

Counting Atoms with Coefficients
Presentation
•
Science
•
6th - 8th Grade
•
Hard
Joseph Anderson
FREE Resource
18 Slides • 24 Questions
1
Chemical Reactions/Counting Atoms

2

3
Open Ended

List anything you know about atoms.
4

5

6
Terminology: Element
A chemical that cannot be broken down into another substance
Each unique capital letter represents a different element.

7
Multiple Choice

What do the CAPITAL letters represent in a molecular formula?
molecules
elements
atoms
8
Atom
The smallest part of a chemical element.
Combining atoms creates molecules.

9
Multiple Choice
Combining _____ creates molecules.
Legos
nucleus
atoms
10

11
Multiple Choice
How many atoms make a molecule?
only 1
2 or more
12

13

14
Multiple Choice
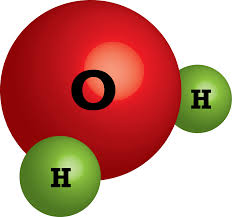
15

16
Multiple Choice
How many atoms of Hydrogen are in H2O
1
2
3
more than 3
17

18
Multiple Choice
19

20
Molecular Formula Examples

21

22

23
Law of conservation of matter
- Energy cannot be created or destroyed
-It can be moved from one object to another

24
Multiple Choice

There are 2 (O) oxygen atoms in the reactants. How many will be in the PRODUCT?
1
2
3
25
Multiple Choice

26
Multiple Choice

Write the chemical formula for the picture:
F2O
O2F
2 FO
USE YOUR NOTES
27
Multiple Choice

Write the chemical formula for the picture:
N3F
3 FN
F3N
USE YOUR NOTES
28
Multiple Choice

The chemical formula is C4H10. How many HYDROGEN (H) atoms are in one molecule of this?
1
4
10
29
Multiple Choice

What is the chemical formula for this picture? (1 green circle & 4 purple circles to represent atoms).
CH4
NH3
H2O
30
Multiple Choice

How many total atoms are in the compound for ammonia, NH3
2
3
4
5
31
Multiple Choice

The right side of the equation (NaCl) is called the
product
reactants
32
Multiple Choice
The ______ are numbers found on the lower right-hand side of an element symbol.
Coefficient
Exponent
Multiplier
Subscript
33
Multiple Choice

How many total ATOMS are shown in this picture?
1
3
4
34
Multiple Choice
How many total atoms are in H2SO4?
3
5
6
7
35
Potential &
Kinetic Energy
https://www.youtube.com/watch?v=zCKeniklH_c

36
Multiple Choice
What is the definition for energy in MOTION (moving)?
Potential energy
Kinetic energy
37
Multiple Choice

A slingshot that has been pulled back, but has not been let go.
potential
kinetic
38
39
Multiple Choice

An arrow is pulled back but hasn't been released from the bow yet.
potential
kinetic
40
Multiple Choice

When a roller coaster is at the top of a hill it has
high potential energy
high kinetic energy
low potential energy
equal amounts of kinetic and potential energy
41
Multiple Choice

42
Multiple Choice

At which point is potential energy greatest?
W
X
Y
Z
Chemical Reactions/Counting Atoms

Show answer
Auto Play
Slide 1 / 42
SLIDE
Similar Resources on Wayground

34 questions
Calculating Density
Presentation
•
6th - 8th Grade

36 questions
Levels of Organization - Cells to Biome
Presentation
•
KG

38 questions
20.3 Energy in Ecosystems
Presentation
•
6th - 8th Grade

36 questions
Ecological Succession & Human Impact on Ground&Surface water
Presentation
•
7th Grade

37 questions
Virtual Lab Assignment (Physical and Chemical Changes)
Presentation
•
6th - 8th Grade

36 questions
Relationships in the Universe
Presentation
•
6th - 8th Grade

37 questions
Elements of the Earth
Presentation
•
5th - 8th Grade

37 questions
Substances and Mixtures
Presentation
•
6th - 9th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

20 questions
Rocks and The Rock Cycle
Quiz
•
6th Grade

20 questions
genetics, punnett squares, heredity
Quiz
•
7th Grade

16 questions
Photosynthesis and Cellular Respiration Review
Quiz
•
7th Grade

30 questions
GMAS Physical Science Review
Quiz
•
8th Grade

20 questions
Human Body Systems
Quiz
•
6th Grade

15 questions
Punnett Squares
Quiz
•
6th Grade

20 questions
Food Webs + Energy Pyramids
Quiz
•
7th Grade