

Chemistry Exam Preparation
Presentation
•
Chemistry
•
8th Grade
•
Easy
+3
Standards-aligned
Adonia Halel
Used 12+ times
FREE Resource
28 Slides • 64 Questions
1

2

3

4

5

6

7
Multiple Choice
What is weather?
The amount of moisture in the air
The number of clouds in the sky
The speed of the wind
The state of the atmosphere at any particular time.
8

9
Multiple Choice
climate
day-to-day changes in atmospheric conditions, including temperature, air pressure, humidity, wind, cloud coverage, or precipitation
moisture in the air
measures air pressure
long-term average of weather in an area
10

11

12
Multiple Choice

Which climate zone gets the least amount of precipitation?
Tropical Rain Forest
Tundra
Highland
Desert
13
Multiple Choice

Mild winter and hot, dry summer
Mediterranean
Arid
Tropical
Tundra
14
Multiple Choice

Found in the extreme north of North America, this climate zone has permafrost (permanently frozen ground).
Tropical
Arid
Desert
Tundra
15
Multiple Choice
Polar
Cold winter and mild summer
Very cold and dry all year
Very cold all year
Hot and wet all year
16
Multiple Choice
Temperate
Cold winter and mild summer
Very cold and dry all year
Very cold all year
Hot and wet all year
17
Multiple Choice
Tropical
Cold winter and mild summer
Very cold and dry all year
Very cold all year
Hot and wet all year
18

19
Multiple Choice
What is a glacial period?
A glacial period is a period of time when the climate was warmer and glaciers melted rapidly.
A glacial period is a period of time when the climate was stable and glaciers did not change in size.
A glacial period is a period of time when the Earth's surface was covered in volcanic ash.
A glacial period is a period of time in the Earth's history when the climate was colder and glaciers covered a large part of the Earth's surface.
20

21
Multiple Choice
What is an interglacial period?
An interglacial period is a time when the Earth is closest to the sun
An interglacial period is a period of extreme cold and ice formation
An interglacial period is a short-term period of drought and desertification
An interglacial period is a geological interval of warmer global average temperature lasting thousands of years that separates consecutive glacial periods within an ice age.
22
Multiple Choice
What is the difference between a glacial period and an interglacial period?
The main difference is that a glacial period is a long period of cold climate and extensive ice sheets, while an interglacial period is a shorter period of warmer climate between glacial periods.
An interglacial period is a long period of cold climate and extensive ice sheets
A glacial period is warmer than an interglacial period
There is no difference between a glacial period and an interglacial period
23
Multiple Select
Which of the evidences supports that the Earth was colder in the past? (multiple answers)
Rock layers
The glaciers carry rocks with them.
The rocks were left behind when glaciers melted.
None of the above
24
Multiple Choice
How do scientists study past climates and ice ages?
By studying the migration patterns of birds
By analyzing the growth of coral reefs
By observing the behavior of polar bears
By analyzing ice cores, tree rings, sediment layers, and other geological records.
25

26
Multiple Choice
In New Zealand, a scientist extracted a core of soil from a
peat bog
rock
plants
glaciers
27
Multiple Select

Which of the following statements are true about peat bog? (multiple answers)
The deeper the peat, the older it is.
The layers of the peat bog represent the different periods of histroy.
The core must be removed carefully to keep the correct order.
Undetermine
28

29
Multiple Choice
Gold jewelry is marked to tell you how pure the gold is. Which is the marking that has the most gold?
30
Multiple Choice
To calculate the purity of the gold, you can divide the carat marking by the 24 carat (pure gold) and times it by 100. What is the purity of 12K gold?
31

32

33
Multiple Choice
Which two particles would be attracted to each other?
protons and neutrons
electrons and protons
electrons and neutrons
All particles are attracted to each other.
34
Multiple Choice
Which of the following is NOT a subatomic particle?
proton
neutron
electron
megatron
35
Multiple Choice
Which subatomic particle has a positive charge?
proton
neutron
electron
36
Multiple Choice
Which subatomic particle has a neutral (no) charge?
electron
neutron
electron
37
Multiple Choice
The center of an atom is called the __________ ?
neutron
nucleus
number
38
Multiple Choice

How many protons are in one atom of Carbon?
6
12
18
39
Multiple Choice

How many neutrons are in one atom of Carbon?
6
12
18
40
Multiple Choice

How many electrons are in one atom of Carbon?
6
12
18
41
Multiple Choice
An element has electronic structure of 2, 8, 4. Which group does this element belongs to?
Group 4
Group 8
42
Multiple Choice
Chlorine has 17 electrons. Which group does chlorine belongs to?
Group 7
Group 8
43
Multiple Choice

State the melting and boiling points trend of group 1.
44
Multiple Choice

State the melting and boiling point trends of group 7.
45
Multiple Choice

Predict the electronic structure of Bromine.
2, 8, 6,
2, 8, 8, 7
46
Multiple Choice

State the melting and boiling points trends in group 8.
47
Multiple Choice
Which group is known as non reactive group?
48

49
Multiple Choice
What makes Sodium atom become Sodium ion? (Na = 11 electrons)
50

51
Multiple Choice
52
Multiple Choice
53
Multiple Choice
54
Multiple Choice
55
Multiple Choice
56
Multiple Choice
Atoms that loses electrons become...
57

58
Multiple Choice
59
Multiple Choice
60
Multiple Choice
How do covalent bonds form?
Donating & receiving electrons between atoms.
Scientists are still not sure how they form.
Sharing electron between atoms.
61
Multiple Choice
In covalent bonding, electrons are
shared
transferred
wiped out
62
Multiple Choice
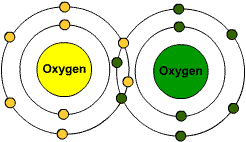
63
Multiple Choice

64
Multiple Choice
65
Multiple Choice
State one difference between ionic bond and covalent bond.
Ionic bonds are weaker than covalent bonds.
66

67
Multiple Choice
Which of the following diagram represents Rutherford's Model of Atom?
68
Multiple Choice
Which of the following diagram represents Bohr's Model of Atom?
69

70

71

72

73

74
Multiple Choice

Salts that are made with hydrochloric acid are called ______________.
chlorides
sulfates
nitrates
carbonates
75
Multiple Choice

What are the products if magnesium reacts with hydrochloric acid?
Magnesium chloride + Hydrogen
Magnesium sulfate + Hydrogen
Magnesium oxide + Hydrogen
Magnesium sulfide + Hydrogen
76
Multiple Choice
Complete the following word equation:
Metalcarbonate + acid -->
Salt + Hydrogen
Salt + Water + Carbon Dioxide
Salt + Water
77
Multiple Choice
Complete the following word equation:
Metal + Acid -->
Salt + water
Salt + hydrogen
Salt + Water + Carbon dioxide
78
Multiple Choice
What is the test for Carbon Dioxide?
Add Litmus paper
Lime water turning milky
Squeaky pop
79
Multiple Choice
80

81
Multiple Choice
How do you remove the water to leave solid copper sulfate crystals?
Filter
Evaporate
Distill
82
Multiple Choice
How do you separate an excess insoluble reactant from a solution?
Filtration
Precipitation
Crystalisation
Evaporation
83
Multiple Choice
Why do you need to leave a saturated salt solution for few days?
To allow the salt to crystallize.
84

85
Multiple Choice
Iron + copper oxide =
Iron + copper oxide
Iron oxide + copper oxide
Iron oxide + copper
Magic
86
Multiple Choice

Which of the following is more reactive?
Calcium
Magnesium
Zinc
Magnesium
87
Multiple Choice
Which one of the following combinations result in a displacement reaction?
Iron with magnesium chloride
magnesium with iron chloride
Iron with Zinc Sulphate
gold with silver nitrate
88
Multiple Choice
89
Multiple Choice
What will the products of this reaction be?
calcium + zinc nitrate --------->
Calcium + zinc nitrate
Zinc + calcium nitrate
there will be no reaction
Zinc + calcium chloride
90
Multiple Choice
91

92


Show answer
Auto Play
Slide 1 / 92
SLIDE
Similar Resources on Wayground

89 questions
Science 8 Quarter 2 L1-B
Presentation
•
8th Grade

85 questions
16-3 - The Rise of Sectionalism 2023
Presentation
•
8th Grade

86 questions
Comparative and superlative adjectives
Presentation
•
8th Grade

86 questions
PhySci Final Review 7: Compounds
Presentation
•
9th Grade

85 questions
Experimental Variables (IV & DV)
Presentation
•
8th Grade

87 questions
Naming Compounds
Presentation
•
8th Grade

87 questions
Review Naming Compounds
Presentation
•
8th Grade

86 questions
F3 Special Relationships L41
Presentation
•
8th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade