

7.PS1.1 Practice for CFA
Presentation
•
Science
•
6th - 8th Grade
•
Medium
+4
Standards-aligned
Stephanie Barahona
Used 3+ times
FREE Resource
1 Slide • 32 Questions
1
Multiple Choice

Which choice has the subatomic particles in the correct order?
A is Neutron
B is Electron
C is Proton
A is Proton
B is Electron
C is Neutron
A is Proton
B is Neutron
C is Electron
A is Electron
B is Neutron
C is Proton
2
Multiple Choice
Which particles are so small that they are almost non-existent?
electrons
negatrons
neutrons
protons
3
Multiple Choice
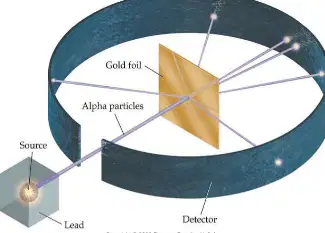
Rutherford's Gold foil experiment led him to discover ___________ .
plum pudding
the nucleus
the electron cloud
the atomic cloud
4
Multiple Choice
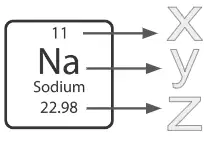
Label X is pointing to...
atomic mass
atomic number
atomic weight
atomic symbol
5
Multiple Choice

Thomson's Plum Pudding Model was best represented as _________.
The plum pieces represent positive protons. The cake-like (pudding) represents a negative nucleus
The plum pieces represent neutral neutrons. The cake-like (pudding) represents a negative electron
The plum pieces represent negative protons. The cake-like (pudding) represents a positive electron
The plum pieces represent negatively charged electrons. The cake-like (pudding) represents a positive charge.
6
Multiple Choice
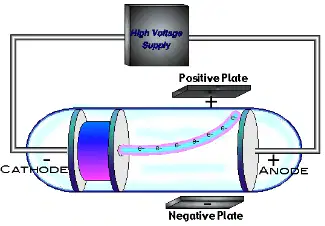
The cathode ray experiment by J. J. Thompson led him to discover ________.
plum pudding
the nucleus
the electron
the atomic cloud
7
Multiple Choice

Label Z is pointing to...
atomic mass
atomic number
number of neutrons
atomic symbol
8
Multiple Choice

Which element is represented by this atomic model?
9
Multiple Choice

How many PROTONS and NEUTRONS are in an atom of the element?
17 protons and 17 neutrons
17 protons and 18 neutrons
18 protons and 17 neutrons
18 protons and 18 neutrons
10
Multiple Choice
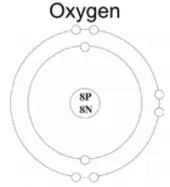
This is a diagram of an oxygen atom.
What is the mass number of oxygen?
0
8
16
24
11
Multiple Choice
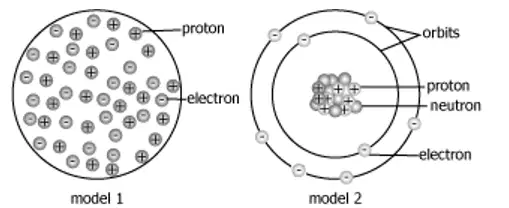
The image shows two different atomic models made by two students.
What can be concluded about the models?
Both models are accurate because they show protons and electrons inside the nucleus.
Model 1 is accurate because the numbers of protons and electrons inside the atom are the same.
Both models are accurate because they contain positively charged protons and negatively charged electrons.
Model 2 is accurate because it shows protons and neutrons at the center of the atom and electrons in orbits around the center.
12
Multiple Choice
Which charge is POSITIVE?
Proton
Neutron
Electron
Cell
13
Multiple Choice
Which charge is NEGATIVE?
Proton
Neutron
Electron
Cell
14
Multiple Choice
Which particle is NEUTRAL - NO CHARGE?
Proton
Neutron
Electron
Cell
Nucleus
15
Multiple Choice
All of the COLUMNS on the Periodic Table are called?
Periods
Rows
Groups
Lines
16
Multiple Choice
All of the ROWS on the Periodic Table are called what?
Periods
Groups
Columns
Lines
17
Multiple Choice
Which type of particle would you find floating around the nucleus, outside of the atom with its negative charges?
Proton
Neutron
Electron
Nucleus
18
Multiple Choice
Which TWO particles are found housed in the nucleus of the atom?
Protons and Electrons
Protons and Neutrons
Electrons and Neutrons
Protons, Neutrons, and Electrons
19
Match
Match the Following
Element
Atom
Nucleus
Proton
Neutron
A substance that cannot be broken down into simpler substances by chemical means.
The smallest unit of an element that keeps the element’s chemical properties.
The dense center of an atom containing protons and neutrons.
Positively charged particles found in the nucleus of an atom.
Particles with a neutral charge found in the nucleus of an atom.
A substance that cannot be broken down into simpler substances by chemical means.
The smallest unit of an element that keeps the element’s chemical properties.
The dense center of an atom containing protons and neutrons.
Positively charged particles found in the nucleus of an atom.
Particles with a neutral charge found in the nucleus of an atom.
20
Multiple Choice
21
Multiple Choice
The three subatomic particles are
proton, nucleus, electron
atom, element, compound
atom, element, molecule
proton, neutron, electron
22
Multiple Choice
The nucleus is ____________ charged.
positively
negatively
neutral
23
Multiple Choice
The area around the nucleus of an atom where the atom's electrons are most likely to be found is called
electron cloud
quark
electron
nucleus
24
Multiple Choice
99.9% of the atom's mass is found where?
nucleus
protons
electron cloud
neutrons
25
Multiple Choice

What is 'E' pointing to?
electron cloud
electron
nucleus
proton
neutron
26
Multiple Choice

27
Multiple Choice
What is the overall charge of an ion with 9 protons, 10 neutrons and 10 electrons?
0
+9
+1
-1
28
Multiple Choice
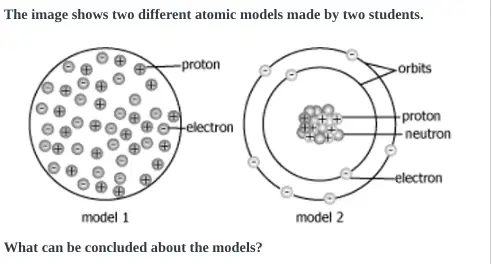
Which model is correct. Pick the reason below that states why correctly
Both models- because they show protons and electrons inside the nucleus.
Model 1-
because the numbers of protons and electrons inside the atom are the same
Both models- because they contain positively charged protons and negatively charged electrons.
Model 2-
because it shows protons and neutrons at the center of the atom and electrons in orbits around the center.
29
Multiple Choice
An atom has an atomic number of 3 and an atomic mass that rounds to 7.
How many protons and neutrons are in the atom?
3 protons and
3 neutrons
4 protons and
3 neutrons
4 protons and
4 neutrons
3 protons and
4 neutrons
30
Multiple Choice

31
Multiple Choice
32
Match
Match the charge with the correct subatomic particle.
Electron
Neutron
Proton
- 1
0
+ 1
- 1
0
+ 1
33

Which choice has the subatomic particles in the correct order?
A is Neutron
B is Electron
C is Proton
A is Proton
B is Electron
C is Neutron
A is Proton
B is Neutron
C is Electron
A is Electron
B is Neutron
C is Proton
Show answer
Auto Play
Slide 1 / 33
MULTIPLE CHOICE
Similar Resources on Wayground

25 questions
What is Technology?
Presentation
•
6th - 8th Grade

27 questions
Gravitational Forces
Presentation
•
6th - 8th Grade

28 questions
Lab Safety Lesson/Quiz
Presentation
•
6th - 8th Grade

26 questions
1.2.3 Moving Cellular Material
Presentation
•
6th - 8th Grade

26 questions
Lab Safety
Presentation
•
6th - 8th Grade

27 questions
The Geologic Time Scale
Presentation
•
6th - 8th Grade

27 questions
Refraction and Lenses
Presentation
•
6th - 8th Grade

25 questions
SOL Review: Earth, Moon and Sun System
Presentation
•
6th - 8th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

20 questions
Rocks and The Rock Cycle
Quiz
•
6th Grade

20 questions
genetics, punnett squares, heredity
Quiz
•
7th Grade

16 questions
Photosynthesis and Cellular Respiration Review
Quiz
•
7th Grade

30 questions
GMAS Physical Science Review
Quiz
•
8th Grade

20 questions
Human Body Systems
Quiz
•
6th Grade

15 questions
Punnett Squares
Quiz
•
6th Grade

20 questions
Food Webs + Energy Pyramids
Quiz
•
7th Grade