

Lesson on Moles
Presentation
•
Chemistry
•
9th - 12th Grade
•
Hard
Joseph Anderson
FREE Resource
23 Slides • 21 Questions
1
Moles - Part 1
What is a mole and why do we need it?

2
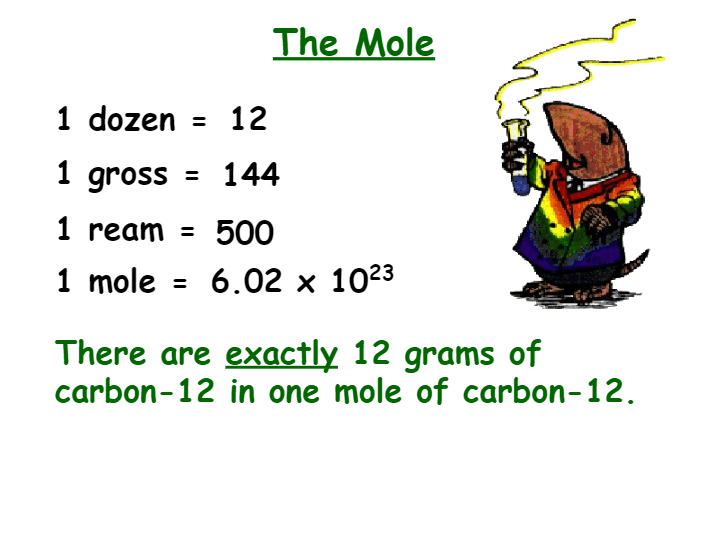
3

4

5
Multiple Choice
Which would have more atoms?
1 mole of Li
1 mole of Au
1 mole of Si
None, all are equal
6
Multiple Choice
How many particles are in a mole?
602
602 million
602 moles
6.02 x 1023
7
Multiple Choice
Which of the following statements regarding the mole is INCORRECT?
A mole is a unit of quantity equal to 6.02 x 1023 particles.
The number of particles in a mole is known as Avogadro’s number.
A mole of particles of an element is numerically equal to the atomic mass of the element.
none of the above
8

9

10

11
12
Multiple Choice
What is the molar mass of AuCl3?
96 g
130 g
232.5 g
303.3 g
13
Multiple Choice
Calculate the molar mass for Al(OH)3
78.0 g/mol
72 g/mol
28 g/mol
25 g/mol
14
Multiple Choice
Calculate the molar mass for Al(OH)3
78.0 g/mol
72 g/mol
28 g/mol
25 g/mol
15
Multiple Choice
What is the molar mass of CO2?
12
16
32
44
16
Multiple Choice
What is the mass of one mole of aluminum?
27 g
13 g
54 g
14 g
17
Multiple Choice
The molar mass of an element is equal to its...
Atomic mass
Atomic number
Oxidation number
Valence electrons
18
Multiple Choice
The molar mass of an element is equal to its...
Atomic mass
Atomic number
Oxidation number
Valence electrons
19

20

21
22
Multiple Choice

Find the mass of 5.82 mol of MgCl2. Round to the nearest tenth.
552.9 g
436.8 g
55.2 g
555.9 g
23
Multiple Choice

Find the mass of 5.82 mol of MgCl2. Round to the nearest tenth.
552.9 g
436.8 g
55.2 g
555.9 g
24

25

26
27
Multiple Choice
How many moles are 2.85 x 1018 atoms of iron?
4.73 x 10-6 moles
1.72 x 1042 moles
1.72 x 10-42 moles
4.73 x 106 moles
28
Multiple Choice
Which conversion factor should be used for the following question " How many molecules are there in 4.00 moles of glucose, C6H12O6?
1 mole = 78.12 g
1 mole = 22.4 L
1 mol = 6.02x 1023 particles
more than one
29

30

31

32
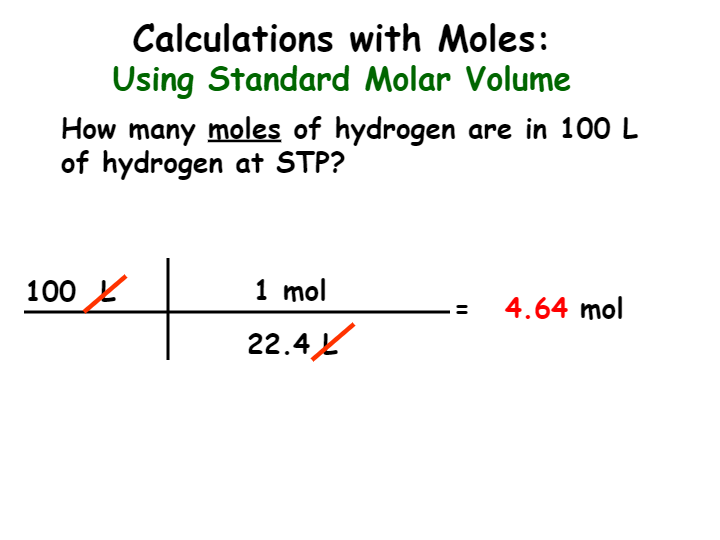
33
34
Multiple Choice

If you use of 0.672 moles of magnesium. How many liters of magnesium would you need in the graduated cylinder?
0.15L Mg
15.05L Mg
105L Mg
19,05L Mg
35
Multiple Choice

How many liters will 3.59 moles of N2 occupy?
80.4 L N2
40.8 L N2
0.408 L N2
0.804 L N2
36

37
38

39
40
Multiple Choice

Which of the following dimensional analysis setups will correctly convert 4.00x1023atoms of cobalt to moles of cobalt? How many moles of colbalt are there?
setup B : 0.664 mol Co
setup A: 2.41x1047mol Co
setup A : 0.664 mol Co
setup B: 2.41x1047mol Co
41
Multiple Choice

Calculate the number of atoms in 0.0340 g Zn.
5.20 x 10-4 atoms Zn
3.13 x 1023 atoms Zn
3.13 x 1020 atoms Zn
2.05 x 1022 atoms Zn
42
Multiple Choice
What is the mass of 1.2 x 1024 atoms of C?
2.0 grams
24 grams
0.17 grams
1.4 x 1025 grams
43
Multiple Choice

Which of the following dimensional analysis setups will correctly convert 27.76 g of Li to atoms of Li?
A
B
C
D
44
Multiple Choice
16.05 grams of methane (CH4), would occupy what volume as a gas?
160.5 Liters
1.605 Liters
It is impossible to even estimate with only this information.
22.4 Liters
Moles - Part 1
What is a mole and why do we need it?

Show answer
Auto Play
Slide 1 / 44
SLIDE
Similar Resources on Wayground

38 questions
Rhetorical Appeals and Strategies
Presentation
•
9th Grade

36 questions
Unit 2 Lesson 5: Nuclear Chemistry and Radiation
Presentation
•
10th - 12th Grade

35 questions
Naming Ionic Compounds
Presentation
•
10th - 12th Grade

44 questions
Phase Diagrams and Heating/Cooling Curves
Presentation
•
9th - 12th Grade

38 questions
Periodic Table 1
Presentation
•
9th - 12th Grade

37 questions
Classifying Chemical Reactions
Presentation
•
9th - 12th Grade

40 questions
Writing and Naming Acids
Presentation
•
9th - 12th Grade

39 questions
Spanish 1 Review: Para Empezar Part 1
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Chemistry

25 questions
Waygrounds 5/20 PS.8.1.2
Quiz
•
9th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

20 questions
Naming & Writing Chemical Formulas
Quiz
•
10th Grade

15 questions
Types of chemical reactions
Quiz
•
9th - 12th Grade

32 questions
Acids/Bases, Electrochem, Orgo
Quiz
•
9th - 12th Grade