

Moles and Molar Mass
Presentation
•
Science
•
9th Grade
•
Hard
Joseph Anderson
FREE Resource
15 Slides • 25 Questions
1

Moles and Molar
Mass
2

Moles
◦Because the number of
particles involved in most
chemical reactions is so large,
chemists use a counting unit
called the mole (mol)
◦One mole (mol) is the amount
of a substance that contains
6.02 x 1023 particles of that
substance
2
3

Molar Mass
◦The mass in grams of one mole
of a substance is called its
molar mass
◦Just as the mass of a dozen
eggs is different from the
mass of a dozen watermelons,
different substances have
different molar masses
3
4
Fill in the Blanks
Type answer...
5
Multiple Choice
602
602 million
602 moles
6.02 x 1023
6
Multiple Choice
grams
grams/mole
mole
moles/gram
7
Multiple Choice
1 mole of Li
1 mole of Au
1 mole of Si
None, all are equal
8
Multiple Choice
Find the molar mass of H2
1.01g
2.02g
1.02g
2.01g
9
Multiple Choice
Find the molar mass of CaCO3
100.09g
100g
100.2g
101g
10
Multiple Choice
Find the molar mass of NH3
17.04g
17g
14.07g
17.03g
11

Calculating
Molar Mass
Example
Calculate the molar mass of CO2.
4
12

Calculating
Molar Mass
Example 2
Calculate the molar mass of
C6H10O5.
5
13

Mole-mass
conversions
◦Given the mass of a substance,
you can use the molar mass as
a conversion factor to
calculate the number of moles
6
14
Multiple Choice
Find the number of moles in 50.0g of NO2
2.50
1.08
50.0
3.08
15
Multiple Choice
Find the number of moles in 24 grams of NaHCO3 (round to nearest thousandths)
.286
.285
.226
.326
16

Mole-mass
conversions
Example 2
What is the mass of 0.2020 mol of
NO2?
8
17
Multiple Choice
What is the mass of 0.2020 mol of NO2?
10.454
4
9.292
8.10
18
Multiple Choice
what is the mass of 1.61 of H2O
28.98
82.12
22.981
3
19

Chemical
Reactions and
Energy
20

Energy
Exchanges
◦All chemical reactions release
or absorb energy!!!
◦This energy can take many
forms such as thermal energy,
light, sound, or electricity
10
21
Fill in the Blanks
Type answer...
22

More
Energy Out
◦Many reactions involve the
release of energy
◦Exergonic reactions: chemical
reactions that release energy
11
23

More
energy out
as thermal
energy
◦In many reactions, the energy
given off is thermal energy.
◦Exothermic reaction: when
the energy given off is primarily
in the form of thermal energy
◦Ex. wood burning and the
explosion of dynamite are
exothermic reactions
12
24
Multiple Choice

_____ reactions usually feel hot!
endothermic
exothermic
25
Multiple Choice
taken in
given out
26
Multiple Choice
Stay the same
Increase
Decrease
27

More
energy in
◦Sometimes a chemical reaction
requires more energy to break
bonds than is released
◦Endergonic reactions:
chemical reactions that absorb
energy
13
28
Multiple Choice
If a chemical reaction absorbs light in a reaction, it is an
exergonic reaction
exothermic reaction
endergonic reaction
endothermic reaction
29

More
energy in
as thermal
energy
◦When the energy needed to
keep a reaction going is in the
form of thermal energy, the
reaction is called an
endothermic reaction
◦Ex. Baking bread, cooking an
egg, evaporating water
14
30
Multiple Choice

taken in or absorbed
given out or released
31
Multiple Choice

A student mixed two chemicals to allow them to react. The temperature before the reaction was 25 ° C. The temperature after the reaction was 18° C. Which of the following is true?
The temperature increased
It is an endothermic reaction
It is an exothermic reaction
32
Multiple Choice
Warm
Cold
33
Multiple Choice
In an endothermic reaction, heat is ...
taken in
given out
34

Endothermic
vs.
Exothermic
15
35

Conservation
of energy in
chemical
reactions
◦Recall, energy can change from
one form to another, but the
total amount of energy never
changes
◦The total amount of energy
before and after the reaction
remains the same
16
Chemical energy of reactants = chemical energy of products + energy released
36
Multiple Choice

Endothermic processes
Exothermic processess
None of these
37
Multiple Choice

Exothermic
Isothermic
Endothermic
None of the above
38
Multiple Choice
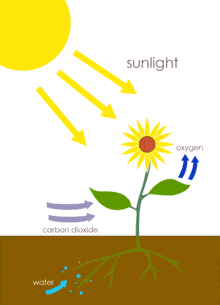
Endothermic
Exothermic
39
Multiple Choice

What type of reaction occurs in a hand warmer?
exothermic
endothermic
40
Multiple Choice

It is an exothermic reaction
It is an endothermic reaction
It is made of ice
It is dissolving your mouth

Moles and Molar
Mass
Show answer
Auto Play
Slide 1 / 40
SLIDE
Similar Resources on Wayground

31 questions
Determining Half-Life Lesson
Presentation
•
9th Grade

34 questions
Carbon, Nitrogen, and Oxygen Cycles
Presentation
•
8th Grade

33 questions
Atoms and the periodic table
Presentation
•
8th Grade

38 questions
NINE WEEK REVIEW
Presentation
•
9th Grade

34 questions
Review
Presentation
•
9th Grade

36 questions
Atomic theory and the periodic table
Presentation
•
9th Grade

34 questions
Describing Matter
Presentation
•
9th Grade
Popular Resources on Wayground

20 questions
STAAR Review Quiz #3
Quiz
•
8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

6 questions
Marshmallow Farm Quiz
Quiz
•
2nd - 5th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for Science

46 questions
Unit 4 Geosphere Test Review
Quiz
•
9th - 12th Grade

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

16 questions
Ecosystem Stability
Quiz
•
9th Grade

20 questions
Mendelian Genetics Review
Quiz
•
9th Grade

12 questions
Unit 6 Quiz #3 (Life Cycles of Stars)
Quiz
•
9th Grade

34 questions
Human Impact on Ecosystems Quiz
Quiz
•
9th Grade

10 questions
Exploring Alfred Wegener's Continental Drift Theory
Interactive video
•
6th - 10th Grade