

Parts of a Chemical Equations
Presentation
•
Science
•
7th Grade
•
Hard
James Gonzalez
FREE Resource
2 Slides • 19 Questions
1

2
Multiple Choice

Students make pizzas to model different atoms. They use pepperoni slices to represent protons, mushrooms to represent electrons, and pieces of onion to represent neutrons. Which pizza
most accurately models an atom containing 3 protons, 3 electrons, and 3 neutrons?
3
Multiple Choice

Desalination is a method of making fresh water from salt water. A student makes a model desalination factory. The student heats salt water in container 1 and collects fresh water in container 2. The diagram shows the model desalination factory, and the table contains the boiling points of water and salt.
How does the process of desalination demonstrate that salt water is a mixture?
The salt water's temperature can increase when placed above a burner.
The volume of fresh water in container 2 is less than the salt water in container 1.
Two diff erent types of pure substances are collected in container 2.
Water can be separated from salt because water has a lower boiling point.
4
To Do:
Today we will look at different word problems and work some of them out with our skittles.
Once done, you may eat your skittles.
Now think back to what we have been working on with chemical equations: Coefficients, Subscripts, Parenthesis, Balanced or Unbalanced
5
Fill in the Blanks
6
Fill in the Blanks
7
Fill in the Blanks
8
Fill in the Blanks
9
Draw
Take 3 different colored skittles and lay out 1 color for each element. Ex: Na, you choose red skittles. How many red skittles do you need for 2Na?
10
Multiple Choice

Does the following equation support the Law of Conservation of Mass? Show with your skittles.
Yes
No
11
Multiple Choice

Does the following equation support the Law of Conservation of Mass? Show your work with your skittles.
Yes
No
12
Multiple Choice

Which side is the reactant?
13
Multiple Choice

Which side is the product?
14
Multiple Choice
If 153 g of iron reacted with 22 g of oxygen gas to form iron oxide, how much iron oxide would be produced if no reactants were left over?
153 g - 22 g = 131 g
153 g + 22 g = 175 g
15
Multiple Choice
In an experiment, students started with 176.3 grams of reactants. In a totally closed system, what will be the mass of the products?
0 g
176.3 g
16
Draw
Count the atoms in the equation pictured. Once done, show your work with your skittles.
17
Multiple Choice
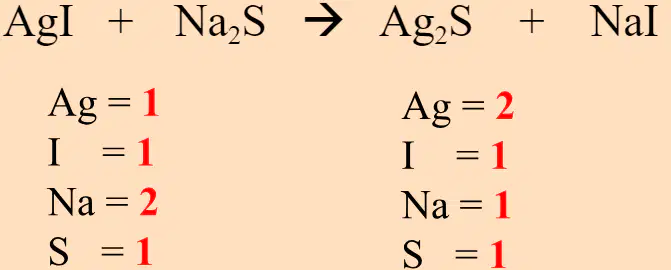
What is wrong with the previous equation?
There is an extra silver (Ag) atom on the reactant side and a missing sulfur (S) atom on the product side.
There is an extra silver (Ag) atom on the reactant side and an extra sodium (Na) atom on the product side.
There is a missing silver (Ag) atom on the reactant side and a missing sodium (Na) atom on the product side.
There is an extra iodine (I) atom on the reactant side and a missing sodium (Na) atom on the product side.
18
Multiple Choice
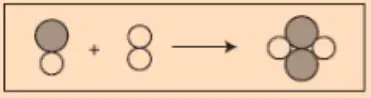
Does the image follow LOCOM? Model with your skittles.
19
Multiple Choice

Does this image follow LOCOM? Model with your skittles.
Yes
No
20
Multiple Choice
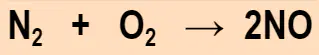
Evaluate the equation attached. Which model demonstrates the Law of Conservation of Mass according to the equation? Use your skittles.
21
Poll
How are you feeling about LOCOM so far?
Great!
Good!
Okay!
Need more help!

Show answer
Auto Play
Slide 1 / 21
SLIDE
Similar Resources on Wayground

18 questions
7.8B Weathering, Erosion, and Deposition
Presentation
•
7th Grade

18 questions
Levels of Organization
Presentation
•
7th Grade

19 questions
7.7B Forces in Organisms Lesson
Presentation
•
7th Grade

16 questions
Roman Numerals
Presentation
•
7th Grade

16 questions
Net Force
Presentation
•
7th Grade

15 questions
Chemical Compounds
Presentation
•
7th Grade

17 questions
7th Science- Human Impacts (Water Edition)
Presentation
•
7th Grade

15 questions
Bacteria
Presentation
•
7th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

20 questions
genetics, punnett squares, heredity
Quiz
•
7th Grade

16 questions
Photosynthesis and Cellular Respiration Review
Quiz
•
7th Grade

20 questions
Food Webs + Energy Pyramids
Quiz
•
7th Grade

10 questions
Exploring the Rock Cycle
Interactive video
•
6th - 8th Grade

10 questions
Exploring Earth's Seasons and Their Causes
Interactive video
•
6th - 8th Grade

26 questions
Amplify Chemical Reactions
Quiz
•
6th - 8th Grade

19 questions
Natural and Artificial Selection
Quiz
•
7th Grade