

Concentrations and Dilutions Review
Presentation
•
Chemistry
•
9th - 12th Grade
•
Practice Problem
•
Easy
Katherine Petersen
Used 2+ times
FREE Resource
35 Slides • 44 Questions
1

2

3

4

5

6

7

8

9

10

11

12

13

14

15

16

17

18
Reorder
Order the following steps to show how properly make a solution.
Determine the mass of solute needed to have the correct number of moles in the solution
Use a balance to measure out the correct mass of solute.
Add the solute to a graduated cylinder or volumetric flask
Add water until the desired volume is reached
Stir the solution until all the solute is dissolved
19

20

Question #1

21
Hotspot
Select the variables that you are GIVEN in the equation.
22
Multiple Choice
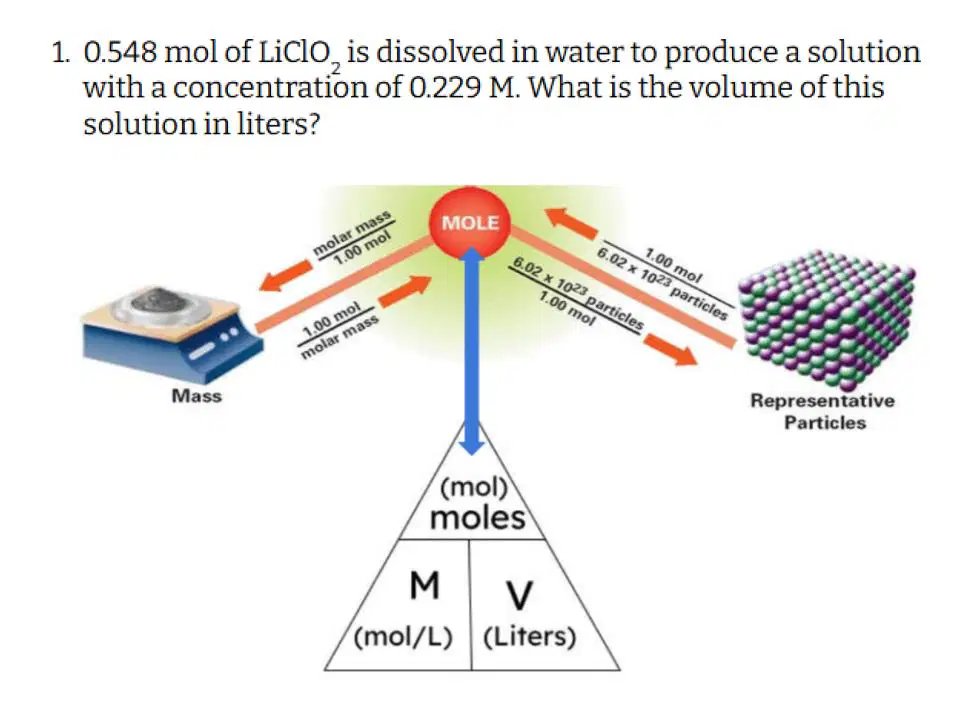
Which of the following correctly describes the steps you need to take to solve this problem?
Convert grams to moles, and then divide moles by concentration to get volume.
Divide moles by concentration to get volume
Multiply concentration by volume to get moles, then convert moles to grams
Convert grams to moles, and then divide moles by volume to get concentration
Multiply mles by concentration to get volume
23
Fill in the Blanks

Type answer...
24

Question #2

25
Hotspot
Select the variables that you are GIVEN in the equation.
26
Multiple Choice
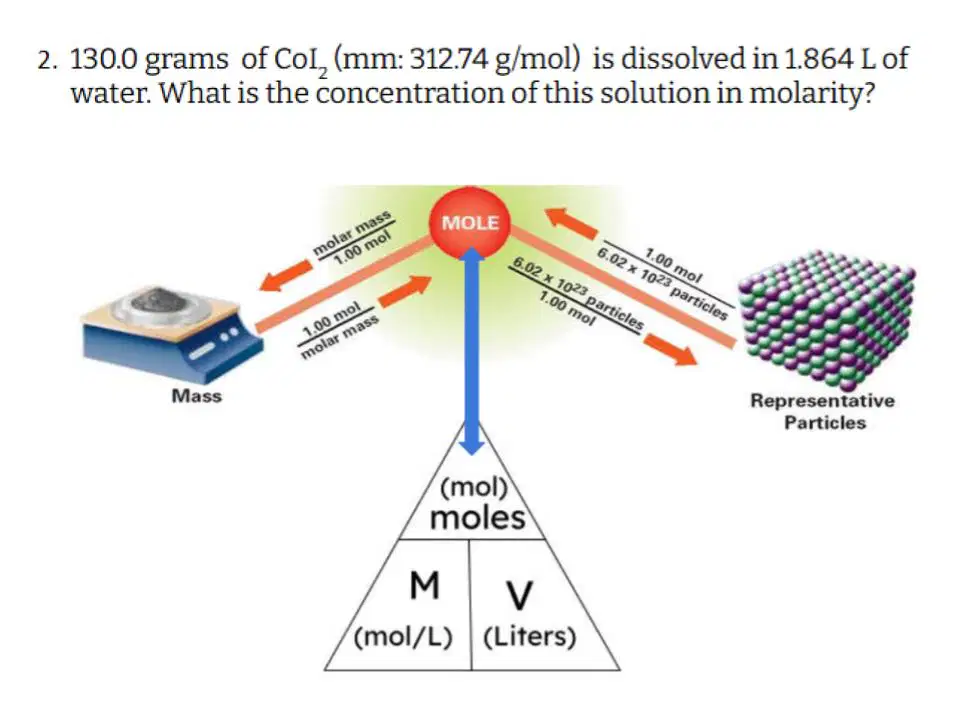
Which of the following correctly describes the steps you need to take to solve this problem?
Convert grams to moles, and then divide moles by concentration to get volume.
Divide mass by volume to get concentration
Multiply concentration by volume to get moles, then convert moles to grams
Convert grams to moles, and then divide moles by volume to get concentration
Multiply mass by volume to get concentration
27
Fill in the Blanks
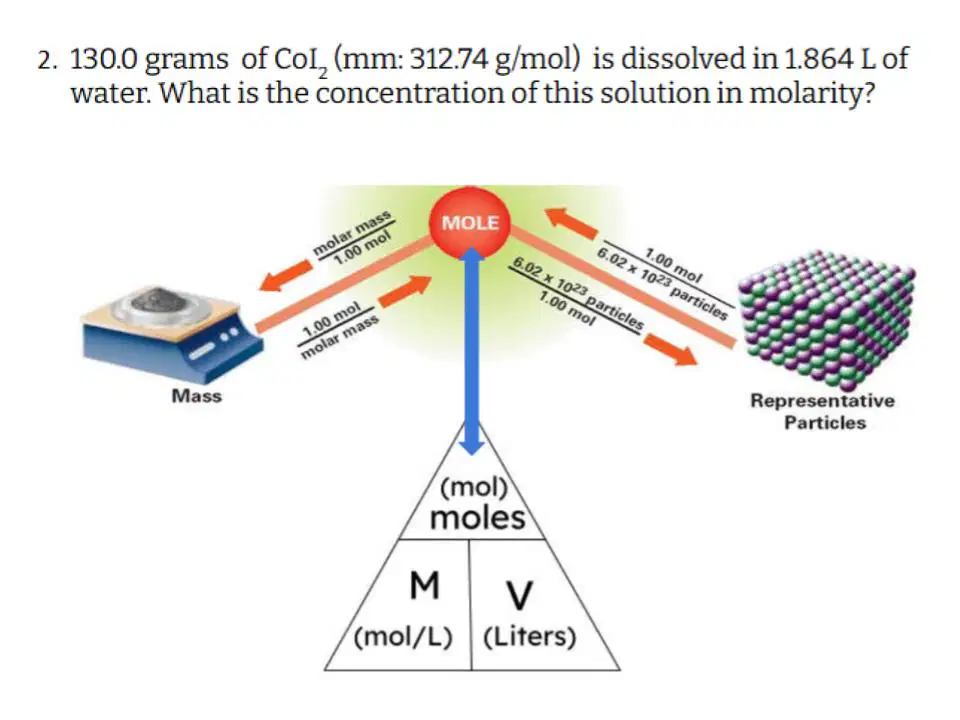
Type answer...
28

Question #3

29
Hotspot
Select the variables that you are GIVEN in the equation.
30
Multiple Choice
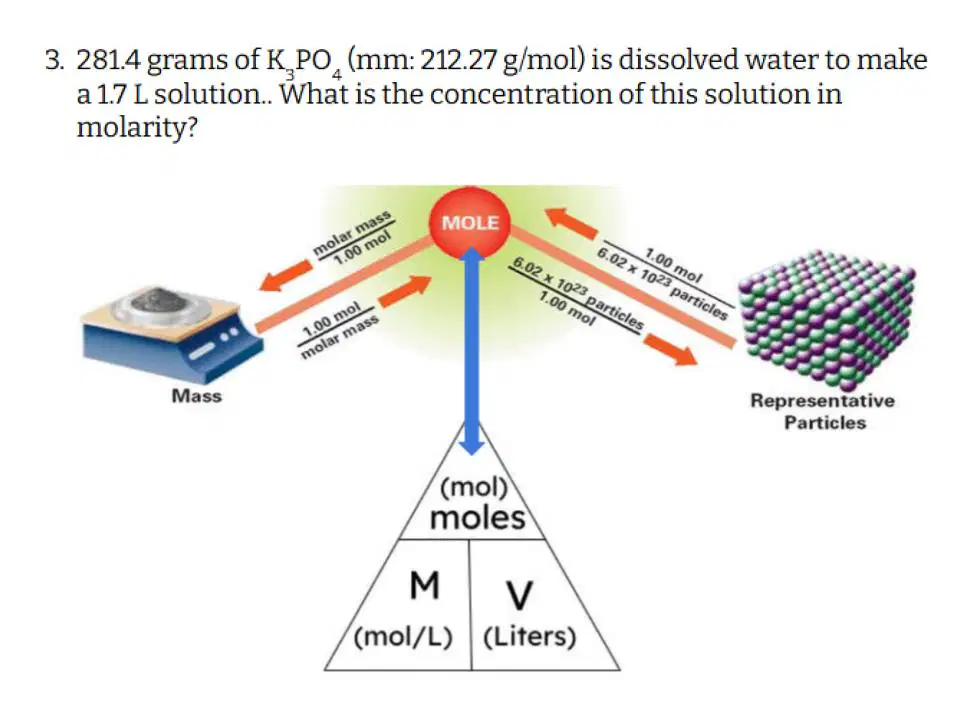
Which of the following correctly describes the steps you need to take to solve this problem?
Convert grams to moles, and then divide moles by concentration to get volume.
Divide mass by volume to get concentration
Multiply concentration by volume to get moles, then convert moles to grams
Convert grams to moles, and then divide moles by volume to get concentration
Multiply mass by volume to get concentration
31
Fill in the Blanks
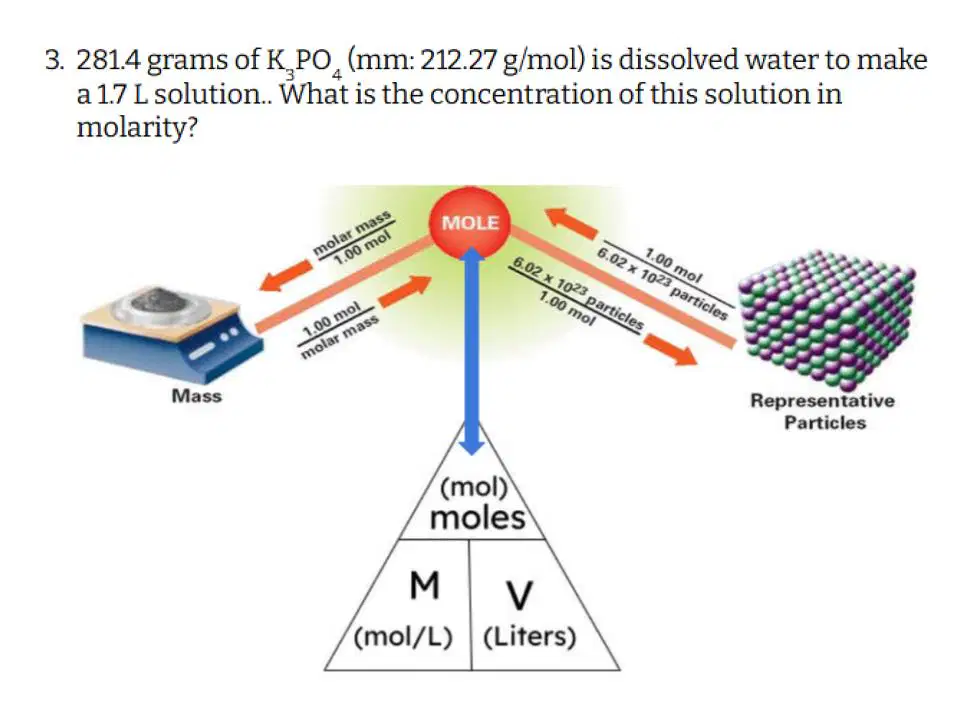
Type answer...
32

Question #4

33
Hotspot
Select the variables that you are GIVEN in the equation.
34
Multiple Choice
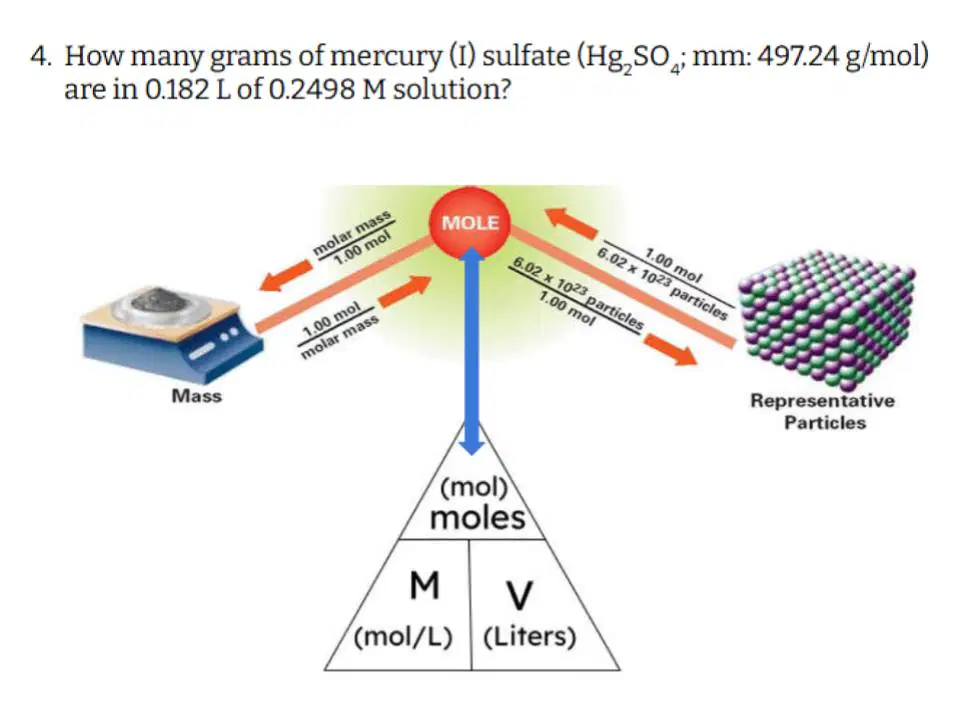
Which of the following correctly describes the steps you need to take to solve this problem?
Multiply concentration by volume to get moles
Divide concentration by volume to get grams
Multiply concentration by volume to get moles, then convert moles to grams
Divide concentration by volume to get moles, then convert moles to grams
Multiply concentration by volume to get grams
35
Fill in the Blanks
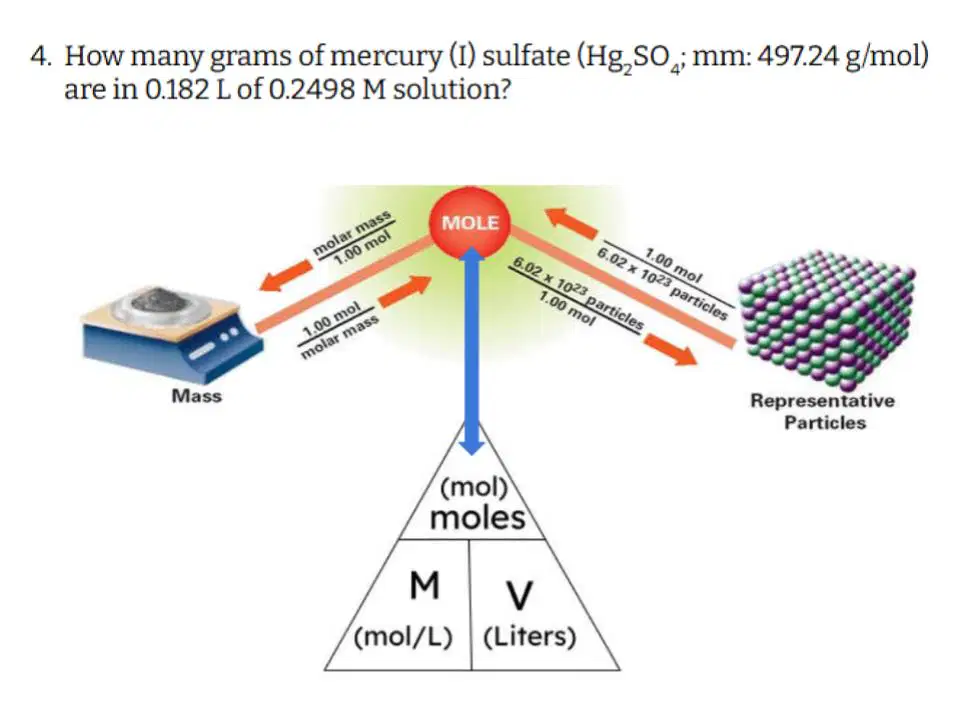
Type answer...
36

Question #5

37
Hotspot
Select the variables that you are GIVEN in the problem.
38
Multiple Choice
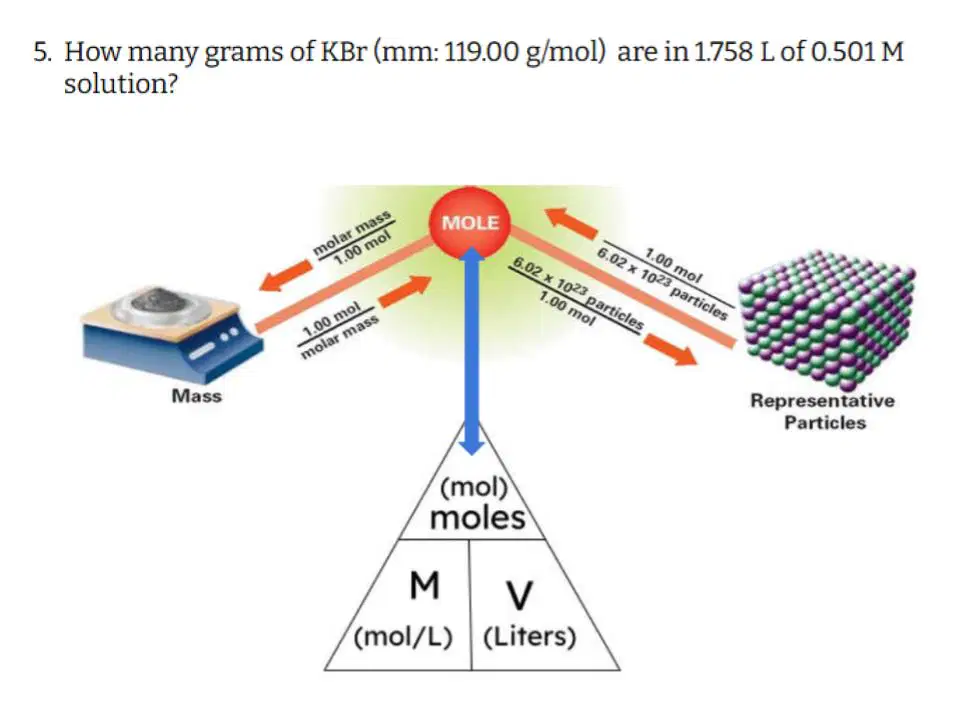
Which of the following correctly describes the steps you need to take to solve this problem?
Multiply concentration by volume to get moles
Divide concentration by volume to get grams
Multiply concentration by volume to get moles, then convert moles to grams
Divide concentration by volume to get moles, then convert moles to grams
Multiply concentration by volume to get grams
39
Fill in the Blanks
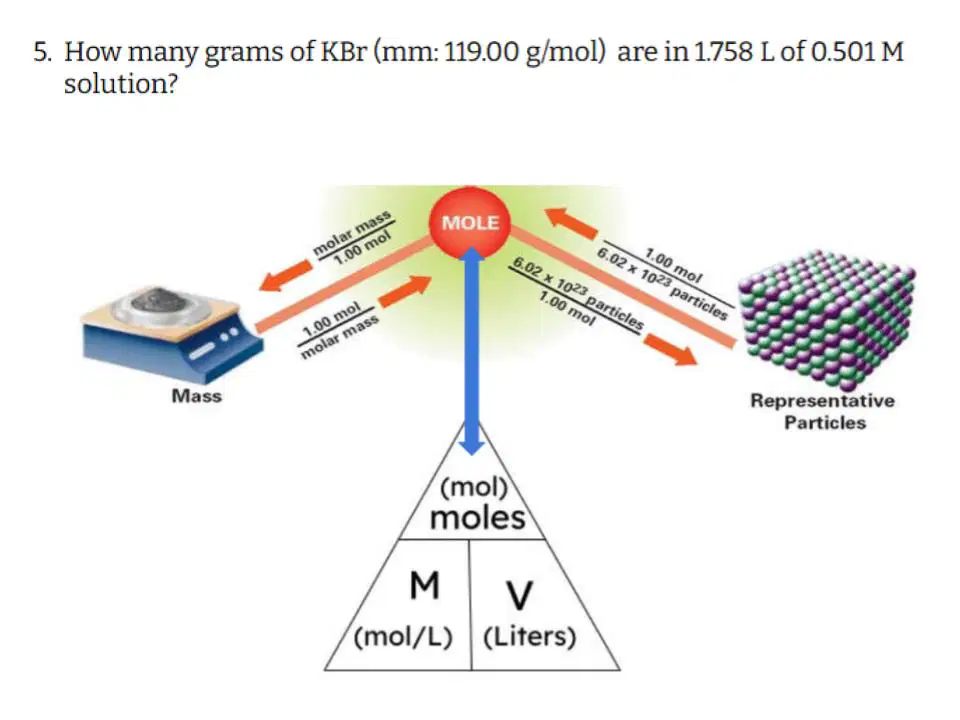
Type answer...
40

Question #6

41
Hotspot
Select the variables that you are GIVEN in the problem.
42
Multiple Choice
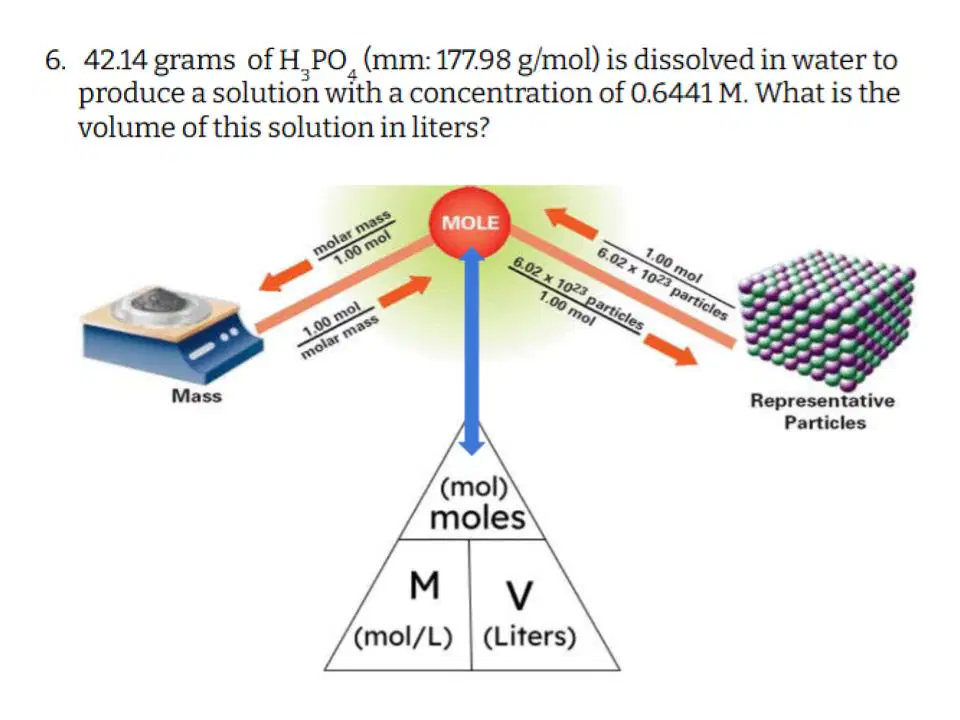
Which of the following correctly describes the steps you need to take to solve this problem?
Convert grams to moles, and then divide moles by concentration to get volume.
Divide mass by concentration to get volume
Convert grams to moles, and then multiply moles by concentration to get volume/
Convert grams to moles, and then divide moles by volume to get concentration
Multiply mass by concentration to get volume
43
Fill in the Blanks
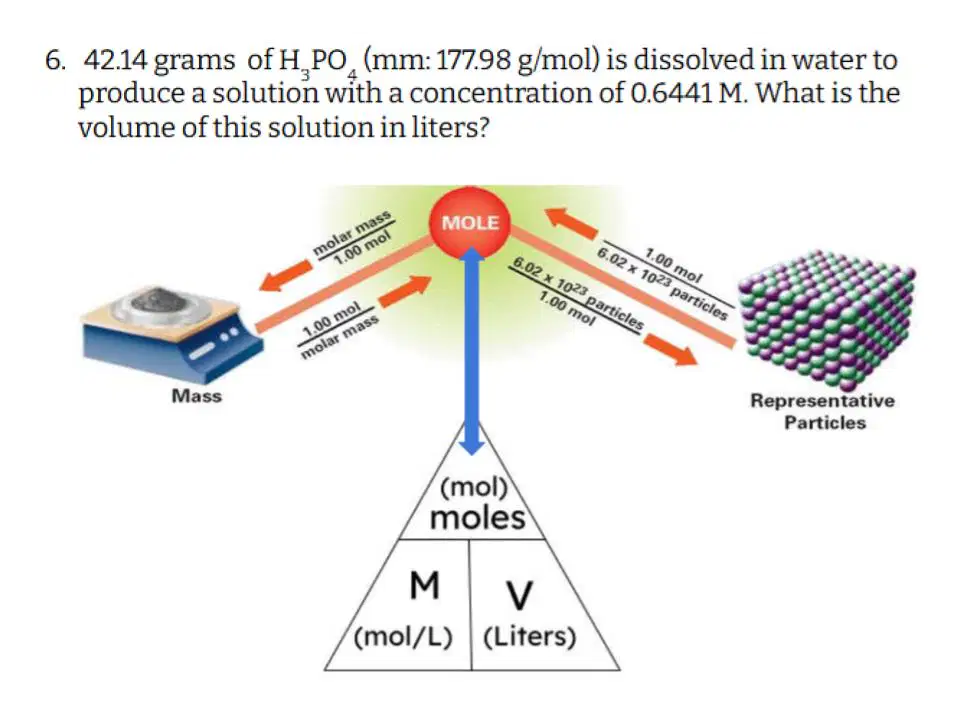
Type answer...
44

45

46

47

48

Example A

!!!!CHANGE 20 mL to 0.020 L!!!!!
49
Dropdown

M1 =
V1 =
M2 =
V2 =
50
Multiple Choice

Which equation shows the proper way to solve Example Problem A?
(2.5 L)(4.0 M)(0.020 L)
(0.020 L)(4.0 M)(2.5 L)
(4.0 M)(0.020 L)(2.5 L)
(0.020 L)(2.5 L)(4.0 M)
(4.0 M)×(2.5 L)×(0.020 L)
51
Fill in the Blanks

Type answer...
52
Multiple Choice

What are the units to the answer o Example Problem A?
Lmol
mLmol
L
mL
mol
53

Example B

54
Dropdown

M1 =
V1 =
M2 =
V2 =
55
Multiple Choice

Which equation shows the proper way to solve Example Problem B?
(0.5 M)(4.0 M)(0.100 mL)
(0.100 mL)(4.0 M)(0.5 M)
(4.0 M)(0.100 mL)(0.5 M)
(0.100 mL)(0.5 M)(4.0 M)
(4.0 M)×(0.5 M)×(0.100 mL)
56
Fill in the Blanks

Type answer...
57
Multiple Choice

What are the units to the answer of Example Problem B?
Lmol
mLmol
L
mL
mol
58

Question #1

59
Dropdown

M1 =
V1 =
M2 =
V2 =
60
Multiple Choice

Which equation shows the proper way to solve Question #1?
(1.0 M)(2.0 M)(1.0 L)
(1.0 L)(2.0 M)(1.0 M)
(2.0 M)(1.0 L)(1.0 M)
(1.0 L)(1.0 M)(2.0 M)
(2.0 M)×(1.0 M)×(1.0 L)
61
Fill in the Blanks

Type answer...
62
Multiple Choice

What are the units to the answer of Question #1?
Lmol
mLmol
L
mL
mol
63

Question #2

64
Dropdown

M1 =
V1 =
M2 =
V2 =
65
Multiple Choice

Which equation shows the proper way to solve Question #2?
(0.050 L)(0.453 M)(0.550 L)
(0.550 L)(0.453 M)(0.050 L)
(0.453 M)(0.550 L)(0.050 L)
(0.550 L)(0.050 L)(0.453 M)
(0.550 L)×(0.050 L)×(0.453 M)
66
Fill in the Blanks

Type answer...
67
Multiple Choice

What are the units to the answer of Question #2?
Lmol
mLmol
L
mL
mol
68

Question #3

69
Dropdown

M1 =
V1 =
M2 =
V2 =
70
Fill in the Blanks

Type answer...
71
Multiple Choice

What are the units to the answer of Question #3?
Lmol
mLmol
L
mL
mol
72

Question #4

73
Dropdown

M1 =
V1 =
M2 =
V2 =
74
Fill in the Blanks

Type answer...
75
Multiple Choice

What are the units to the answer of Question #4?
Lmol
mLmol
L
mL
mol
76

Question #5

77
Dropdown

M1 =
V1 =
M2 =
V2 =
78
Fill in the Blanks

Type answer...
79
Multiple Choice

What are the units to the answer of Question #5?
Lmol
mLmol
L
mL
mol

Show answer
Auto Play
Slide 1 / 79
SLIDE
Similar Resources on Wayground

76 questions
North Korea Lesson
Presentation
•
9th - 12th Grade

72 questions
43 JavaScript Obiekty
Presentation
•
KG

70 questions
La familia y las descripciones
Presentation
•
9th - 12th Grade

75 questions
Photosynthesis
Presentation
•
9th Grade - University

69 questions
PSAJ FISIKA
Presentation
•
9th - 12th Grade

72 questions
Safety & Government Regulations_HST
Presentation
•
9th - 12th Grade

73 questions
The Rise of Big Business and Corruption
Presentation
•
9th - 12th Grade

72 questions
Lesson 1: Electricity
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Interpreting Solubility Curves
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
10th Grade

43 questions
Acids and Bases Review
Quiz
•
10th - 11th Grade

20 questions
Thermochemistry Review
Quiz
•
10th Grade

25 questions
Unit 4 (Mid) Rumble
Quiz
•
10th Grade