

Chromatography
Interactive Video
•
Chemistry
•
12th Grade
•
Practice Problem
•
Easy
Tom Barclay
Used 3+ times
FREE Resource
17 questions
Show all answers
1.
SLIDE QUESTION
30 sec • Ungraded
2.
LABELLING QUESTION
1 min • 1 pt
Label the two examples as adsorption or absorption
Adsorption
Absorption
3.
SLIDE QUESTION
30 sec • Ungraded
4.
MULTIPLE SELECT QUESTION
45 sec • 1 pt
What is required to make a molecule polar?
At least one bond between atoms with a difference in electronegativity of 0.5 or higher
One polar bond in a large molecule is enough to make the molecule very polar overall
At least one bond between atoms with any difference in electronegativity
Molecule shape around a polar polar bond that puts the centres of the partial negative and partial positve charges in different places
Large molecules require multiple polar centres to be polar overall
5.
MATCH QUESTION
30 sec • 1 pt
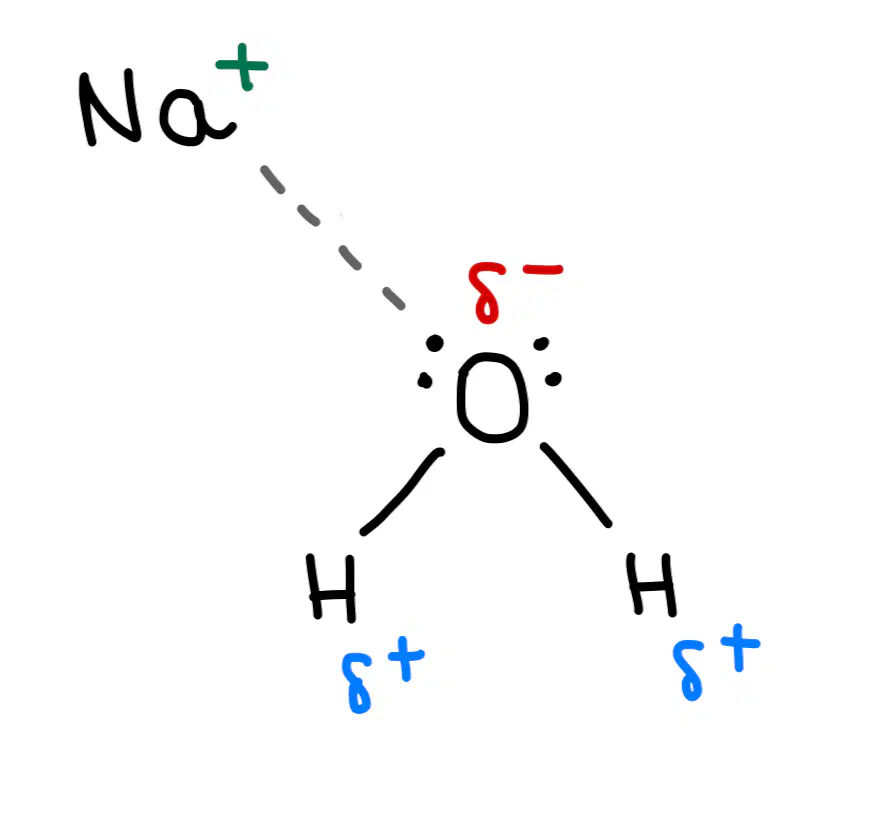
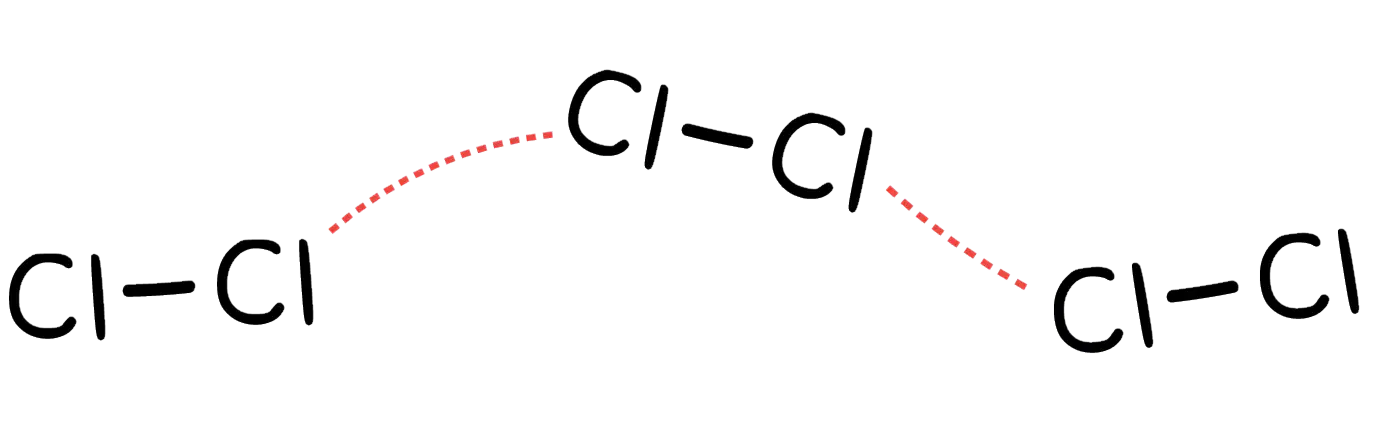
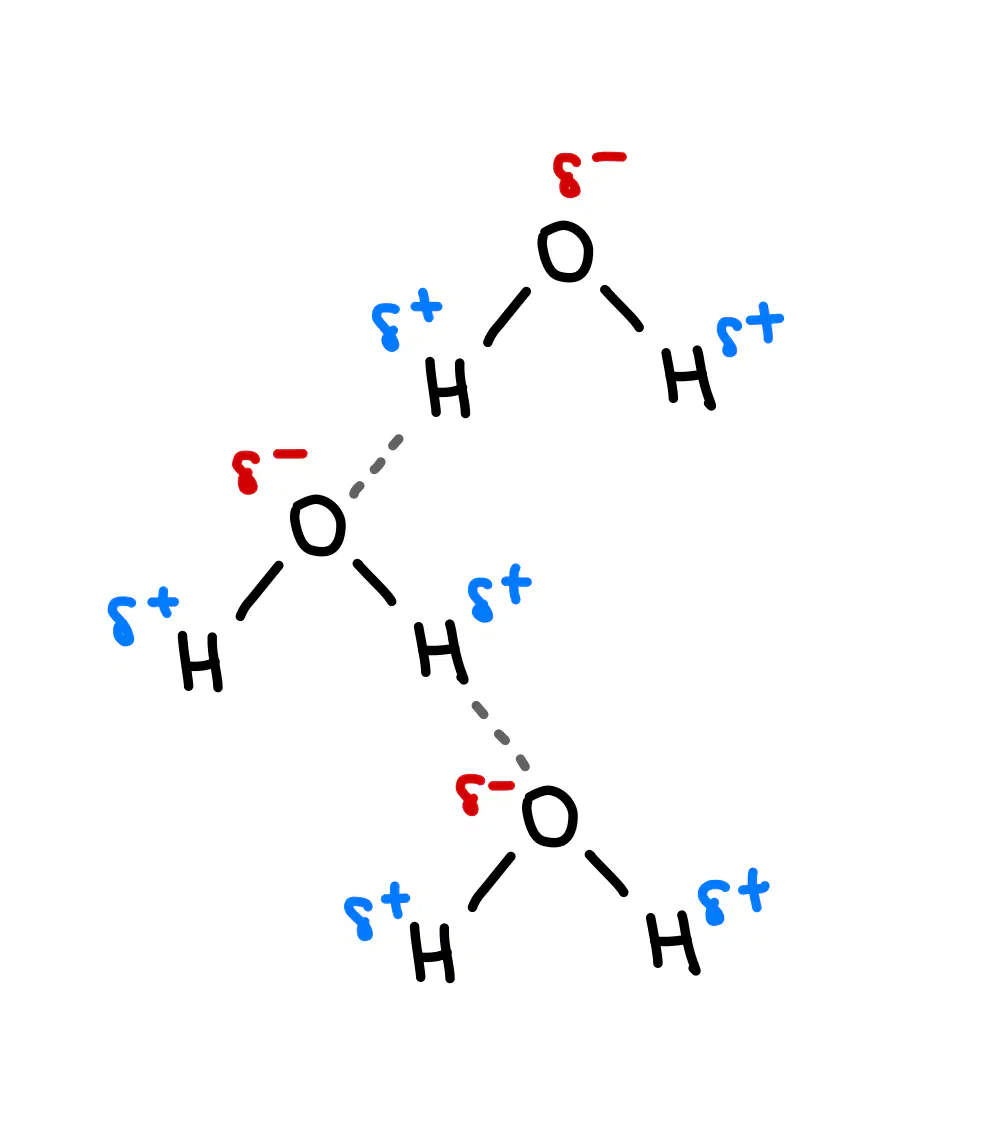
Give the best match between the image and the secondary interactions
Hydrogen bond
Dispersion force

Dipole-dipole

Ion-dipole
Trick answer - covalent bonds shown
6.
REORDER QUESTION
30 sec • 1 pt
Place the secondary interactions in order of strength (1 being the strongest)
Ion-dipole
Hydrogen bonding
Dipole-dipole
Dispersion forces
7.
SLIDE QUESTION
30 sec • Ungraded
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
