

Average Atomic Mass and Relative Abundance
Presentation
•
Chemistry
•
9th - 12th Grade
•
Hard
Joseph Anderson
FREE Resource
10 Slides • 10 Questions
1
Atomic Number, Average Atomic Mass, Mass Number, Isotopes

2
3

4
5
Multiple Choice
How is average atomic mass determined?
By adding all of the protons and neutrons together.
by summing the masses of the element's isotopes, each multiplied by its natural abundance on Earth
By multiplying of often protons and neutrons occur and adding up the averages.
6
Multiple Choice
What are AMU's
Atomic Mass University
Atomic Mass Units
Atoms mean unity
All Mass United
7
Multiple Choice
What are AMU's
The unit used to measure the mass of atoms and molecules. Equal to 1/12 the mass of Carbon-12
Unit to measure the radius of an atom
Equal to a gram
The unit used to measure large quantities of atoms
8
Multiple Choice
What does the atomic number represent?
The number of electrons
The number of protons
The number of neutrons
The number of protons and neutrons together.
9

10

11

12

13
Isotope Symbols
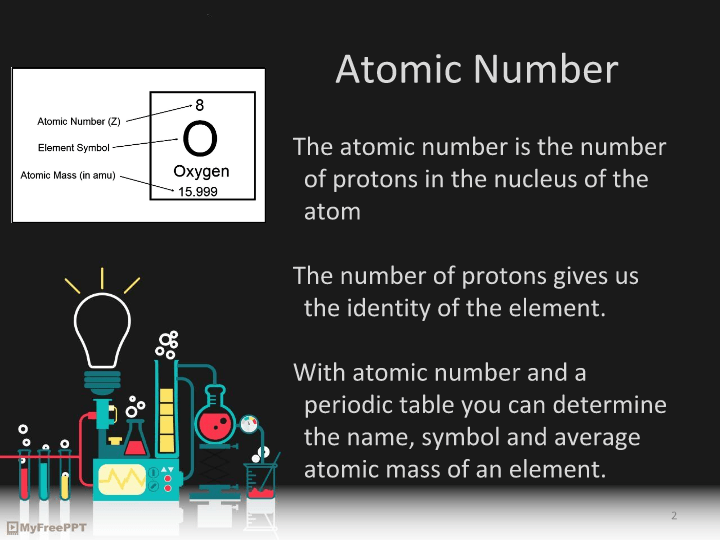
As you can see to the right there are certain ways elements and isotopes are represented as symbols. There is the element symbol (the biggest part). Then the upper number will the the mass number (number of protons + number of neutrons) and the lower number is just the atomic number (number of protons).

14
Isotope Symbol Examples
As you can see here the element is Cu (copper), and it has a mass number of 63 and atomic number of 29. With this information I can determine that this copper atom has 34 neutrons.

15
Multiple Choice
What does the mass number represent?
The number of electrons
The number of protons
The number of neutrons
The number of protons and neutrons together.
16
Multiple Choice
What does the atomic number represent?
The number of electrons
The number of protons
The number of neutrons
The number of protons and neutrons together.
17
Multiple Choice
If an atom has 5 protons and 6 neutrons what is the proper name of the isotope?
Boron-1
Boron-11
Carbon-11
Carbon 11
Boron Carbonate
18
Multiple Choice

How many neutrons does an atom of symbol pictured have?
26
-26
70
19
Multiple Choice

How many protons does an atom of symbol pictured have?
22
26
70
20
Multiple Choice

What is the total number of particles in the nucleus atom of symbol pictured have?
22
70
48
Atomic Number, Average Atomic Mass, Mass Number, Isotopes

Show answer
Auto Play
Slide 1 / 20
SLIDE
Similar Resources on Wayground

14 questions
Acid Naming & Formula Writing
Presentation
•
10th - 11th Grade

14 questions
Intro to Matter
Presentation
•
9th - 12th Grade

15 questions
Compound-complex sentences
Presentation
•
9th - 12th Grade

15 questions
Finding Zeros of a Function Lesson
Presentation
•
9th - 12th Grade

15 questions
Completing the Square
Presentation
•
9th - 12th Grade

17 questions
Percent Composition
Presentation
•
9th - 12th Grade

19 questions
Structure of Atom
Presentation
•
9th - 12th Grade

16 questions
Mole Conversions & Percent Composition Practice
Presentation
•
10th - 12th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Interpreting Solubility Curves
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
10th Grade

43 questions
Acids and Bases Review
Quiz
•
10th - 11th Grade

20 questions
Thermochemistry Review
Quiz
•
10th Grade

25 questions
Unit 4 (Mid) Rumble
Quiz
•
10th Grade