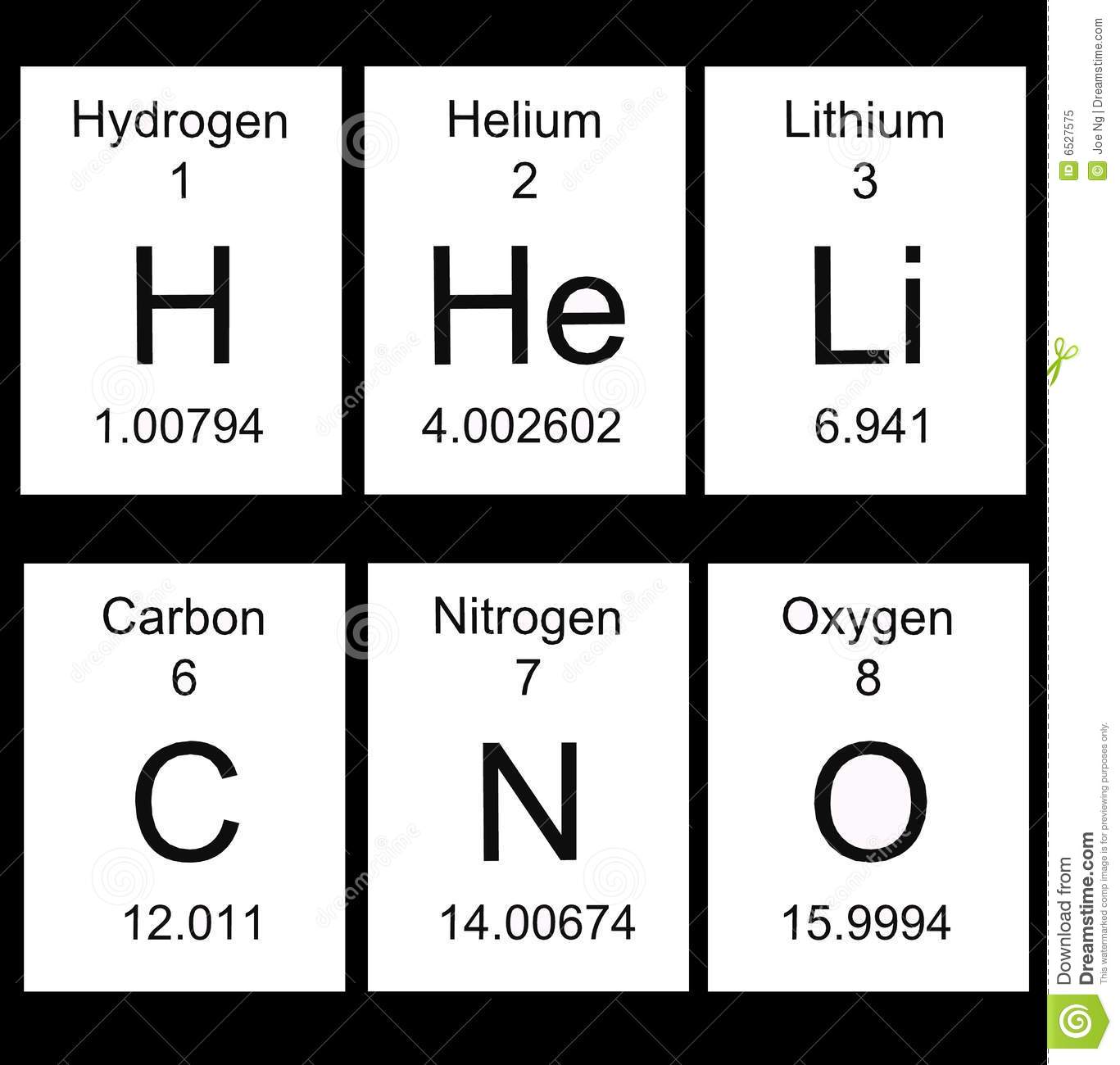
11B/09/27
Presentation
•
Science
•
9th - 12th Grade
•
Medium
+6
Standards-aligned
Osman Mutlu
Used 1+ times
FREE Resource
12 Slides • 37 Questions
1
2
Multiple Choice
3
Multiple Choice
4
Multiple Choice
5
Multiple Choice
6
Multiple Choice
7
Multiple Choice
8
Multiple Choice
9
Multiple Choice
10
Multiple Choice
11
Multiple Choice
12
Multiple Choice
13
Multiple Choice
14
Multiple Choice
15
Multiple Choice
16
Multiple Choice
17
Multiple Choice
18
Multiple Choice
19
Multiple Choice
20
Multiple Choice
21
Multiple Choice
22
Multiple Choice
23
Multiple Choice
24
Multiple Choice
25
Multiple Choice
26
Multiple Choice
27
28
29
30
Multiple Choice
31
32
33
34
Multiple Choice
35
36
37
38
Multiple Choice
39
40
41
Multiple Choice
42
Multiple Choice
43
Multiple Choice
44
Multiple Choice
45
Multiple Choice
46
Multiple Choice
47
Multiple Choice
48
Multiple Select
49
Multiple Select
Show answer
Auto Play
Slide 1 / 49
SLIDE
Similar Resources on Wayground

42 questions
Energy and Life Day 1-ATP
Lesson
•
9th - 12th Grade

48 questions
Meteors, comets and debris
Lesson
•
10th - 12th Grade

47 questions
The Periodic Table
Lesson
•
10th - 12th Grade

43 questions
Introduction to the Holocaust Notes & Lesson
Lesson
•
9th - 12th Grade

45 questions
Intro to Ecology
Lesson
•
9th - 12th Grade

46 questions
Mr. Bradley' pH Acids and Bases Lesson
Lesson
•
9th - 12th Grade

44 questions
PSci 4.1
Lesson
•
9th - 12th Grade

43 questions
Poetry, Prose, and Drama
Lesson
•
9th - 12th Grade
Popular Resources on Wayground

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

10 questions
Probability Practice
Quiz
•
4th Grade

15 questions
Probability on Number LIne
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

22 questions
fractions
Quiz
•
3rd Grade

6 questions
Appropriate Chromebook Usage
Lesson
•
7th Grade

10 questions
Greek Bases tele and phon
Quiz
•
6th - 8th Grade
Discover more resources for Science

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

10 questions
Exploring Weathering, Erosion, and Deposition Processes
Interactive video
•
6th - 10th Grade

26 questions
Unit 8b Review
Quiz
•
9th Grade

14 questions
Natural Selection and Adaptation
Lesson
•
9th - 12th Grade

10 questions
Exploring Water Pollution and Conservation
Interactive video
•
6th - 10th Grade

10 questions
Exploring the Human Respiratory System
Interactive video
•
6th - 10th Grade

30 questions
Unit 2C Progress Check (Biosphere 3)
Quiz
•
9th - 12th Grade

14 questions
Genetics Practice (Mendelian & Non-mendelian)
Quiz
•
10th Grade