

Atoms and Elements
Presentation
•
Science
•
8th Grade
•
Hard
Joseph Anderson
FREE Resource
8 Slides • 10 Questions
1
Atoms and the Periodic Table

2
What is an atom?
An atom is the building block of all matter.
Atoms are very small; smaller than the tip of your pencil.
Atoms are composed of particles called protons, electrons and neutrons.
Atoms can join together to form molecules, which in turn form most of the objects around you.

3
Multiple Choice

What is at the center of every atom?
an electron
only protons
a molecule
a nucleus
4
The Periodic Table of the Elements
display of the chemical elements, which are arranged by atomic number, electron configuration, and recurring chemical properties
The seven rows of the table, called periods, generally have metals on the left and nonmetals on the right.
The columns, called groups, contain elements with similar chemical behaviors.

5
Multiple Choice

What is the Periodic Table?
a chart that lists all the molecules that exist in the universe
a chart that shows all the elements scientists have discovered
a chart that shows all the compounds found in the world
a chart that shows what time periods elements were discovered in
6
Chemical Element
Atomic number- tells the number of protons, electrons and the location of the element on the Periodic Table
Atomic Mass- the sum of the protons and neutrons in the nucleus of the atom
To find the number of neutrons, subtract the atomic number from the atomic mass
APE MAN

7
Multiple Choice
What does an element's atomic number represent?
the element state at room temperature
how metallic an element is
the number of atoms in a compound
the number of protons in an element
8
Electron Shell Rules:
1st shell can hold up to 2 electrons
2nd shell can hold up to 8 electrons
3rd shell can hold up to 18 electrons

9
Multiple Choice
Which of the following is the correct atomic model for Boron?
10
Multiple Choice

How many neutrons are in Potassium?
*Click the image to make it larger.
19
20
39
58
11
The three basic states of matter

12

13
Multiple Choice
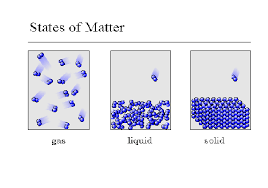
At higher temperatures
particles in an object have less energy
particles in an object move faster
a gas contracts
14
Multiple Choice
How is a gas defined?
no shape, no volume
Definite shape, definite volume
Found in stars
Definite volume, no shape
15
All matter have properties.

16
Multiple Choice
Physical properties are
are properties that can be observed without changing the matter's identity
are properties that can be observed that change the matter into new matter with new properties
17
Multiple Choice
Which of the following is a sign that a chemical reaction has occurred?
change in shape
melting
formation of a gas
dissolving
18
Poll
How would you rate your understanding of atoms and the Periodic Table?
Atoms and the Periodic Table

Show answer
Auto Play
Slide 1 / 18
SLIDE
Similar Resources on Wayground

15 questions
Weather Fronts
Presentation
•
8th Grade

13 questions
PMA Review-Rock Cycle
Presentation
•
8th Grade

16 questions
Intro to the Solar System
Presentation
•
8th Grade

12 questions
Lesson 9: Newton's Second Law
Presentation
•
8th Grade

14 questions
Environmental Changes
Presentation
•
7th - 8th Grade

12 questions
Genes, Chromosomes, and DNA
Presentation
•
7th - 8th Grade

12 questions
Adaptations
Presentation
•
8th Grade

12 questions
Which Chemical Reaction is Occuring
Presentation
•
8th Grade
Popular Resources on Wayground

15 questions
Grade 3 Simulation Assessment 1
Quiz
•
3rd Grade

22 questions
HCS Grade 4 Simulation Assessment_1 2526sy
Quiz
•
4th Grade

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

20 questions
Math Review
Quiz
•
3rd Grade
Discover more resources for Science

20 questions
8th Grade Science STAAR Review
Quiz
•
8th Grade

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

20 questions
Earth Day
Quiz
•
3rd - 12th Grade

30 questions
GMAS Physical Science Review
Quiz
•
8th Grade

100 questions
GMAS Science Review
Quiz
•
8th Grade

5 questions
History of Earth Day
Passage
•
6th - 8th Grade

100 questions
STAAR Review
Quiz
•
8th Grade

10 questions
Exploring Earth's Seasons and Their Causes
Interactive video
•
6th - 8th Grade