

PhySci Final Review 7: Compounds
Presentation
•
Science, Chemistry
•
9th Grade
•
Medium
William Jared Lovering
Used 3+ times
FREE Resource
41 Slides • 45 Questions
1
PhySci Final Review 7: Compounds
By William Lovering
2
Ions are elements with a positive (+) or negative (-) charge.
3

4
5

6

7

8
Valence Electrons and Ions
Atoms gain or lose electrons from the highest energy level.
The atoms in the highest energy level are called valence electrons.
To find the number of valence electrons in an atom of a representative element, simply look at its representative group number.
Elements in group 15 like nitrogen have 5 valence electrons.
9
Atoms want to have a "full" outer shell (orbital) of 8 electrons.
Hydrogen (H) and Helium (He) are the only exceptions- They have 2 in the first shell (orbital).
10
Octet Rule
The octet rule determines if an atom will gain or lose electrons.
The outer level of an atom has eight electrons.
An atom will gain or lose electrons to have a full outer energy level.
Atoms of metals tend to lose their valence electrons. Nonmetals tend to gain or share electrons to achieve a full octet.
11
Will an atom gain or lose an electron?
Atoms in group 1, 2, and 13 lose electrons. Remember the atom want a full outer energy level.
For atoms that lose electrons, they empty the outer most energy level.
For groups 1 - 13: atoms lose electrons forming a positive ion.
For groups 15 - 17: atoms gain electrons forming a negative ion.
For group 14: atoms can gain or lose electrons forming a positive or negative ion.
12
Chemical Formulas
In chemical reactions, the atoms in molecules separate and recombine to form new compounds.
To tell which and how many elements are in compounds, we use chemical formulas.
Chemical formulas are a mix of element symbols and subscripts.

13

14
Chemical Formulas
When you look at a chemical formula, it is very similar to an XYZ formula in Math class.
Na2SO4
Na2 = 2 Sodium Atoms
S = 1 Sulfur Atom (when there is no subscript, it is understood to be 1).
O4 = 4 Oxygen Atoms

15

16

17

18

19
Electrons are the Key
If valence electrons are shared by atoms, it is called COVALENT bonding
If valence electrons move freely between atoms, it is called METALLIC bonding
If valence electrons are transferred from one atom to another, it is called IONIC Bonding

20
Ionic Bonds
Form Crystalline Lattice (crystals)
Have high melting and boiling points
Hard
Brittle
Conduct electricity when dissolved in water

21
What you need to remember...
Metals + Nonmetals
Make IONIC bonds
Transfer electrons
High electronegativity difference

22
Covalent Compounds
Have Lower melting and boiling points
Tend to be flammable
When dissolved in water they do not conduct electricity
Soft or brittle solid
Can also form crystals

23
What you need to remember
Nonmetal + Nonmetal
Make COVALENT bonds
Share electrons
Low electronegativity difference

24
Metallic Bonds
good conductors of heat and electricity
malleable and ductile
lustrous
opaque

25
What you need to remember
Metal ions near each other share valence electrons
These valence electrons flow freely between all of the ions
These delocalized electrons are why metals conduct heat and electricity
It is also why metals are malleable and ductile

26
Properties of ionic and covalent molecular substances

27
When naming or writing compounds, the first step is to determine if the compound is ionic or covalent.
What is the difference?
28
Naming Ionic Compounds

29
Rules for Naming Cations
When metals lose electrons they become ions, but their name does not change.

30
Rules for Naming Anions
When nonmetals gain electrons they become ions, and their name does change.

31
Rules of Naming Ions
The names of metals do not change.
Changing the name of nonmetals:
root of element name + -ide = name of anion
Examples:
The name of chlorine’s ion: chlor- + -ide = chloride
The name of calcium’s ion: calcium
(The names of metals don’t change!)
The name of nitrogen’s ion: nitr- + -ide = nitride
32
Steps for Naming Ionic Compounds
Step 1: Write the name of the metal ion.
Step 2: Write the name of the nonmetal ion.
Step 3: YOU ARE DONE! It is that easy.
Example: CaBr2
Calcium Bromide
33
Writing Ionic Compound Formulas
physical science

34
35
36
naming covalent compounds

37

38

39

40

41

42
Multiple Choice
Na ionizes to a charge of _______
+1
+2
-1
-2
43
Multiple Choice
K ionizes to a charge of _________.
+1
+2
-1
-2
44
Multiple Choice
Mg ionizes to a charge of ______.
+1
+2
-1
-2
45
Multiple Choice
Al ionizes to a charge of _______.
+3
-3
+2
-2
46
Multiple Choice
P ionizes to a charge of _______.
+3
-3
+2
-2
47
Multiple Choice
O ionizes to a charge of ________.
+1
+2
-1
-2
48
Multiple Choice
S ionizes to a charge of _______.
-2
-1
+1
+2
49
Multiple Choice

50
Multiple Choice

51
Multiple Choice

52
Multiple Choice

53
Multiple Choice

What is the name of this ion?
oxide
oxygen
54
Multiple Choice

Why are ions formed?
To make our lives difficult
Because atoms want to be stable with 8 valence electrons
Because atoms have the same number of protons and electrons
Because atoms gained neutrons
55
Multiple Choice
An atom with 2 protons, 3 neutrons, and 4 electrons has a charge of ____.
+2
-2
+3
0
56
Multiple Choice

57
Multiple Choice
A bond between a metal and a nonmetal is called a(n)
covalent bond
ionic bond
metallic bond
transfer bond
58
Multiple Choice
What type of bond involves the sharing of electrons between atoms?
covalent bond
ionic bond
metallic bond
transfer bond
59
Multiple Choice
Identify the following compound as ionic or covalent: MgO
ionic
covalent
60
Multiple Choice
Identify the following compound as ionic or covalent: Na2SO4
ionic
covalent
61
Multiple Choice
Identify the following compound as ionic or covalent: SO2
ionic
covalent
62
Multiple Choice
When naming covalent compounds...
You need roman numerals and no prefixes
You do not need roman numerals or prefixes
You need roman numerals and prefixes
You do not need roman numerals but you do need prefixes
63
Multiple Choice
64
Multiple Choice
65
Multiple Choice
66
Multiple Choice
67
Multiple Choice
68
Multiple Choice
69
Multiple Choice
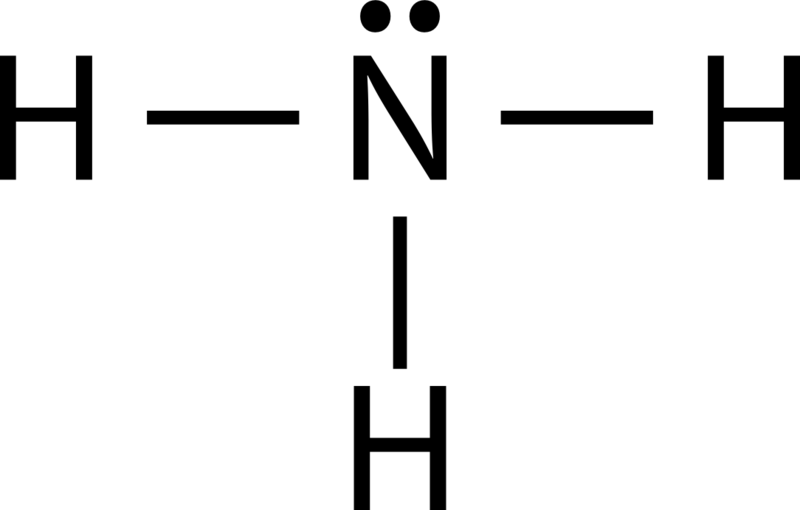
70
Multiple Choice
71
Multiple Choice
72
Multiple Choice
73
Multiple Choice
74
Multiple Choice
75
Multiple Choice
76
Multiple Choice
The correct name for P2O5 is
potassium oxide
dipotassium pentoxide
diphosphorus pentoxide
phosphorus oxide
77
Multiple Choice
The correct name for CaBr2 is
calcium dibromide
calcium (II) bromide
calcium bromide
carbon bromide
78
Multiple Choice
The formula for sulfur hexafluoride is
S6F
SF6
SuF5
SFl6
79
Multiple Choice
What is the name of the compound Mg3N2?
manganese (II) nitride
magnesium nitride
magnesium nitrogen
magnesium (II) nitride
80
Multiple Choice
81
Multiple Choice
82
Multiple Choice
83
Multiple Choice
84
Multiple Choice
Phosphorous trichloride (covalent)
PCl3
P3Cl
P3Cl3
PCL
85
Multiple Choice
What is the NAME of this ionic compound....
Na2S
sodide (II) sulfide
sodium sulfide
disodium sulfide
sodium sulfur
86
Multiple Choice
What is the NAME of this ionic compound....
Mg3N2
magnesium (III) nitrogen
magnide nitride
magnesium (II) nitrogen
magnesium nitride
PhySci Final Review 7: Compounds
By William Lovering
Show answer
Auto Play
Slide 1 / 86
SLIDE
Similar Resources on Wayground

87 questions
Bonding and Nomenclature Unit Review
Presentation
•
10th Grade

87 questions
Chapter 2 Section 6: Investigate
Presentation
•
8th Grade

87 questions
Forces Objectives
Presentation
•
8th Grade

72 questions
AKS 10 - Electricity and Magnetism
Presentation
•
8th Grade

84 questions
EOT REVIEW LESSON - GRADE 8
Presentation
•
8th Grade

84 questions
Physics Motion- Yr 10
Presentation
•
10th Grade

75 questions
Properties of matter
Presentation
•
9th Grade

77 questions
Energy Unit Review 25/26
Presentation
•
8th Grade
Popular Resources on Wayground

20 questions
STAAR Review Quiz #3
Quiz
•
8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

6 questions
Marshmallow Farm Quiz
Quiz
•
2nd - 5th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for Science

46 questions
Unit 4 Geosphere Test Review
Quiz
•
9th - 12th Grade

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

16 questions
Ecosystem Stability
Quiz
•
9th Grade

20 questions
Mendelian Genetics Review
Quiz
•
9th Grade

12 questions
Unit 6 Quiz #3 (Life Cycles of Stars)
Quiz
•
9th Grade

34 questions
Human Impact on Ecosystems Quiz
Quiz
•
9th Grade

10 questions
Exploring Alfred Wegener's Continental Drift Theory
Interactive video
•
6th - 10th Grade