

Chemistry Unit Review
Presentation
•
Science
•
8th Grade
•
Hard
Joseph Anderson
FREE Resource
5 Slides • 58 Questions
1
Chemistry Unit Review

Atoms, Periodic Table, Formulas, and Changes
2

Parts Of The Atom
3
Multiple Choice
4
Multiple Choice
5
Multiple Choice
6
Multiple Choice
7
Multiple Choice
8
Multiple Choice
9
Multiple Choice
10
Multiple Choice
11
Multiple Choice

12
Multiple Choice

13
Multiple Choice

14
Multiple Choice

15
Multiple Choice

16
Multiple Choice
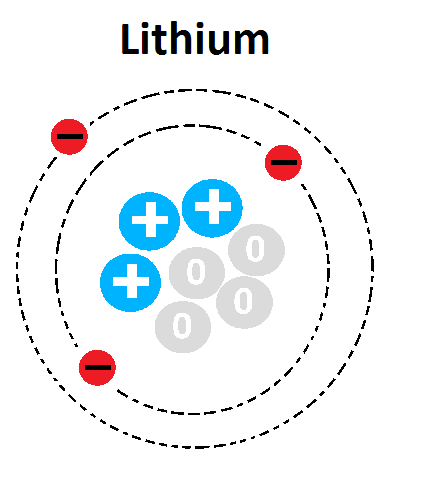
What is the mass number of this atom?
1
3
4
7
17
Multiple Choice

Name the element
Boron
Carbon
Nitrogen
Oxygen
18
Multiple Choice

19

The Periodic Table
20

The GROUP an atom is in tells you the number of VALENCE ELECTRONS and how it will REACT
The PERIOD an atom is in tells you how many ENERGY LEVELS an atom has.
21
Multiple Choice
22
Multiple Choice

23
Multiple Choice
24
Multiple Choice
25
Multiple Choice
26
Multiple Choice
27
Multiple Choice
28
Multiple Choice
29
Multiple Choice
30
Multiple Choice
31
Multiple Choice
32
Multiple Choice
33
Multiple Choice
34
Multiple Choice
35
Multiple Choice
36
Multiple Choice

37
Multiple Choice

38
Multiple Choice

39
Multiple Choice

40
Multiple Choice
41
Multiple Choice
42

Chemical Formulas And Reactions
43
Multiple Choice

44
Multiple Choice
45
Multiple Choice

46
Multiple Choice

47
Multiple Choice

48
Multiple Choice

49
Multiple Choice

50
Multiple Choice

51
Multiple Choice

52
Multiple Choice
53
Multiple Choice
Al + O2 --> 2Al2O3
54
Multiple Choice
55
Multiple Choice
2H2 + O2 --> 2H20
5g 20g ?
56
Multiple Choice
H2 + O2 → H2O
57
Multiple Choice
58
Multiple Choice
59
Multiple Choice
60
Multiple Choice
CaO + 3 C → CaC2 + CO
61
Multiple Choice
HCl + Zn → H2 + ZnCl2
62
Multiple Choice
63
Multiple Choice
Chemistry Unit Review

Atoms, Periodic Table, Formulas, and Changes
Show answer
Auto Play
Slide 1 / 63
SLIDE
Similar Resources on Wayground

56 questions
Physical Science
Presentation
•
8th Grade

56 questions
Strata and Fossils
Presentation
•
8th Grade

56 questions
Fossils and Geologic Time
Presentation
•
8th Grade

56 questions
Element, Molecule, Compound
Presentation
•
8th Grade

56 questions
Petrified Fossils
Presentation
•
8th Grade

58 questions
Ecology
Presentation
•
9th Grade

60 questions
Spheres, Layers, Rocks
Presentation
•
8th Grade

56 questions
HR Diagram
Presentation
•
8th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

12 questions
PS.8.1.2, PS.8.1.3, PS.8.1.5 Review
Quiz
•
8th Grade

20 questions
Moon Phases and Eclipses
Quiz
•
6th - 8th Grade

22 questions
6th & 8th Grade Science Material SOL Review
Quiz
•
6th - 8th Grade

49 questions
NC Released EOG 8 Science (2024)
Quiz
•
8th Grade

48 questions
8th Grade NC Science EOG Review 2
Quiz
•
8th Grade

19 questions
Renewable and Nonrenewable Resources (CK)
Quiz
•
8th Grade

20 questions
8th Grade Science STAAR Review
Quiz
•
8th Grade