

Atomics
Presentation
•
Chemistry
•
11th Grade
•
Hard
Joseph Anderson
FREE Resource
5 Slides • 27 Questions
1
Test #1 Review: Atomic Concepts
TEST TOMORROW 10/7!
2
Atomic Theory Discoveries
JJ Thompson
- Discovered electrons (negative)
Rutherford
- Discovered nucleus is positive and contains protons
- atom is mostly empty space
Neils Bohr
-planetary model
3
Subatomic Particles
Protons
- positive charge
- equal to the atomic number
Electrons
- negative charge
- equal to the atomic number
Neutrons
- no charge
- equal to the atomic mass - atomic number
4

5
Multiple Choice
Which device did JJ Thomson use to discover the electron?
X-ray machine
scanning tunneling electron microscope
spectrometer
cathode ray tube
6
Multiple Choice
JJ Thomson was credited for the discovery of the
center of the universe
proton
electron
neutron
7
Multiple Choice
Which is a finding attributed to Rutherford's Gold Foil Experiment?
atoms are indivisible and have a dense positive nucleus.
atoms are divisible and have multiple layers of electrons scattered uniformly about the atom.
atoms are like plum pudding with positive and negative charges uniformly scattered throughout the atom.
atoms are mostly empty space with a dense positive nucleus.
8
Multiple Choice
What evidence did Rutherford use to support the idea that the atom was mostly empty space?
the vast majority of the alpha particles went through the foil unaffected.
the vast majority of the alpha particles were deflected at 90 degrees.
one alpha particle was deflected by the gold foil.
beams of alpha particles were deflected by magnets.
9
Multiple Choice
According to the planetary model of the atom, electrons_____?
are all packed inside the nucleaus of the atom
freely move in and out of the nucleus
revolve around the nucleus in specific orbits
electrons do not exist, atoms only contain protons and neutrons
10
Multiple Choice
Which subatomic particle has a positive charge?
proton
neutron
electron
midichlorian
11
Multiple Choice
The nucleus of an atom contains which subatomic particles?
protons and electrons
protons and neutrons
electrons and neutrons
protons, electrons, and neutrons
12
Multiple Choice

13
Multiple Choice

14
Isotopes
Isotopes are defined as having the same number of protons, but different number of neutrons
- atomic number is the same
- atomic mass is different

15
Multiple Choice
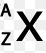
atomic mass
16
Multiple Choice
17
Multiple Choice
Isotopes are atoms of the same element with the same # of _________, different number of ______.
neutrons; protons
protons; neutrons
18
Multiple Choice
19
Multiple Choice

What is the planetary model also known as?
Thomson model
Plum pudding model
Bohr model
Nuclear model
20
Multiple Choice
21
Multiple Choice
22
Multiple Choice
23
Multiple Choice
24
Multiple Choice
Which 2 subatomic particles make up most of an atom's mass (mass number)?
protons and neutrons
all subatomic particles are equal in number
neutrons and electrons
protons and electrons
25
Multiple Choice
How many electrons does a Copper atom have?
63
29
92
34
26
Multiple Choice
27
Multiple Choice
An element is defined by its number of ...
protons
electrons
neutrons
protons + electrons
28
Multiple Choice
Negative ions ____ electrons.
Gain
Lose
29
Multiple Choice
30
Multiple Choice
31
Multiple Choice
What is the average mass of this element?
32
Multiple Choice
Naturally occurring chlorine that is put in pools is: 75.53% chlorine with a mass of 34.969 amu and 24.47% chlorine with a mass of 36.966 amu. Calculate the average atomic mass.
35.46 amu
35.00 amu
37.00 amu
17.00 amu
Test #1 Review: Atomic Concepts
TEST TOMORROW 10/7!
Show answer
Auto Play
Slide 1 / 32
SLIDE
Similar Resources on Wayground

24 questions
Electronic Structure of Atoms & The Periodic Table
Presentation
•
11th Grade

28 questions
History of the Periodic Table
Presentation
•
10th - 12th Grade

22 questions
Dimensional Analysis
Presentation
•
10th Grade

25 questions
Sistem Koloid
Presentation
•
11th Grade

27 questions
Chemistry Chapter 3 Section 1
Presentation
•
11th Grade

27 questions
Periodic Table and Valence electrons
Presentation
•
University

27 questions
Survey of Chemistry
Presentation
•
11th Grade

26 questions
Quantum Theory and the Atom
Presentation
•
11th Grade
Popular Resources on Wayground

15 questions
Grade 3 Simulation Assessment 1
Quiz
•
3rd Grade

22 questions
HCS Grade 4 Simulation Assessment_1 2526sy
Quiz
•
4th Grade

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

20 questions
Math Review
Quiz
•
3rd Grade
Discover more resources for Chemistry

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Redox Reactions
Quiz
•
10th - 12th Grade

20 questions
pH and pOH Calculations
Quiz
•
10th - 11th Grade

36 questions
Unit 4 (Part 2) Solutions
Quiz
•
10th - 12th Grade

30 questions
Final Exam Prep S22: 2.1.A.1 Isotopes
Quiz
•
9th - 12th Grade

25 questions
Stoichiometry Mass to Mass
Quiz
•
10th - 11th Grade

15 questions
Calculating pH and pOH
Quiz
•
10th - 12th Grade

15 questions
Balancing Chemical Equations
Quiz
•
10th - 12th Grade