

Titration Calculations
Presentation
•
Chemistry
•
11th - 12th Grade
•
Hard
Joseph Anderson
FREE Resource
13 Slides • 14 Questions
1
Titration
2
Titration is a laboratory technique used to determine the concentration of a substance in a solution.
It involves adding a known volume of a solution with a known concentration (the titrant) to a solution of the substance being analyzed (the analyte) until a reaction occurs, and the concentration of the analyte can be calculated based on the amount of titrant needed to complete the reaction.

3
Titration involves several steps, including:
Preparation
Indicator Selection
Titration
Calculation
4
Preparation of the solutions: The unknown solution is placed in a flask or beaker, and a known volume of the titrant is measured out.
The titrant is usually added to a burette, which is a long, graduated glass tube used for precise measurement of liquid volumes.
5
Indicator selection: An indicator is added to the unknown solution, which will change color or signal some other change when the reaction between the unknown and titrant is complete.
6
Titration: The titrant is gradually added to the unknown solution while stirring until the indicator signals that the reaction is complete.
The point at which this occurs is called the equivalence point and is the point at which the number of moles of titrant added is stoichiometrically equivalent to the number of moles of the substance being titrated.
7
Calculation of results: The volume of titrant required to reach the equivalence point is measured and used to calculate the concentration of the unknown solution.
8
In titration, a burette is typically used to add the titrant slowly and precisely to the analyte solution, while the analyte solution is continuously stirred or swirled.
The reaction between the two solutions is typically monitored using an indicator, which changes color or other properties when the reaction is complete.
9
Multiple Choice

What is this piece of apparatus called
Pipette
Burette
Janette
Cuvette
10
Multiple Choice

What is the reading on this burette?
24.0cm3
25.8cm3
24.2cm3
23.9cm3
11
Multiple Choice
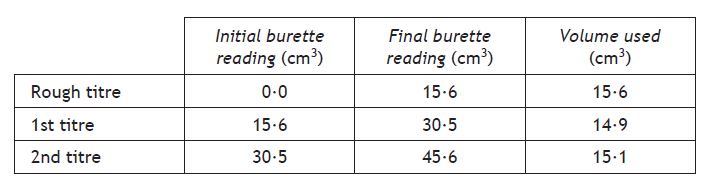
Calculate the average volume of acid needed for this neutralisation
15.2cm3
15.0cm3
35.5cm3
30.1cm3
12
Multiple Choice

4.40mL
3.50mL
3.60mL
4.50mL
13
Multiple Choice
salt + hydrogen
salt + water
salt + carbon dioxide + water
salt
14
Multiple Choice
NaH + ClOH
NaCl + H2
NaCl + H2O
NaCl + Cl2
15
Multiple Choice
Phenolphthalein
Bromothymol Blue
Litmus
Universal
16
Multiple Choice
What is the white tile or white paper for in titration?
To help clearly see the color change
To protect the conical flask
To add extra height
To assist the reaction
17
Multiple Choice
an acid
a base
a neutral
18
Multiple Choice
pH 1
pH 4
pH 8
pH 13
19
If, for example, your results were:
25.0cm3 of sodium hydroxide required 20.0cm3 of 0.100 mol dm-3 hydrochloric acid for neutralisation.
What is the concentration of the sodium hydroxide?
20

21

22

23

24
Multiple Choice
30mL of NaOH is neutralized by 12.3mL of 0.2M HCl. What is the concentration of the NaOH.
82 mol/l
0.82 mol/l
0.49 mol/l
0.082 mol/l
25
Multiple Choice
The reaction of perchloric acid (HClO4) with lithium hydroxide (KOH) is described by the equation
HClO4 + KOH → KClO4 + H2O
Suppose 100 mL of perchloric acid is neutralized by exactly 50.0 mL of 1.0 M of Potassium hydroxide. What is the concentration of the perchloric acid?
0.5M
50M
2.0M
1.0M
26
Multiple Choice
If it takes 50 mL of 0.5 M Ca(OH)2 to neutralize 125 mL of hydrochloric acid (HCl), what is the concentration of the acid?
0.2 M
5 M
0.5 M
0.4 M
27
Multiple Choice
1.392 M
0.155 M
0.718 M
Titration
Show answer
Auto Play
Slide 1 / 27
SLIDE
Similar Resources on Wayground

19 questions
Electron configuration
Presentation
•
11th - 12th Grade

20 questions
Mixed Numbers
Presentation
•
4th - 5th Grade

21 questions
ohms law
Presentation
•
11th - 12th Grade

21 questions
Significant Figure and Scientific Notation Review
Presentation
•
10th - 11th Grade

19 questions
More Venn Diagrams
Presentation
•
11th - 12th Grade

19 questions
Homeostasis
Presentation
•
11th - 12th Grade

20 questions
Oxidation State Practice
Presentation
•
10th - 12th Grade

23 questions
Ecosystems and Biomes
Presentation
•
11th - 12th Grade
Popular Resources on Wayground

15 questions
Grade 3 Simulation Assessment 1
Quiz
•
3rd Grade

22 questions
HCS Grade 4 Simulation Assessment_1 2526sy
Quiz
•
4th Grade

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

20 questions
Math Review
Quiz
•
3rd Grade
Discover more resources for Chemistry

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Redox Reactions
Quiz
•
10th - 12th Grade

20 questions
pH and pOH Calculations
Quiz
•
10th - 11th Grade

36 questions
Unit 4 (Part 2) Solutions
Quiz
•
10th - 12th Grade

30 questions
Final Exam Prep S22: 2.1.A.1 Isotopes
Quiz
•
9th - 12th Grade

25 questions
Stoichiometry Mass to Mass
Quiz
•
10th - 11th Grade

15 questions
Calculating pH and pOH
Quiz
•
10th - 12th Grade

15 questions
Balancing Chemical Equations
Quiz
•
10th - 12th Grade