy stored in chemical bonds
The energy released during a chemical reaction
115
Multiple Choice

What is the meaning of boiling point?
The temperature at which a substance changes from a liquid to a gas
The temperature at which a substance changes from a gas to a liquid
The temperature at which a substance changes from a solid to a gas
The temperature at which a substance changes from a solid to a liquid
116
Multiple Choice

What does condensation mean?
The process by which a solid changes into a liquid
The process by which a solid changes into a gas
The process by which a liquid changes into a gas
The process by which a gas or vapor changes into a liquid
117
Multiple Choice

What does 'critical point' mean?
The temperature at which liquid and gas phases have the same density
A point in a system where the temperature is extremely high
A point in a system where multiple substances combine
A point in a system where there is a sudden increase in pressure
118
Multiple Choice

What is the triple point in chemistry?
The point at which a substance undergoes a phase change from liquid to gas.
The point at which a chemical reaction reaches its maximum rate.
The temperature and pressure at which a substance can exist as a solid, liquid, and gas simultaneously.
The point at which a substance undergoes a phase change from solid to liquid.
119
Multiple Choice

What is the definition of density?
The ability of an object to float in water.
The total energy of an object.
The amount of mass per unit volume of a substance.
The force of gravity acting on an object.
120
Multiple Choice

What are extensive properties?
Extensive properties are characteristics that remain constant regardless of the object or system's size.
Extensive properties are only applicable to living organisms.
Extensive properties refer to the internal composition of an object or system.
Extensive properties depend on the amount or size of the object or system.
121
Multiple Choice

What is freezing in chemistry?
The process in which a substance changes from a liquid to a gaseous state due to an increase in temperature.
The process in which a substance changes from a gaseous to a liquid state due to a decrease in temperature.
The process in which a substance changes from a liquid to a solid state due to a decrease in temperature.
The process in which a substance changes from a solid to a liquid state due to an increase in temperature.
122
Multiple Choice

What is sublimation in chemistry?
The process in which a substance transitions directly from a solid to a gas without passing through the liquid state.
The process of converting a gas into a liquid.
The process of converting a solid into a liquid.
The process of converting a liquid into a solid.
123
Multiple Choice

What are intensive properties?
Properties that are independent of the quantity of the substance
Properties that depend on the quantity of the substance
Properties that are only applicable to liquids
Properties that change with temperature
124
Multiple Choice

What does malleability mean?
The ability of a substance to conduct electricity
The ability of a substance to dissolve in water
The ability of a substance to change color under certain conditions
The ability of a substance to be deformed into different shapes without breaking
125
Multiple Choice

Which process involves the passive movement of molecules from an area of high concentration to an area of low concentration?
Facilitated Diffusion
Active Transport
Osmosis
Diffusion
126
Multiple Choice

Which process involves the movement of water across a semipermeable membrane from an area of low solute concentration to an area of high solute concentration?
Osmosis
Endocytosis
Diffusion
Active Transport
127
Multiple Choice

Which process involves the combination of substances to form a single product?
Synthesis
Double displacement
Single displacement
Combustion
128

129
Multiple Choice

Which type of chemical reaction involves the breakdown of a compound into simpler substances?
Decomposition
Single displacement
Synthesis
Double displacement
130
Multiple Choice

Which type of chemical reaction involves the combination of a substance with oxygen?
Synthesis
Double displacement
Single displacement
Combustion
131
Multiple Choice

What is the main difference between single displacement and double displacement reactions?
Single displacement reactions are spontaneous reactions, while double displacement reactions are non-spontaneous reactions.
Single displacement reactions involve the exchange of atoms or ions between two compounds, while double displacement reactions only occur in the presence of a catalyst.
Single displacement reactions involve the transfer of energy through the formation of a precipitate, while double displacement reactions involve the release of energy through a combustion reaction.
In single displacement reactions, one element or ion displaces another element or ion in a compound. In double displacement reactions, atoms or ions from two different compounds switch places.
132
Multiple Choice

What is deposition in chemistry?
The process in which a gas transitions directly into a solid without passing through the liquid state.
The process of converting a solid into a liquid.
The process of converting a liquid into a gas.
The process of converting a liquid into a solid.
133
Multiple Choice

Which of the following is a source of UV radiation?
Microwave ovens
Incandescent light bulbs
The sun
Television screens
134
Multiple Choice

How does UV radiation affect the skin?
It makes the skin produce more melanin
It has no effect on the skin
It can make the skin more oily
It can cause sunburn and increase the risk of skin cancer
135
Multiple Choice
How can you protect your skin from UV radiation?
By wearing sunglasses
By wearing sunscreen and protective clothing, and by seeking shade
By staying indoors all the time
By using tanning beds regularly
TEAS Test Study Guide - Chemistry
Show answer
Auto Play
Slide 1 / 135
SLIDE
Similar Resources on Wayground

134 questions
Realidades 3 capítulo 4 Relaciones personales
Presentation
•
12th Grade

136 questions
Common Infection relevant to DentalOffice
Presentation
•
University

137 questions
Livestock Judging
Presentation
•
12th Grade

122 questions
CSE 309- HTML summer2024
Presentation
•
University

128 questions
review 1002 50 questions
Presentation
•
University

126 questions
DC US History
Presentation
•
University

128 questions
sôn lình 12 v2
Presentation
•
KG

125 questions
HS II Honors_ Unit 4.01 PowerPoint
Presentation
•
KG - University
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade