

7-Physical Science Review-TCAP
Presentation
•
Engineering
•
6th - 8th Grade
•
Medium
Jessica Freeman
Used 2+ times
FREE Resource
37 Slides • 279 Questions
1
7th Grade
TCAP Review
Physical Science
By Jessica Freeman
2
Multiple Choice

element
compound
mixture of elements
mixture of compounds
mixture of elements and compounds
3
Multiple Choice
Water is made up of hydrogen and oxygen, two elements that were chemically combined. This makes water a(n)
atom
compound
element
mixture
4
Multiple Select
[check all that apply] Pure substances include:
Elements
Homogeneous Mixtures
Compounds
Heterogeneous Mixtures
5
Multiple Choice

What is the model scientists refer to when looking at the structure of an atom?
The electron cloud model
The atom model
The structure of our world model
The theory model
6
THe Boher model of an Atom
Electrons are located in electron orbits, called valence shells.
Electrons have a negative charge.
Protons are located in the nucleus and have a positive charge.
Neutrons are also located in the nucleus, they have a neutral charge

7
Multiple Choice
The center of an atom has
both electrons and protons
a negative charge
electrons
a nucleus
8
Multiple Choice
Electrons have this type of charge
positive
neutral
negative
9
Multiple Choice
Particles in an atom that have a positive charge are
nuetrons
electrons
protons
ion
10
Multiple Choice
Where are elctrons in an atom located
in the nucleus
in the outer shells
everywhere
11
Multiple Choice
Protons and neutrons are located
in the nucleus
in the outer shells
everywhere
12
Elements on the periodic table have a neutral charge.
Neutral atoms have equal numbers of protons and electrons.
Ions of an atom, are atoms with one more electron than its number of protons. Ions have a negative charge.
Cations of an atom, are atoms with one less electron than its number of protons. Cations have a positive charge
13
Multiple Choice
A neutral atom has
Equall amounts of protons and electrons
Equall amounts of protons and neutrons
Equall amounts of electrons and neutrons
14
Multiple Choice
Ions are atoms with one more electron than protons and will have a
positive charge
negative charge
neutral charge
15
Multiple Choice
Cations are atoms with one less electron than protons and will have a
positive charge
Negative charge
neutral charge
16
Changing the number of protons in an atom, changes the type of element

Notice Hydrogen has 1 proton
and Helium has 2, andLithium has 3.
The number of protons in an atom is its atomic mass
The number of protons and neutrons an atom has is its Atomic mass or weight.
17
Multiple Choice
Adding a proton to the nucleus of an atom changes
the charge of the atom
The stability of the atom
The type of element
18
Multiple Choice
The atomic number of an atom is its
number of protons
number of electrons
numbers of neutrons
number of both its protons and its neutrons
19
Multiple Choice
The atomic mass of an atom is its
number of protons
number of electrons
numbers of neutrons
number of both its protons and its neutrons
20
Multiple Choice
Challenge question True or false:
Unstable atoms have more neutrons than protons in its nucleus
True
False
sometimes
21
Multiple Choice
An atom of oxygen has a -2 charge. How many electrons does it have?
2
8
6
10
22
Multiple Choice
An atom has a negative charge. Which of the following MUST be true?
it has the same number of protons and neutrons
it has more protons than electrons
it has more electrons than protons
it has more neutrons than protons
23
Multiple Select
Select all of the following that affects mass number.
protons
neutrons
electrons
how common they are in the world
24
Multiple Choice
If two atoms are different elements, they must have...
different mass numbers
different numbers of protons
different charges
different colors
25
Multiple Choice
The atomic number is...
the number of protons + neutrons
the number of protons - electrons
the number of protons
the imbalance of protons and electrons
26
Multiple Choice

Particles in an atom that are positively charged
neutron
proton
atom
electron
27

The Periodic Table
Vocabulary and Practice Questions
28
Valance Electrons & Reactivity
I can explain that the valence electrons describe the reactivity of the element. 8.5(B)
29
state of matter and properties of matter

30
Multiple Choice
Matter is made up of tiny particles called_
particles
molecules
atoms
cells
31
Multiple Choice
A property that can be observed WITHOUT changing the chemical composition of an object
chemical property
physical property
32
Multiple Choice

Is this a chemical or physical change?
Physical Change
Chemical Change
33
Multiple Choice

Anthony is performing an experiment. He adds 0.25 grams of baking soda to 5 grams of water and stirs. Anthony watches as the baking soda dissolves in the water. If Anthony weighs the water and baking soda solution, what will its mass be?
4.75 grams
5.25 grams
5.0 grams
7.5 grams
34
Multiple Choice
If a reaction starts with a total 54g of reactants it should produce
a total of 54g of products
35
Multiple Choice
All solids have a closely packed, fixed arrangement of particles.
True
False
36
Multiple Choice
37
Multiple Choice

Charles's Law says that when the temperature of a gas rises (increases), its
volume also increases
mass increases
volume decreases
volume stays the same
38
Multiple Choice
The way molecules in a substance move around relative to each other
a. kinetic energy
b. freedom of movement
c. potential energy
d. freedom of speech
39
Multiple Choice
when matter changes from a solid to a gas and skips the liquid phase
sublimation
depostion
endothermic
exothermic
40
Multiple Choice
What happens in a chemical reaction?
Bonds between atoms break and/or are formed
New atoms are created and/or removed
Protons move from one atom to another
Electrons change into protons
41
Types of Chemical Reactions
physical science

42
Multiple Choice

element
compound
mixture of elements
mixture of compounds
mixture of elements and compounds
43

44
Multiple Choice
How are chemical bonds formed?
Atoms duplicate themselves
Electrons move from one atom to another
The number of protons and electrons in an element is changed
Through changes in an atom's nucleus
45
Multiple Choice
What reaction has the following general formula:
A + B --> AB
Synthesis
Decomposition
Single Displacement
Double Displacement
Combustion
46
Multiple Choice
when frozen carbon dioxide goes from a solid to a gas
freezing
melting
vaporization
frost
47
Multiple Choice
What type of reaction is the following:
2H2+O2−>2H2O
Synthesis
Decomposition
Single Displacement
Double Displacement
Combustion
48
Multiple Choice
Which of the following would indicate that a chemical reaction has occurred?
A new element has been created
A different substance has formed
The total mass has either increased OR decreased
The total number of elements has either increased OR decreased
49

50
Multiple Choice
How is particle motion related to temperature.
a. if the particles in two pure substances have the same average speed, the substances must have the same average speed
b. all the particles inside a substance are moving at the same speed, and that speed determines the same temperature
c. how hot or cold a substance is depends on how fast the particles are moving
d. the total kinetic energy of the particles in a substance determines the temperature of the substance
51
Multiple Choice
What is the general reaction scheme for a decomposition reaction?
A + B --> AB
AB --> A + B
A + CD --> C + AD
AB + CD --> CB + AD
CxHy+O2 −> CO2+H2O
52
Multiple Choice

These are all models of the same molecule. What would this molecule look like in a chemical/molecular formula?
CH
C4H
CH4
4CH
53
Multiple Choice
2 NO2 --> 2 O2 + N2
Synthesis
Decomposition
Single displacement
Double displacement
54
Multiple Choice
these particles are shaped, rigged, compacted, vibrate back and forth
liquid
gas
solid
55

56
Multiple Choice

Which would indicate a chemical equation is balanced?
If the number of atoms in the reactants is the same as the number of atoms in the products
If the atoms have ionic OR covalent bonds
If the atoms have full valence shells
If the total energy has remained the same
57
Multiple Choice
What reaction has the following general formula:
CxHy + O2 −>CO2 + H2O
Combination
Decomposition
Single Displacement
Double Displacement
Combustion
58
Multiple Choice

When particles of a gas are heated
they speed up and collide with the walls of their container
slow down and won't collide with the walls of their container
there will be no change
59
Multiple Choice
What type of reaction is the following:
2C4H10 +13O2 −> 8CO2+10H2O
Combination
Decomposition
Single Displacement
Double Displacement
Combustion
60
Multiple Choice

BEFORE the equation is balanced, how many Hydrogen atoms are present on the reactant side?
6
2
12
8
61

62
Multiple Choice
these particles have an indifferent shape, and definite volume
solid
liguid
gas
63
Multiple Choice
What reaction has the following general formula:
A + CD --> C + AD
Combination
Decomposition
Single Displacement
Double Displacement
Combustion
64
Multiple Choice

Why does the number of Carbon atoms need to be the same on both sides of the reaction?
In a chemical reaction, no new atoms can be made
Because the number of Oxygen atoms must be balanced with the number of Carbon and Hydrogen atoms
Because the coefficients and the subscripts need to be equal
It does NOT need to be the same
65
Multiple Choice
What type of reaction is the following:
3CuS +2Al −> Al2S3+3Cu
Combination
Decomposition
Single Displacement
Double Displacement
Combustion
66
Multiple Select
Temperature is a measure of kinetic energy. Every substance is made up of particles. The temperature of a substance is related to the kinetic energies of these particles. If a substance is hot, its particles tend to have a large amount of kinetic energy.
The particles in a substance have kinetic energy because they are moving. The particles are constantly in motion, even if the substance does not appear to be moving.
Select all of the statements that are true about the rubber ball shown above.
a. if the ball is cold, its particles tend to have a small amount of kinetic energy
b. the particles that make up the ball are moving even if the ball is not moving
c. the temperature of the ball depends on the kinetic energies of the particles that make up the ball
67

68
Multiple Choice

What coefficient should go in the last blank space in order for the equation to be balanced?
1
2
3
4
69
Multiple Choice
What reaction has the following general formula:
AB + CD --> CB + AD
Combination
Decomposition
Single Displacement
Double Displacement
Combustion
70
Multiple Choice
these particles have indefinite shape, indefinite volume. and expans
solid
liquid
gas
71
Multiple Choice
72
Multiple Choice

How many sulfur atoms are present in this molecule?
4
3
12
1
73
Multiple Choice
74
Multiple Select

In the molecule below, which of the following choices is a subscript? (Select all that apply)
3
2
4
SO
75
Multiple Choice
going from a liquid to a solid is called
melting
condensation
freezing
vaporization
76
Multiple Choice
What does the Law of Conservation of Mass state?
No new substances can be made or destroyed
No new compounds can be made or destroyed
No new atoms can be made or destroyed
Mass cannot be lost from a physical reaction
77
Multiple Choice
Lily is doing a science experiment with a substance in a sealed jar. At first, the substance’s molecules are moving around each other. What will happen to Lily’s substance if she transfers energy into the substance and causes a phase change?
After the phase change, the substance’s molecules will move . . .
a. faster, and the substance will be a liquid
b. faster, and the substance will be a gas
c. slower, and the substance will be a solid
d. slower, and the substance will be a liquid.
78
Multiple Choice
Which of the following follows the Law of Conservation of Mass?
CH₄ + 2 O₂ → 2CO₂ + 2 H₂O
2 CH₄ + 2 O₂ → CO₂ + 4 H₂O
CH₄ + 2 O₂ → CO₂ + 2 H₂O
All the above
79
Multiple Choice
going from a gas to a solid and skips the liquid phase is called
deposition
sublimination
endothermic
80
Multiple Choice
In a chemical reaction, ________________________ are changed into _______________________.
Reactants; Products
Electrons; Molecules/Compounds
Coefficients; Subscripts
Original Elements; New Elements
81
Multiple Choice

when the speed of particles of gas in a container increases,
the pressure decreases
the pressure increases
there will be no change in the pressure inside the container
82
Multiple Choice
Bacteria in milk causes it to curdle over time. What type of change is this?
Chemical
Physical
Both
Neither
83
Multiple Choice
when heat is exiting the system
endothermic
exothermic
sublimination
condensation
84
Multiple Choice
Which type of reaction pulls in thermal energy from the environment around it?
Endothermic
Exothermic
85
Multiple Choice
Temperature is a measure of the average kinetic energy (represented here by speed) of molecules in a substance
a. True
b. False
86
Multiple Choice
When building a HEATING pack, what type of reaction is it?
Exothermic
Endothermic
87
Multiple Choice
when heat is entering the system
endothermic
exothermic
sublimination
deposition
88
Multiple Choice

Moo like a cow for 2 seconds.
Did it!
No Way!
89
Multiple Select
The particles in liquids and gases are free to move while the particles in a solid have a fixed arrangement. This is why _____ take the shape of their containers while solids do not.
liquids
gases
solids
colors
90
Multiple Choice
these particles have atoms that lose their electrons. the mixture of electrons and nuclei that results is this
solid
liquid
gas
plasma
91
Multiple Choice
When a substance does not appear to be moving, the molecules of a substance are NOT moving also.
a. True
b. False
92
Multiple Choice
the particles in a solid are rigid and do not move
true
false
93
Multiple Choice

The pressure of a gas in increased, so the
volume will increase
volume will stay the same
volume will decrease
94
Multiple Choice
a liquid does not have a shape of its own
true
false
95
Multiple Choice
Anthony measured the temperature of a pond near his house. Before he left for school, the water in the pond was 18°C. When he came home from school, the temperature of the pond was higher than it was in the morning.
What happened to the water molecules while he was at school?
a. The molecules got closer together
b. The molecules started moving faster.
c. The water lost cold molecules.
d. The molecules became larger
96
Multiple Choice
decreasing heat energy can cause a phase change
true
false
97
Multiple Choice
98
Multiple Choice
decreasing heat energy can cause a phase change
true
false
99
Multiple Choice
What happens to molecules when their kinetic energy decreases?
a. They move slower
b. They move faster
c. They get heavier
d. It depends on the type of molecules
100
Multiple Choice
increasing heat energy can cause a phase change
true
false
101
Multiple Choice

If heat is added to the air in a sealed container, the air pressure will
stay the same
go up (increase)
go down (decrease)
102
Multiple Choice
the evaporation of water over time is an example of sublimation
true
false
103
Multiple Choice
Jamil’s bicycle is made of aluminum. When are the aluminum molecules in his bicycle moving?
The aluminum molecules are . . .
a. only moving when Jamil is riding his bicycle.
b. only moving when Jamil is not riding his bicycle
c. always moving
d. never moving
104
Multiple Choice
placing a balloon in a freezer will cause the balloon to expand
true
false
105
Multiple Choice
O + H2 → H2O
32 g of Oxygen completely reacts with 8 g of Hydrogen to produce _____ g of water.
8
12
24
40
106
Multiple Choice
this is anything that has a mass and takes up space by having volume. it is made up of particles which can be atoms, molecules, or electrically charged particles called ions
mass
volume
matter
velocity
107
Multiple Choice
Scientists put a sample of water into a sealed tank. Water can be a solid, liquid, or gas. At first, the water was a liquid. Then, the scientists transferred energy out of the substance, and the water changed phase. What happened to the movement of the water molecules?
After the phase change, the water molecules moved . . .
a. faster and started moving away from each other.
b. faster and started moving around each other.
c. slower and started moving in place.
d. slower and started moving around each other.
108
Multiple Choice

When using a bicycle pump, the air pressure within the pump is ___________ as the handle is pushed down. This is compression.
remains the same
increases (goes up)
decreases (goes down)
109
Multiple Choice

110
Multiple Choice

Describes the relationship between a gas's temperature and volume when its pressure remains constant
Charles's Law
Boyles Law
The Law of Thermodynamics
111
Multiple Choice
112
Multiple Choice
In the wintertime, the air pressure in the tires on a car can decrease. This is because the temperature outside has _____________
decreased (went down)
increased (went up)
stayed the same
113
Multiple Choice

Which law?
Boyle
Charles
Landis
Boykin
114
Multiple Choice

What state of matter undergoes changes in volume most easily? (It can be most easily compressed)
solid
liquid
gas
115
Multiple Choice
Matter can not be created nor destroyed: it can only be
Destroyed a little bit
Invisible
Transformed, changed
None of the above
116
Multiple Choice

the amount of space that matter fills up
solid
liquid
volume
gas
117
Multiple Choice

Which law?
Charles
Boyle
Diegel
Mitchell
118
Multiple Choice

This happens when particles gain enough energy to move independently. This results in either evaporation or boiling
vaporization
condensation
evaporation
freezing
119
Multiple Choice
120
Multiple Choice
121
Multiple Choice
Mg + F → MgF
24 g of magnesium reacts with 38 g of fluorine to produce _____ g magnesium fluoride.
122
Multiple Choice
123
Multiple Choice
124
Multiple Choice
125
Multiple Choice

What evidence supports that this is a PHYSICAL change?
It creates a new substance
It changes the state of matter to a liquid
It is started on fire
126
Multiple Choice
127
Multiple Choice
128
Multiple Choice
129
Multiple Choice
How many elements does PbNO7 have?
Hint: Every capital letter😉
130
Multiple Choice
131
Multiple Choice
What state of matter are the particles moving the fastest and are farthest apart?
Solid
Liquid
Gas
132
Multiple Choice

133
Multiple Choice
If reaction starts with 20g of reactants it should produce
a total of 40g of products
a total of 80 g of products
a total of 10g of products
a total of 20g of products
134
Multiple Choice

135
Multiple Choice
In this state of matter the particles "slip past" each other and take the shape of it's container.
Solid
Gas
Liquid
136
Multiple Choice

137
Multiple Choice
138
Multiple Choice

139
Multiple Choice
140
Multiple Choice
Which of the following represents the amount of space and object takes up?
density
volume
mass
141
Multiple Choice

Which state of matter has particles that are somewhat close together and have enough kinetic energy to slide past each other?
solid
liquid
gas
142
Multiple Choice
Substances present at the start of a chemical reaction are called ____.
143
Multiple Choice
144
Multiple Choice

Which of the models of a chemical reaction best represents the Law of Conservation of Mass?
A
B
C
D
145
Multiple Choice
Which of the following is NOT a part of the Kinetic Theory of Matter?
All matter is made up of tiny particles.
Particles are always moving in random paths
Occasionally, particles stop moving.
The type of movement of particles depends on the amount of energy.
146
Multiple Choice
Substances that form as the result, or at the end, of a chemical reaction are called ____.
147
Multiple Choice
Which of the following states of matter has the LOWEST level of energy?
Solid
Liquid
Gas
Plasma
148
Multiple Choice

Is this a chemical or physical change?
Physical Change
Chemical Change
149
Multiple Choice
The energy of an object as it is in motion is defined as
Potential Energy
Elastic Energy
Electric Energy
Kinetic Energy
150
Multiple Choice
How many iron (Fe) atoms are in
Fe(NO3)2
151
Multiple Choice

What state of matter is shown in the image?
Solid
Liquid
Gas
Thermal Energy
152
Multiple Choice

Which of the models of a chemical reaction best represents the Law of Conservation of Mass?
A
B
C
D
153
Multiple Choice

All matter is made of _____ that are constantly moving.
insulators
particles
convection
pieces of plastic
154
Multiple Choice
How many nitrogen (N) atoms are in
Fe(NO3)2
155

156
Multiple Choice
How many oxygen (O) atoms are in
Fe(NO3)2
157
Multiple Select
Which of the following is an open system?
A
B
C
D
158
Multiple Choice
Transformed, rearranged
159
Multiple Choice

What evidence supports that this is a CHEMICAL change when the white stuff is set on fire?
It creates a new substance of ash
It changes the state of matter to a solid
The product has the same properties as the reactant
160
Labelling
Label the reactants and products of the chemical reaction shown.
Products
Reactants
161
Multiple Select
Which of the following is close system?
A
B
C
D
162
Labelling
Label the reactants and products in the chemical equation shown.
Products
Reactants
163
Multiple Choice
What is the definition of conductivity?
ability of a material to resist breaking
how reflective (shiny) a surface is
ability to complete an electric circuit
ability to react to forces exerted by magnets
164
Multiple Choice

What are the reactants in this chemical equation?
6 CO2 + 6 H2O
H2O
C6H12O6
C6H12O6 + O2
165
Multiple Choice

Which of the models of a chemical reaction best represents the Law of Conservation of Mass?
A
B
C
D
166
Multiple Choice

What are the products in this chemical equation?
CO2 + H2O
H2O
C6H12O6
C6H12O6 + O2
167
Multiple Choice

What are the reactants in this animation?
The squiggly ash that comes off the top
The white powder on the sand
The smoke that is created
168
Multiple Choice

Select the chemical formula for this molecule?
B1F3
BF
BF3
FB
169
Multiple Choice
Q.
True or False
The mass of the reactants in a chemical equation must equal the mass of the products.
TRUE
FALSE
170
Multiple Choice
How many atoms of hydrogen are in
C6H12O6
6
12
18
2
171
For example- There are 3 Valance Electrons in Aluminum, Al, shown in the image ->
Valance Electrons are on the outmost energy level

172
Dropdown

The experiment shows a
The mass at the end of the experiment will be
173
Multiple Choice
A student heated a 10 gram sample of a chemical in an open container. A chemical reaction occurred, and the mass of the sample was measured again. The mass of the sample was found to be less than before the reaction. Which of the following best explains the decrease in mass of the sample?
The heat caused the chemical to become less dense.
Some of the lighter particles were destroyed.
The reaction gave off more heat than was added.
Some of the particles escaped as a gas formed.
174
Multiple Choice

How many grams of Zn(NO3)2 was produced?
3 g
4 g
0 g
5 g
175
Multiple Choice

Is this a chemical or physical change?
Its filling up with a bubbling blue liquid!
Physical Change
Chemical Change
176
Multiple Choice

If AB is 18 grams and reacts in a closed system, which of the following could be the grams of A and B?
A is 13 grams and B is 2 grams
A is 18 grams and B is 5 grams
A is 9 grams and B is 11 grams
177
Multiple Choice
A chemical reaction that releases energy _________ and a chemical reaction that absorbs energy is called _____________.
exothermic; endothermic
endothermic; exothermic
exothermic; accumulative
liberal; endothermic
178
Multiple Choice
Which of the following is NOT a physical property?
flammability
melting point
density
luster
179
Multiple Choice
A system where only energy can be exchanged but not matter
Close system
Open system
180
Multiple Select

What evidence supports that this is a CHEMICAL change? There is more than 1 answer
It creates a new substance of blue liquid
It changes the state of matter to a liquid
It creates a new gas that is bubbling out of the liquid
181
Multiple Choice
A system which can be exchange both matter and energy with the surrounding.
Close system
Open system
182

183
Multiple Choice
Q.
True or False
In a chemical reaction there is always ONE product.
TRUE
FALSE
184
Multiple Choice

Is this a chemical or physical change?
Physical Change
Chemical Change
185
Multiple Choice
In combustion reaction what element is always the reactant
Oxygen
Carbon
Gold
Helium
186
Multiple Choice
A property that can only be observed when a substance reacts or does not react with another substance
chemical property
physical property
187
Multiple Choice

What evidence supports that this is a PHYSICAL change?
It creates a new substance
It changes its shape
It is started on fire
188

Use your own paper
Write each term and the definition.
Do NOT shorten the definitions.
Vocabulary
189
Multiple Choice

Are these chemical or physical changes?
Physical Change
Chemical Change
190
Fill in the Blanks
Type answer...
191
Multiple Choice

What evidence supports that this is a CHEMICAL change?
They are the same matter, are not a gas
Burning stuff is always a chemical change!
The product has the same properties as the reactant
192

193
Multiple Choice

Is this a chemical or physical change?
Physical Change
Chemical Change
194
Multiple Choice
Which of the following represents the amount of matter in an object?
density
mass
volume
195
Multiple Choice

What evidence supports that this is a PHYSICAL change?
It creates a new substance
It changes its shape
It changes the state of matter
196
Multiple Choice

How many valance electrons does Te have?
52
127
6
5
197
Multiple Choice

Is this a chemical or physical change?
Physical Change
Chemical Change
198
Multiple Choice
The temperature at which a liquid turns into a gas
melting poing
boiling point
199
Multiple Choice

What evidence supports that this is a PHYSICAL change?
It creates a new substance
It changes its shape
It changes the state of matter
200

201
Multiple Choice

Are these chemical or physical changes?
Physical Change
Chemical Change
202
Multiple Choice
If you place a thermometer into a glass of boiling water, what temperature should the thermometer read in degrees Celsius?
0
32
100
212
203
Multiple Choice

What evidence supports that this is a CHEMICAL change?
A gas is created
Burning stuff is always a chemical change!
Its still soda, its just in the pool
204
Multiple Choice

particles in an atom that are negatively charge
electron
proton
neutron
nucleus
205
Multiple Choice

What material from is the reactant in this chemical reaction
the Soda and Mento candy
The boy
the foam
The pool
206
Multiple Choice
At what temperature does water freeze?
0ᵒC or 32ᵒF
0ᵒF or 32ᵒC
100ᵒC or 212ᵒF
100ᵒF or 212ᵒC
207

208
Multiple Choice
Which of the following is NOT an example of a chemical reaction?
flammability
oxidation
reactivity
melting point
209
Reactivity - A measure of how much a substance reacts when it is mixed with another substance
Reactivity of an element is determined by the number of Valance Electrons.
210
Multiple Choice
Which of the following is defined as the likelihood of a substance to undergo a chemical reaction?
oxidation
rusting
reactivity
flammability
211
Fill in the Blanks
Type answer...
212
Multiple Choice
Which famous US landmark is known for its oxidation?
Mount Rushmore
Eiffel Tower
Disney World
Statue of Liberty
213

THEN: Answer the questions on the following slides. Refer to this reading and your vocabulary.
Read This
214
Multiple Choice
What is the formula for density?
volume/mass
volume x mass
mass/volume
215
Fill in the Blanks
Type answer...
216
Multiple Choice
Which of the following is NOT a unit of measure for volume?
grams (g)
milliliters (mL)
cubed centimeters (cm³)
217
Multiple Choice
Reactivity of an element is determined by the number of ________.
Protons
Valance Electrons
Neutrons
Subatomic Particles
218
Fill in the Blanks
Type answer...
219
Multiple Choice
An element is?
Chemically combined
Pure substances that can be broken down
Pure substances that cannot be broken down into simpler substances
One or more elements chemically combined.
220
Fill in the Blanks
Type answer...
221
Have a FULL outer shell
😃Non-Reactive/Stable Elements
DO NOT have a full outer shell
😡Reactive/Unstable Elements
Reactive vs Non-Reactive


222
Multiple Select
The weighted average of the atomic masses of the naturally occurring isotopes of an element.
average atomic mass
atomic number
number of isotopes
mass number
223
Multiple Choice
_____ elements have a full outer shell of valance electrons?
Reactive or unstable
Non-reactive or stable
224
Multiple Choice

Neutron
particles in an atom that has no charge
particles in an atom that is negatively charge
particles in an atom that is positively charge
225
Multiple Choice
_____ elements that DO NOT have a full outer shell of valance electrons?
Reactive or unstable
Non-reactive or stable
226
Multiple Choice
A one, two, or three letter representation of the name of an element.
atomic number
chemical formula
chemical symbol
periodic table
227


Outer shells are almost full or almost empty.
Lithium only has 1 VE- Highly Reactive
Chlorine has 7 VE- Highly Reactive
Highly Reactive Elements
Outer shells are about half way full
Boron has 3 VE- Less Reactive
Carbon has 4 VE- Less Reactive
Less Reactive


228
Multiple Choice
__ is a pure substance containing two or more kinds of atoms.
Element
compound
mixture
solution
229
*All of the Elements in Group 8A are Stable or nonreactive
Reactivity is determined by number of Valance Electrons

230
Multiple Choice
An arrangement of the elements in order of their atomic numbers so that elements with similar properties fall in the same column, or group.
group
chemical symbol
periodic table
malleable
231
Multiple Choice
Neon, Ne, is in Group 8a. Is Ne stable or reactive? Why?
Ne is stable because it the last energy level is full.
Ne is reactive because it the last energy level is full.
Ne is stable because it the last energy level is almost full.
Ne is reactive because it the last energy level is almost full.
232

233
The elements in Groups 1, 2, 6 & 7 are highly reactive because the outer shells are almost full
Reactivity is determined by number of Valance Electrons

234
Multiple Choice
In chemistry, a horizontal row of elements in the periodic table.
metal
period
metalloid
group
235
Multiple Choice
Which group is more reactive 6a or 7a? Why?
6a because it is closer to having a full shell
7a because it is closer to having a full shell
6a because it has a full outer shell
7a because it has a full outer shell
236
Multiple Choice
The properties of a compound are different from the elements that make them up.
True
False
237
The elements in Groups 3,4 & 5 are less reactive because the outer shells are about halfway full
Reactivity is determined by number of Valance Electrons

238
Multiple Choice
Russian chemist who developed a periodic table of the chemical elements and predicted the discovery of several new elements.
Demitri Mendeleev
Democritus
JJ Thompson
Vladimir Putin
239
Multiple Choice
Which is the least reactive group?
6a
4a
1a
240
Multiple Choice
Electrons in the outermost orbital of an atom are called
Ions
Covalen bonds
Ionic bonds
Valence
241
Lets compare:
Which element is more reactive, Oxygen (O) or Aluminum (Al)?
Why?
Reactivity is determined by number of Valance Electrons

242
Multiple Choice
An element that conducts heat and electricity poorly.
metal
metalloid
nonmetal
malleable
243
Lets compare:
Which element is more reactive, Oxygen (O) or Aluminum (Al)?
Why?
Because it is closer to having a full outer shell
Reactivity is determined by number of Valance Electrons

244
Multiple Choice

element
compound
mixture of elements
mixture of compounds
mixture of elements and compounds
245
Multiple Choice
Use your Periodic Table. Which element is stable or non-reactive?
Carbon (C)
Krypton (Kr)
Sodium (Na)
246
Multiple Choice
An electron that is found in the outermost shell of an atom and that determines the atom's chemical properties.
proton
atom
valence electron
cation
247
Multiple Choice
Use your Periodic Table. Which element is highly reactive?
Carbon (C)
Krypton (Kr)
Sodium (Na)
248
Multiple Choice
A positive charge will attract a ____________ charge
Positive
Negative
Neutral
249
Multiple Choice
Use your Periodic Table. Which element is least reactive?
Carbon (C)
Krypton (Kr)
Sodium (Na)
250
Fill in the Blanks
251
Valance Electrons & Reactivity
Add the reactivity notes to your Periodic Table

252
Multiple Choice

Oxygen is a
Element
Compound
253
Fill in the Blanks
254

255
Fill in the Blanks
256
Multiple Choice
An element is a ___ that cannot be separated into simpler substances by physical or chemical means.
pure substance
mixture
solution
compound
257
Fill in the Blanks
258
Multiple Choice
All atoms are most stable with (or would "prefer") how many electrons in their valence shell?
1
2
8
18
259
Fill in the Blanks
260
Multiple Choice
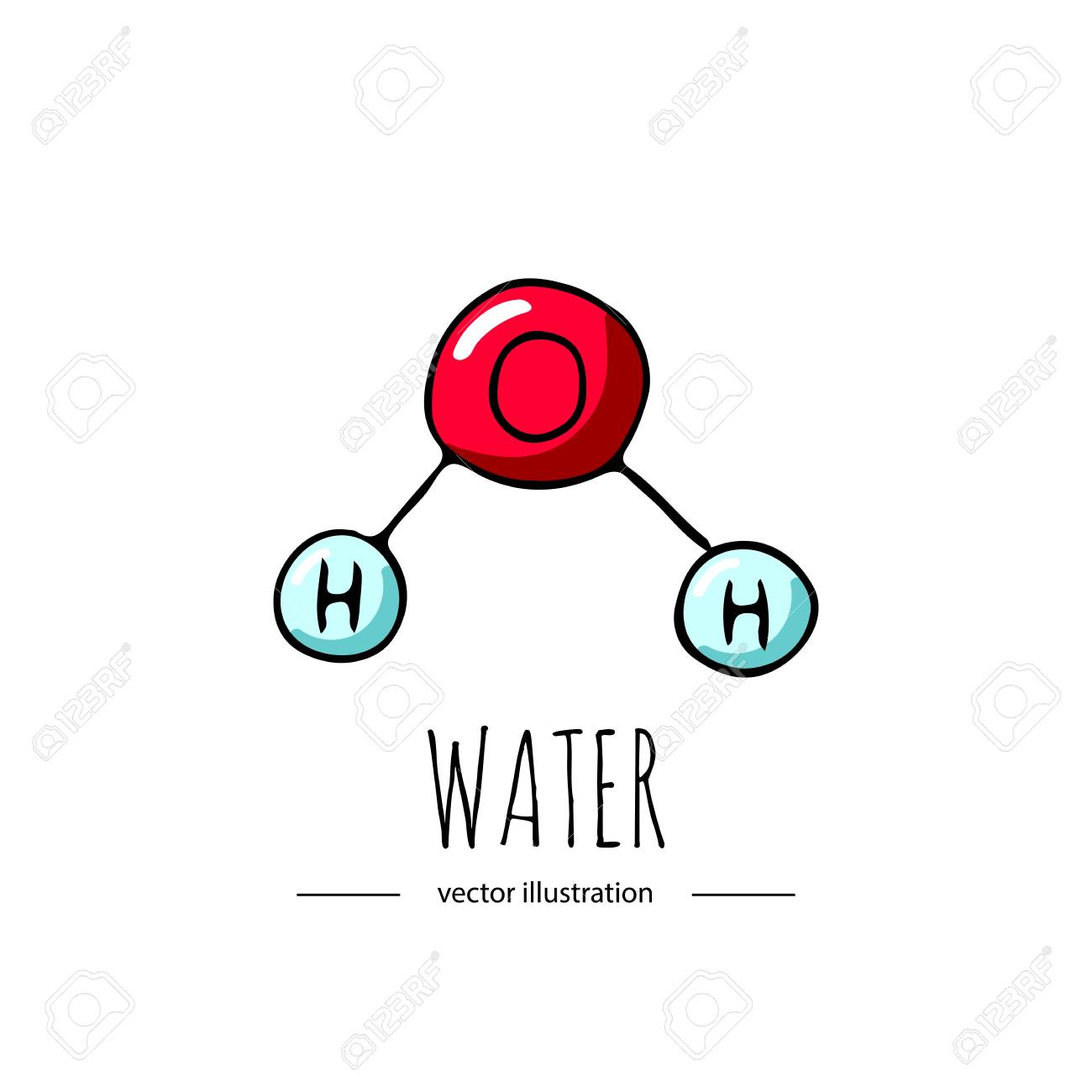
Water (H2O) is a
Element
Compound
261
Fill in the Blanks
262
Multiple Choice

neutrons
protons
atomic mass
neutrons + protons
263
Multiple Choice

element
compound
mixture of elements
mixture of compounds
mixture of elements and compounds
264

265
Multiple Select
[check all that apply] Mixtures are
chemically combined
composed of two or more types of matter that can be present in varying amounts
One or more elements chemically combined
can be separated by physical changes
266
Multiple Choice
A charged atom
Ion
Isotope
Proton
Electron
267
Multiple Choice

Which of the diagrams below represent a mixture?
A
B
C
D
268

269
Multiple Choice

Salt (NaCl) is
Mixture
Compound
270
Multiple Choice
If there are more protons than electrons, the
atom is ______ charged.
positively
negatively
neutral
271
Multiple Choice

element
compound
mixture of elements
mixture of compounds
mixture of elements and compounds
272
Multiple Choice
If there are more protons than electrons, the
atom is ______ charged.
positively
negatively
neutral
273
Multiple Choice

Saltwater is
Mixture
Compound
274
Multiple Choice
If there are more electrons than protons, the atom is _______ charged.
positively
negatively
neutral
275
Multiple Choice

How many different types of atoms are there in the following molecule?
7
6
3
8
276
Multiple Choice
Bonds form because atoms are seeking
Help
Relationships
Stability
Reactions
277
Multiple Choice

Chicken noodle soup is
Homogeneous mixture
Heterogeneous mixture
278
Multiple Choice
Ionic bonds
Share electrons
Trade electrons
Borrow electrons
Transfer electrons
279
Multiple Choice

element
compound
mixture of elements
mixture of compounds
mixture of elements and compounds
280
Multiple Choice
Covalent Bonds
Share electrons
Trade electrons
Borrow electrons
Transfer electrons
281
Multiple Choice

A bottle of dishwashing detergent contains a clear, viscous liquid. The label has a long list of ingredients, including water, sodium lauryl sulfate, fragrance, and Blue dye #5.
What can you infer about the material in the bottle from this label and the appearance of the liquid?
It is a pure substance.
It is a compound.
It is a heterogeneous mixture.
It is a homogeneous mixture.
282
Multiple Choice
How do electrons of an element determine that element's reactivity?
If the valence shell has missing electrons it is very reactive to attract or lose more electrons.
If the valence shell has a full ring of electrons it is very reactive because it needs more.
If the valence shell has a full shell of electrons it is very reactive because it is already full.
If the valence shell has missing electrons it is not reactive because it is missing electrons.
283
Multiple Choice
How are elements and compounds different?
A compound is represented by a chemical formula, and elements are represented by a chemical symbol.
Compounds are represented by chemical symbols of one letter, and elements are represented by symbols of two letters.
Elements are pure substances, and compounds are not.
Elements are always larger units of matter, and compounds are smaller.
284
Multiple Choice

285
Multiple Choice
Pure Substance or Mixture?
made from one type of atom
pure substance
mixture
286
Multiple Choice

287
Multiple Choice

element
compound
mixture of elements
mixture of compounds
mixture of elements and compounds
288
Multiple Choice
Pure Substance or Mixture?
can be made from multiple elements that are chemically combined
pure substance
mixture
289
Multiple Choice
How many elements are in the compound Fe2O3?
0
1
2
3
290
Multiple Choice
Homogeneous or Heterogeneous?
a mixture that is uniform throughout
homogeneous
heterogeneous
291
Multiple Choice

element
compound
mixture of elements
mixture of compounds
mixture of elements and compounds
292
Multiple Choice

Pure Substance or Mixture?
Dr. Pepper
pure substance
mixture
293
Multiple Choice
Hydrogen is considered an element, while hydrogen peroxide is considered a compound. This is because hydrogen peroxide is...
is used for cleaning wounds.
made of two different elements.
is a liquid.
useful to human beings.
294
Multiple Choice

Pure Substance or Mixture?
neon
pure substance
mixture
295
Multiple Choice

element
compound
mixture of elements
mixture of compounds
mixture of elements and compounds
296
Multiple Choice

297
Multiple Choice
The smallest particle of matter
Element
Compound
Atom
Chemical
298
Multiple Choice

299
Multiple Choice

element
compound
mixture of elements
mixture of compounds
mixture of elements and compounds
300
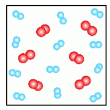
Multiple Choice
301
Multiple Choice
Element or Compound? H6O5
Element
Compound
302
Multiple Choice
303
Multiple Choice
Element or Compound? O6
Element
Compound
304
Multiple Choice
305
Multiple Choice
How many elements? C6H12O6
6
12
3
306
Multiple Choice
How many atoms in TOTAL? C6H12O6
6
12
24
307
Multiple Choice
An element consists of how many types of atoms?
None
1
More than 2
It depends on the element
308
Multiple Choice
Compounds are formed when
Atoms break apart
One atom exists
More than two atoms bond together
More than two atoms combine
309
Multiple Choice
Mixtures can contain which of the following?
Elements
Compounds
Both elements and compounds
310
Multiple Choice
H2O is an example of a
Element
Compound
Mixture
Atom
311
Multiple Choice
What is matter?
anything that has mass and takes up space
anything that has volume and is solid
anything that you can see
312
Explanation Slide...
Matter is EVERYTHING

313
Multiple Choice

Is this a compound or element?
compound
element
314
Explanation Slide...
This is the compound Carbon Dioxide (what we breathe out). It is made of the elements Carbon and Oxygen.

315
Multiple Choice

Is this a compound or element?
compound
element
316
Explanation Slide...
This is a molecule of chlorine - which is an element. When a molecule is only made of one type of atom, it is an element.

7th Grade
TCAP Review
Physical Science
By Jessica Freeman
Show answer
Auto Play
Slide 1 / 316
SLIDE
Similar Resources on Wayground
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Engineering

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

17 questions
guess the logo
Quiz
•
8th Grade

10 questions
Candy
Quiz
•
4th - 8th Grade

10 questions
Fact Check Ice Breaker: Two truths and a lie
Quiz
•
5th - 12th Grade

22 questions
Dot Plots and Histograms
Quiz
•
6th Grade

10 questions
Memorial Day History and Traditions
Interactive video
•
5th - 8th Grade