

Atomic Structure and Periodic Table
Presentation
•
Chemistry
•
9th Grade
•
Practice Problem
•
Medium
Standards-aligned
Lane Derrick
Used 41+ times
FREE Resource
10 Slides • 20 Questions
1



Periodic Table Organization
Periods = rows (across)
- # of periods = 7
- Going across a period, the number of protons goes up
Groups = columns (up and down)
- # of groups = 18
- Elements in a group have similar properties
2


Lanthanide and Actinide Series
● Located under the periodic table, but they actually
belong in periods 6 and 7
● Lanthanides - period 6
● Actinides - period 7
Lanthanides
Actinides
3

Expanded periodic table
Why are lanthanides and actinides on the bottom?
So it's easier to fit the periodic table on piece of paper
LANTHANIDES
ACTINIDES
4
Multiple Choice

What element is in period 2 and group 13?
lithium, Li
beryllium, Be
boron, B
helium, He
5
Multiple Choice

What element is in period 5 and group 11?
Copper, Cu
silver, Ag
Iron, Fe
Vanadium, V
6
Multiple Choice

What element is in group 17 and period 3?
7


1. Metals
2. Metalloids
3. Nonmetals
Elements are organized into 3 classifications.
8



Label your periodic table (use arrows for metals and nonmetals, lightly shade metalloids)
1. Left side of stairs:
metals
2. Right side of stairs:
nonmetals
3. Touching the stair:
metalloids (B, Si,
Ge, As, Sb, Te, Po?, At?)
Not Aluminum!
METALS
Nonmetals
Metalloids
9
Multiple Choice

Most of the elements on the periodic table are
metalloids
metals
nonmetals
gases
10

11

12
Multiple Choice

Which of the following will have a larger radius than Zinc?
Gallium
Aluminum
Magnesium
Strontium
13

14
Fill in the Blanks
Type answer...
15
Multiple Choice
Which of these elements has a larger atomic radius?
Lithium Li
Fluorine F
Carbon C
16
Valence Electrons
These are the number of electrons found on the outermost energy level
If you are still confused about valence electrons, watch the 4-minute video which will explain more about valence electrons. You will need to copy and paste the link into another browser tab.
https://www.youtube.com/watch?v=0hsh0VoFsts
17
To determine the # of valence electrons we look at the group an element is in.
Groups are the vertical columns within a periodic table. All elements in the same group will have the same number of valence electrons.
18
Multiple Choice
How many valence electrons does C have?
2
4
12
14
19
Multiple Choice
How many valence electrons does Ca have?
2
4
12
14
20
Multiple Choice
How many valence electrons does Cs have?
2
55
12
1
21
Multiple Choice
Which element has the same number of valence electrons as Cl?
I
Ar
Al
K
22
Multiple Choice
Which element has the same number of valence electrons as Kr?
I
Ar
Al
K
23
Multiple Choice
24
Multiple Choice
25
Multiple Choice
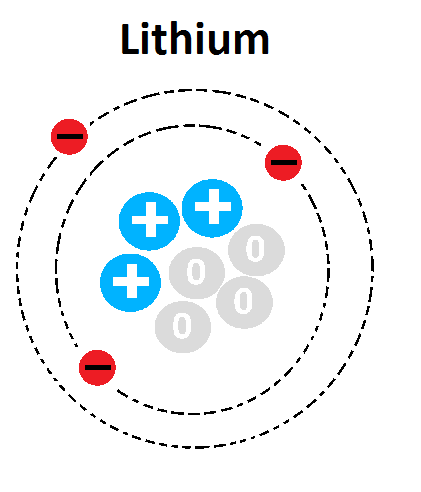
26
Multiple Choice
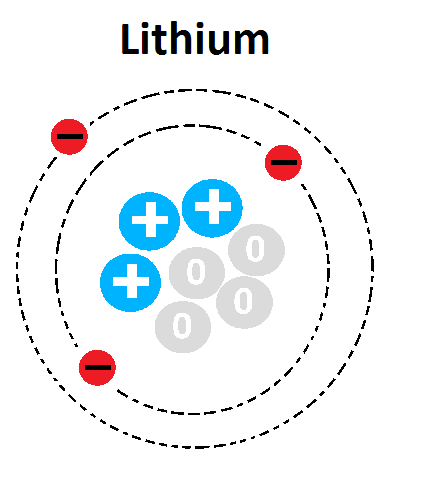
What is the mass number of this atom?
1
3
4
7
27
Multiple Choice
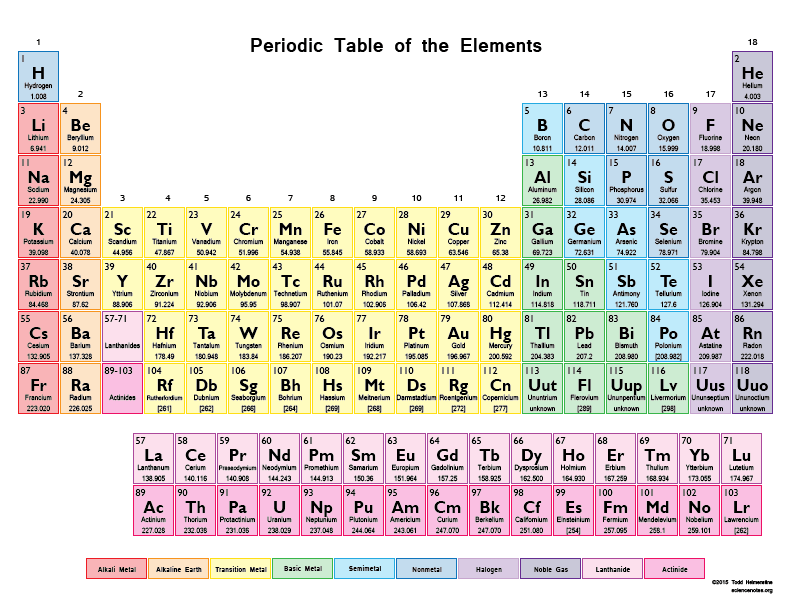
What is the atomic number of Barium, Ba? (enlarge the periodic table)
20
38
56
88
28
Multiple Choice

What is the atomic number of Nickel? (enlarge the periodic table)
110
28
46
78
29
Multiple Choice
30
Multiple Choice
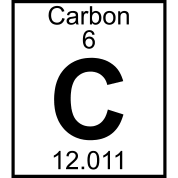



Periodic Table Organization
Periods = rows (across)
- # of periods = 7
- Going across a period, the number of protons goes up
Groups = columns (up and down)
- # of groups = 18
- Elements in a group have similar properties
Show answer
Auto Play
Slide 1 / 30
SLIDE
Similar Resources on Wayground

26 questions
Chemical Bonds
Presentation
•
10th Grade

21 questions
IONIC BONDING_MELSCI
Presentation
•
9th Grade

23 questions
Naming Covalent Compounds
Presentation
•
10th Grade

23 questions
Algebra 1: Algebraic Properties
Presentation
•
9th Grade

24 questions
Age of Exploration
Presentation
•
9th Grade

21 questions
Climate Change
Presentation
•
9th Grade

22 questions
Oceania
Presentation
•
9th Grade

22 questions
Introduction to Relations and Functions
Presentation
•
9th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade