

Unit 4 Test Review: Properties of matter, density, IMF
Presentation
•
Physics
•
11th Grade
•
Medium
+2
Standards-aligned

Leslie Dunham
Used 7+ times
FREE Resource
9 Slides • 31 Questions
1

Unit 4 Review: Properties of matter, density, IMF
2

IntrAmolecularforces
Type of Bond (weakest to strongest)
Nonpolar covalent
Polar covalent
Ionic
3

Polar
Unequal sharing of electrons
Electronegativity difference >0.4 and <1.7
Nonpolar
Equal sharing of electrons
Electronegativity difference 0.0 - 0.4
Polar vs Nonpolar
4
Multiple Choice
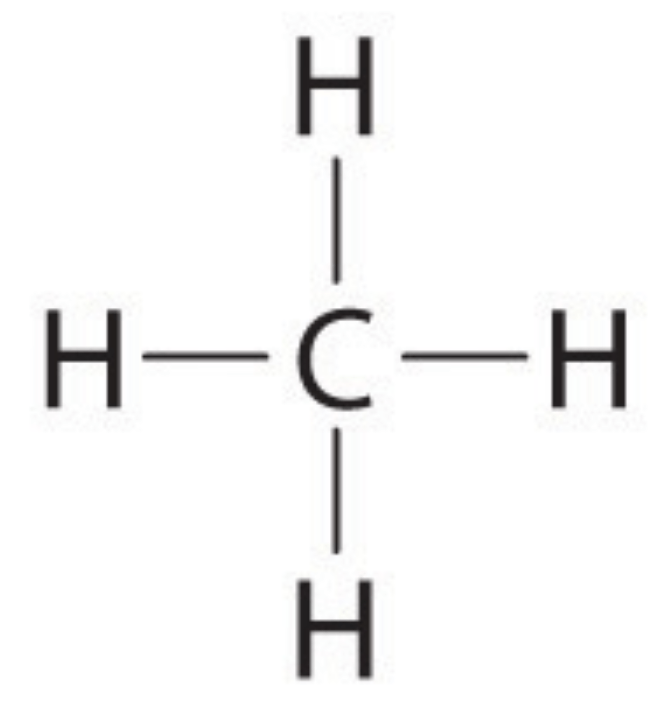
Classify the following molecule.
polar
nonpolar
5
Multiple Choice
What kind of solute will dissolve in a nonpolar solution? (Select all that apply)
polar covalent solvent
nonpolar covalent solvent
ionic solvent
metallic solvent
6
Multiple Select
What kind of solute will dissolve in a polar solution? (Select all that apply)
polar covalent solvent
nonpolar covalent solvent
ionic solvent
metallic solvent
7
Multiple Choice
... bonds happens when electrons are shared EVENLY between the two bonded atoms.
Covalent polar
Covalent nonpolar
Ionic
Metallic
8

London Dispersion
Dipole-dipole
Hydrogen Bond
Ion Dipole
IntErmolecular forces
9
IMF and physical properties
STRONGER IMFs lead to:
Higher viscosity (thickness of a liquid, molecules sticking to each other)
Higher boiling point (change from liquid to gas)
Higher melting point (change from solid to liquid)
10
Multiple Choice
Substance A is a liquid, Subtance B is a solid. Which has higher intermolecular forces?
Substance A
Substance B
11
Multiple Choice
If a substance has stronger intermolecular forces, what would be true of its viscosity?
Viscosity would be lower
Viscosity would be higher
Viscosity is not affect by intermolecular forces
12
Multiple Choice
The stronger the intermolecular forces of a substance the _____________ the boiling point
higher
lower
13
Multiple Select
Select all of the Intermolecular Forces that water has.
London dispersion forces
Dipole-dipole
Hydrogen bond
Ion dipole
14
Multiple Choice
Which substance has the weakest intermolecular forces?
Substance A, boiling point of 75 °C
Substance B, boiling point of 105 °C
Substance C, boiling point of 25 °C
Substance d, boiling point of 45 °C
15
Multiple Choice
Which of the following will NOT have hydrogen bonding?
16
Multiple Choice
Which of the following will have the lowest melting point?
17
Multiple Choice
Hydrogen bonding is a special type of what force?
London dispersion forces
ion-dipole forces
dipole-dipole forces
covalent force
18
Multiple Choice
Which of these is not an intermolecular force?
covalent bonding
London dispersion forces
hydrogen bonding
dipole-dipole forces
19
Multiple Choice
20
Multiple Choice
Type of intermolecular force present in I2, Br2, and Cl2.
dipole dipole
H-bond
London dispersion
metallic
21
Multiple Choice
22

Physical - can be observed without changing the substance
Chemical - describe the ability to change into different substances
Physical and Chemical Properties
23

Intensive - doesn't change based on how much of a substance you have
Extensive - does change based on how much you have
INtensive vs EXtensive physical properties
24
Multiple Choice
Physical properties....
change the matter when studied
can be observed and measured without changing the matter being studied
include the ability to burn and rust
are useless unless studied in a lab
25
Multiple Choice
______________________ can only be recognized when substances react or do not react chemically with one another.
Physical properties
Chemical properties
Flammable properties
Colorful properties
26
Multiple Choice
27
Multiple Choice
All of the following are examples of physical properties EXCEPT
Density
Heat conductivity
pH (acidic or basic)
Boiling point
28
Multiple Choice
This property changes as the amount of matter changes.
Intensive
Extensive
Chemical
Dense
29
Multiple Choice
Which of the following is an extensive property?
Color
Mass
Odor
Luster
30
Multiple Choice
8) Intensive or Extensive Property: Volume
Intensive
Extensive
31
Multiple Choice
9) Intensive or Extensive Property: Density
Intensive
Extensive
32
Multiple Choice
9) Intensive or Extensive Property: Malleability
Intensive
Extensive
33
Multiple Choice
What kind of physical property is this? Length
intensive
extensive
34

Density in an intensive physical property of matter
Density
35

Water displacement for volume of a solid
Mass of a liquid
Measuring lxwxh of a solid
Ways to find density
36
Multiple Choice
A bar of copper has a mass of 216g and a volume of 24 cm3. What is the density of copper?
9g/cm3
5184g/cm3
.11g/cm3
322mL
37
Multiple Choice
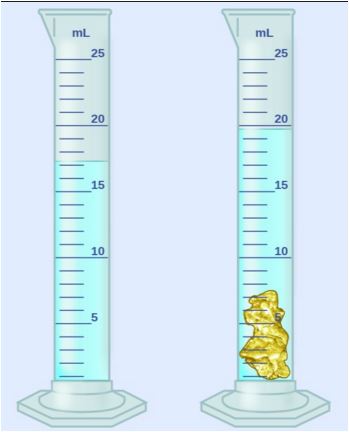
38
Multiple Choice

39
Multiple Choice

Calculate the density of the rectangular prism
2 g/cm3
4 g/cm3
6 g/cm3
8 g/cm3
40
Multiple Choice

Use the density chart to identify the substance. If the substance has a volume of 5 cm3 and a mass of 52.5.
lead
aluminum
silver
water

Unit 4 Review: Properties of matter, density, IMF
Show answer
Auto Play
Slide 1 / 40
SLIDE
Similar Resources on Wayground

37 questions
B.4.2 Lesson 4 Thermodynamic Processes
Presentation
•
11th Grade

31 questions
Conservation of Mechanical Energy
Presentation
•
11th Grade

34 questions
Stoichiometry Review
Presentation
•
10th - 12th Grade

36 questions
The Brain
Presentation
•
10th Grade

35 questions
Physical Science 15.1
Presentation
•
10th Grade

33 questions
Unit 1-1D Motion Review
Presentation
•
KG - 12th Grade

35 questions
Lesson: Electron Configuration
Presentation
•
11th Grade

34 questions
26. Work - PE
Presentation
•
11th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Physics

50 questions
Honors Physics - 2nd Semester Finals Review - Part 1
Quiz
•
11th Grade

30 questions
Kinetic Energy Question
Quiz
•
11th Grade

50 questions
Honors Physics - 2nd Semester Finals Review - Part 2
Quiz
•
11th Grade

80 questions
2nd Semester Finals Review
Quiz
•
11th Grade

45 questions
Spring 2026 Final Exam
Quiz
•
9th - 12th Grade

17 questions
Newton's Laws
Quiz
•
KG - University