

T Level Radiation Recap
Presentation
•
Physics
•
11th - 12th Grade
•
Medium
T Southerden
Used 3+ times
FREE Resource
46 Slides • 32 Questions
1
Radioactive Decay

2
Unstable Nuclei
Some atoms have stable nuclei.
Some atoms have unstable nuclei.
These undergo radioactive decay to try and become more stable.
3
Multiple Choice
What is an isotope?
An atom with the same number of neutrons and electrons, but a different number of protons.
An atom with the same number of protons and electrons, but a different number of neutrons.
An atom with the same number of neutrons and protons, but a different number of electrons.
4
Unstable Nuclei
The nuclei of an atom can be unstable for three reasons.
1. the nuclei is too big
- these emit an alpha particle
2. the nuclei has an incorrect ratio of protons and neutrons
- these emit a beta particle
3. the nuclei has too much energy.
- these emit gamma rays
5
Alpha Particles
Alpha particles (α) consist of 2 neutrons and 2 protons (no electrons).
This is the same as a helium nucleus.
Alpha decay causes the mass number of the nucleus to decrease by four and the atomic number of the nucleus to decrease by two.
The overall charge of an alpha particle is +2.

6
Beta Particles
If a nuclei has too many neutrons, a neutron will turn into a proton and emit a fast-moving electron. This electron is called a beta (β) particle.
A beta particle has a relative mass of zero. Beta decay causes the atomic number of the nucleus to increase by one and the mass number remains unchanged.
The charge of a beta particle is -1.

7
Gamma rays
After emitting alpha/beta particles, nuclei still have a high amount of energy and need to 'cool down'.
So, often they will emit gamma rays (γ). These are electromagnetic (EM) waves.
Gamma ray emission causes no change in the number of particles in the nucleus meaning both the atomic number and mass number remain unchanged.

8
Multiple Choice
which type of decay is also referred to as a helium nuclei?
alpha
beta
gamma
9
Multiple Choice
Which type of decay is a type of electromagnetic wave?
alpha
beta
gamma
10
11
Chernobyl Disaster 1986
During 1986, a nuclear power plant in Ukraine experienced a critical failure in their safety measures. This resulted in a considerable amount of radioactive contamination.
This contamination included alpha, beta and gamma radiation.
Today, tourists can enter limited parts of Chernobyl.

12

13
Geiger-Muller counter
All types of radioactive decay can be detected by a Geiger-Muller tube, or G-M tube.
The radiations ionise the gas inside and the resulting charged particles move across the chamber and get counted as charges rather like an ammeter.

15
Open Ended
Tourists at Chernobyl must wear miniature versions of GM counters - why is this a good idea?
16
Ionising and Penetrating Power
Ionising Power - to ionise means to convert an uncharged atom or molecule into a charged particle by adding or removing electrons.
Penetrating Power - the power of the radiation that demonstrates how far into a material the radiation will go.
17
18
19
Multiple Choice
How ionising is alpha decay?
low
moderate
high
20
Multiple Choice
how penetrating is beta decay?
low
moderate
high
21
Multiple Choice
how ionising is gamma radiation?
low
moderate
high
22

23




Types of Radioactive Emission
24


Alpha Decay
• Radiation through the loss of 2p + 2n or
(helium)
25


Beta Decay
• Radiation where a neutron splits, giving off
an electron and becoming a proton in the
new element
26
Multiple Choice
Alpha particles (alpha decay) are comprised of
2 Protons & 2 Neutrons
4 Neutrons & 2 Electrons
an electron
an energy wave
27
Multiple Choice
Beta particles (Beta decay) are comprised of
2 Protons & 2 Neutrons
4 Neutrons & 2 Electrons
an electron
an energy wave
28


Question 3a
Balance the nuclear equation after alpha
decay
29


Question 3a
Balance the nuclear equation after alpha
decay
30



Question 3b
Balance the nuclear equation after beta decay
Remember in beta decay a neutron changes into a proton by giving off an
electron
31



Question 3b
Balance the nuclear equation after beta decay
Remember in beta decay a neutron changes into a proton by giving off an
electron
32
Multiple Choice
146C --> 0-1e + ________
33
Multiple Choice
34
Multiple Choice
If we start off with element 5024X after an gamma decay we get an element(product) that looks like...
5124X
5023X
5024X
5025X
35
Multiple Choice
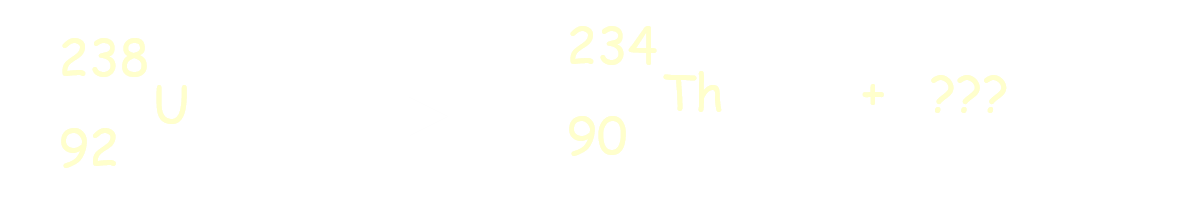
36
Multiple Choice
37
Multiple Choice
24094Pu —› ____ + 42He
23692U
24796Cm
24493Np
24295Am
38
Half Lives
What is it?
the time taken for half of the nuclei to decay
Some radioisotopes have long half lives, like U-238, and some have short ones, like I-131. Long half lives can be used to date ancient artefacts. Carbon-13 is useful in this instance. Short half-lives are useful in medicine.
39
Let's count!
If you have 100g of a radioactive isotope, how much would you have after two half-lives-
a) if the half life is 3 years?
b) if the half life is 3 hours?
40
Multiple Choice
A radioisotope has a half-life of 10 years. In the year 2000, there was 50g of this sample in a container. How much would there be in the year 2020
25g
12.5g
100g
41
Multiple Choice
The half life of plutonium is 24300 years. If nuclear bomb released 8kg of this isotope, how many years would pass before the amount is reduced to 1kg?
72900
50
8100
5
42
Exponential Decay
trendlines in science
Trendlines are used to make clear the trend in the data
You can have linear and non-linear trends
Non-Linear trends can be exponential, logarithmic, quadratic or trigonometric
Linear trends- well, they are just straight line!
43
Here is a non linear exponential decaying trend

44
Here is one that seems to be periodic

45
Radioactive Decay
Radioactive decay is an exponentially decaying function
It is a rapidly decreasing function
But a very useful function because we can determine the half life of these radioactive isotopes
46

Which experiment had a longer half life?
47
Multiple Choice

Determine the amount of the radioisotope left 4 years
approx 60g
approx 6g
approx 155g
10g
48
Multiple Choice

Determine the half life or radioisotope 'x'
approx 5 years
approx 1 year
approx 1/2 year
approximately 50 years
49
Can we use half life to predict amounts?
Suppose we start with 120 atoms of a radioactive sample, how many will remain after three half lives?
What percentage is that?
50
The half life of Carbon-14 is 5730 years. You have a sample that has only 25% of the original amount. How many years has passed?
51
Radioactive Dating

In 1896, Marie and Pierre Curie worked with Henri Becquerel to study the element Uranium. Together, they discovered the property of radioactivity, which is used in a variety of technologies and sciences.
52
What is Radioactive Dating?
Igneous Rocks naturally contain some radioactive elements
Scientists can measure the amount radioactive elements in a rock and compare it to the half-life of those elements
Ohio Grade 8 | Lesson 6.3

53
Potassium-Argon Dating
How scientists often date rocks
Used due to a very long half-life
When Potassium-40 breaks down, it forms argon-40
has a half life of 1.3 Billion Years
Ohio Grade 8 | Lesson 6.3
54
Carbon-14 Dating
Carbon-14 is a radioactive form of carbon
Has a half life of only 5,730 years
All plants and animals have carbon
After a plant or animal dies, Carbon-14 decays into nitrogen-14
Only good for fossils younger than 50,000 years old
after that the amount of carbon is too small to be measured
Ohio Grade 8 | Lesson 6.3

55
Multiple Choice
Scientists use the method of _________ to find the absolute age of a rock
radioactive dating
relative dating
Carbon-dating
lava dating
56
Multiple Choice
True or False: Carbon-14 is used for dating fossils because it has a short half life
True
False
57
58

Fission and Fusion
59
60
Nuclear Fission
Larger nuclei break down into smaller more stable nuclei.
A lot of Energy is released.
Source of all Nuclear Power on Earth.
e.g. Nuclear Bomb, Nuclear Power Plant.

61


62
Multiple Choice

63
Nuclear Fusion
Smaller nucleii combine to form larger nucleii
Lot of Energy is released.
Needs High Temperature and Pressure.
Not Possible on Earth.
Sun and Stars - Nuclear Fusion
e.g Hydrogen Bomb.

64
The Sun
a massive ball of plasma.
It is constantly burning so hot that hydrogen is fused together to form helium.
This happens at such a rate that a huge amount of energy is released.

65
Multiple Choice
When small nuclei are exposed to intense heat and pressure, they join to create a larger nucleus, releasing huge amounts of energy
Nuclear Fusion
Nuclear Fission
66
Multiple Choice
The sun (or any star)
Nuclear Fusion
Nuclear Fission
67
Multiple Choice
Fission is --
fusing together
division
68
Categorize
Atom is divided
Two atoms join
Nuclear waste is an issue
Uses uranium and other radioactive elements
1000 x stronger
Typically uses hydrogen
Naturally occuring on the sun
Produces energy in stars
Requires high temperature and pressure
Produces helium as a byproduct
Experimental energy source
Generates heat and electricity
Used in nuclear power plants
Produces radioactive isotopes
Chain reaction can occur
Requires controlled environment
69
Multiple Choice
70

71

72
Multiple Choice
___________ starts the fission of a uranium atom.
Energy
Protons
One Neutron
All of the above
73
Nuclear Weapons
Atomic Bombs use uncontrolled fission reactions
Thermonuclear (hydrogen) bombs use fission reactions to induce fusion - 100x more powerful than atomic bombs

74
Multiple Select
Which of the following correctly display nuclear fission?
75
Multiple Select
Which of the following correctly display nuclear fusion?
76
Multiple Choice
77
Multiple Choice
78

| STEM
You can open this webpage in a new tab.
Radioactive Decay

Show answer
Auto Play
Slide 1 / 78
SLIDE
Similar Resources on Wayground

71 questions
Pedigree
Presentation
•
11th - 12th Grade

70 questions
Phylum Mollusca Notes
Presentation
•
10th - 12th Grade

72 questions
Senderos 2 Lección 4 - El Cuerpo
Presentation
•
10th - 12th Grade

69 questions
Parts and Function of a Cell
Presentation
•
11th Grade

75 questions
What is Science?
Presentation
•
10th Grade - University

73 questions
Unit 3 Objectives
Presentation
•
11th - 12th Grade

71 questions
TSIA 2 Reading and Writing Overview
Presentation
•
12th Grade

74 questions
Number Systems 1
Presentation
•
12th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Physics

11 questions
Electricity Explained
Interactive video
•
9th - 12th Grade

15 questions
waves and wave properties
Quiz
•
9th - 12th Grade

18 questions
Unit 1 and 2 Concepts Review
Quiz
•
9th - 12th Grade

15 questions
Motion Graphs 101
Quiz
•
8th - 11th Grade

50 questions
AP Physics 1 Semester Exam REVIEW
Quiz
•
11th Grade

12 questions
EOY DAY 1 ELECTRICITY
Presentation
•
10th - 12th Grade

19 questions
Optics Quiz
Quiz
•
11th Grade

10 questions
EOY REVIEW 2- Electrostatics & Momentum
Presentation
•
10th - 12th Grade