

Elements Compound & Mixtures
Presentation
•
Chemistry
•
7th Grade
•
Medium
Standards-aligned
Hiral Mody
Used 2+ times
FREE Resource
59 Slides • 52 Questions
1
Elements Compounds & Mixtures
By Hiral Mody
2
Multiple Choice

What element is present in this necklace?
A. Iron
B. Neon
C. Gold
3
Multiple Choice

What element is this tray made of?
Aluminum
Silver
Nickel
4
Multiple Choice

What element is this wire made of?
Copper
Gold
Silver
5
Multiple Choice

What element is present in this?
Platinum
Iron
Germanium
6
Multiple Choice

What is the red liquid that rise when you get your temperature?
Sulfur
Lithium
Mercury
7
Multiple Choice

What element is present in this can?
Titanium
Boron
Tin
8
Multiple Choice

What element is present in the match stick?
Potassium
Phosphorus
Chlorine
9
Multiple Choice
What is the Periodic Table?
A tabular arrangement of chemical elements
A list of recurring chemical properties
A chart of electron configurations
A map of atomic symbols and numbers
10
The Periodic Table
The Periodic Table is a tabular arrangement of chemical elements, organized based on their atomic number, electron configuration, and recurring chemical properties. It consists of rows called periods and columns called groups. Each element is represented by its atomic symbol and atomic number. The table helps in understanding the relationships between elements and predicting their properties.
11
Periodic Table:
A list of recurring chemical properties. It organizes elements based on their atomic number, electron configuration, and recurring chemical properties. The table is divided into periods (rows) and groups (columns). Each element is represented by its atomic symbol and number. It is a fundamental tool in chemistry.
12
The Periodic Table
13
Multiple Choice
What do periods and groups indicate on the periodic table?
Number of electron shells and similar chemical properties
Atomic number and atomic mass
Number of protons and number of neutrons
Number of valence electrons and reactivity
14
Periodic Table: Atomic Number and Atomic Mass
Trivia: The periods and groups on the periodic table indicate the arrangement of elements based on their atomic number and atomic mass. The atomic number represents the number of protons in an atom, while the atomic mass represents the average mass of the atom. This arrangement helps scientists understand the trends and patterns in the properties of elements. Fun Fact: The periodic table was first developed by Dmitri Mendeleev in 1869.
15
The World of Elements
16
Multiple Choice
Which category of elements includes silicon, arsenic, and germanium?
Metals
Nonmetals
Metalloids
Noble Gases
17
Metalloids
Metalloids are elements that have properties of both metals and nonmetals. They are also known as semimetals. Silicon, arsenic, and germanium are examples of metalloids. They are used in various industries, including electronics and semiconductors. Metalloids play a crucial role in modern technology and scientific research.
18
The Elements: Symbols and Names
19
Naming Elements
Chemical symbols are used to name official names given to elements. They are derived from the element's name in various languages. For example, the symbol for gold (Au) comes from the Latin word 'aurum'. Chemical symbols are essential in scientific research, industry, and everyday life, enabling efficient communication and identification of elements.
20
Sugar is not an element.
When heated, it breaks down into carbon and water vapour.
Some text here about the topic of discussion.
Is Sugar an element?


21
Open Ended
Is Sugar an element?
22
Open Ended
Is air an element? (just a yes or no respond will do)
23
It is a pure substance that cannot be broken down into two or more simpler substances by chemical processes.
Some text here about the topic of discussion.
Element

24
Elements, Compounds and Mixtures

25
Elements, Compounds and Mixtures

26
Elements, Compounds and Mixtures

27
28
Learning goals
At the end of this section, you should be able to:
Write the definition of element
State the chemical symbol of elements using Periodic Table
Subject | Subject
Some text here about the topic of discussion
29
It is a pure substance that cannot be broken down into two or more simpler substances by chemical processes.
Some text here about the topic of discussion.
Element

30
Poll
Is water an element?
Yes
No
not sure
31
Open Ended
Is air an element? (just a yes or no respond will do)
32
Open Ended
Is Sugar an element?
33
Sugar is not an element.
When heated, it breaks down into carbon and water vapour.
Some text here about the topic of discussion.
Is Sugar an element?


34
Two key questions to check whether a substance is an element.
Can the substance be broken down into two or more simpler substances?
Can it be found in the periodic table?
Subject | Subject
Some text here about the topic of discussion
35
Chemists use chemical symbols to represent elements.
Each symbol is unique, consisting of one or two letters.
Some text here about the topic of discussion.
Chemical symbols of elements

36

Elements can be classified into:
Metals
Metalloids
Non-metals
Some text here about the topic of discussion.
Classifying elements
37

Elements are classified based on:
Metallic and non-metallic properties
Physical states (at r.t.p)
Some text here about the topic of discussion.
Classifying elements
38

Some text here about the topic of discussion.
Classifying elements
39
Multiple Choice
Using the Periodic Table, find the symbols of Aluminium.
AL
al
Al
40
Multiple Choice
Using the Periodic Table, find the symbols of Magnesium.
MG
mg
Mg
41
Multiple Choice
Using the Periodic Table, find the symbols of Lead.
PB
pb
Pb
42
Multiple Choice
Using the Periodic Table, find the symbols of Copper.
Cu
CU
cu
43
Multiple Choice
Using the Periodic Table, find the symbols of Nitrogen.
N
n
44
Multiple Choice
Using the Periodic Table, find the symbols of Sulfur.
S
s
45
Elements exist in atoms and molecules.
Some text here about the topic of discussion.
Atoms and Molecules

46
Atoms are the smallest particles of an element that have the chemical properties of that element.
The atoms of an element are different from that of another element.
Some text here about the topic of discussion.
Atoms and Molecules

47

A molecule is a group of two or more atoms that are chemically combined (joined together).
Molecules exist in diatomic or polyatomic.
Some text here about the topic of discussion.
Atoms and Molecules
48
Draw
Circle all the molecules in the picture
49
What is a compound?
A compound is a pure substance that contains two or more elements that are chemically combined in a fixed ratio.
Some text here about the topic of discussion.
Compound


50
Ions
Molecules
What are compounds made of?
Some text here about the topic of discussion


51
A compound has different properties from the elements that form it.
Some text here about the topic of discussion.
Properties of Compounds
52
Names of Compounds
Each compound has a chemical name indicating the elements it contains.
Subject | Subject
Some text here about the topic of discussion

53
Chemical Formulas of Compounds
A compound can be represented by a chemical formula.
The chemical formula shows:
the types of elements present;
the ratio of the different atoms present.
Subject | Subject
Some text here about the topic of discussion

54
Composition of Compounds
Every compound has fixed compositions of the different elements it contains.
Chemical formula can be deduced by the ratio of atoms present in a compound.
Subject | Subject
Some text here about the topic of discussion

55
Decomposition of Compounds
A chemical reaction is necessary to separate the elements in a compound.
In thermal decomposition, heat is used to break down compounds into elements or simpler compounds.
Compounds can also be broken down by using electricity. This process is known as electrolysis.
Subject | Subject
Some text here about the topic of discussion

56
Open Ended
Is water an element? Explain your answer.
57
Multiple Choice
58
Learning goals
At the end of this section, you should be able to:
define a mixture;
differentiate between compounds and mixtures.
Some text here about the topic of discussion
59

Mixtures are made up of two or more substances that are not chemically combined.
Some text here about the topic of discussion.
What are mixtures?
60
There are a few scenarios on how substances are mixed together physically:
mixture of elements
mixture of compounds
mixture of compound and element
Type of mixtures

61
Multiple Select

Which of the following is a pure substance? (you can choose more than one answer)
A
B
C
D
62
Multiple Choice

Which substance is a solid?
A
B
C
D
63
Multiple Choice

Which diagram represent a solution?
A
B
None are solutions
64
Multiple Select
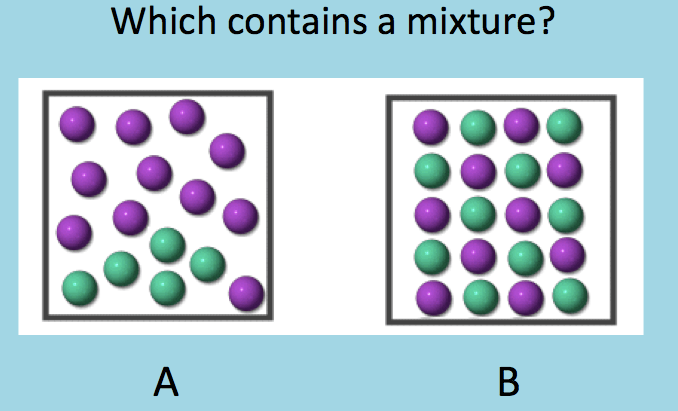
Which of the following is a mixture?
A
B
65
Multiple Select

Which of the following diagram represent a pure substance?
A
B
C
D
66
Multiple Choice

Which of the following contain only elements?
A
B
C
D
67
68
Learning goals
At the end of this section, you should be able to:
Write the definition of element
State the chemical symbol of elements using Periodic Table
Subject | Subject
Some text here about the topic of discussion
69
It is a pure substance that cannot be broken down into two or more simpler substances by chemical processes.
Some text here about the topic of discussion.
Element

70
Poll
Is water an element?
Yes
No
not sure
71
Open Ended
Is air an element? (just a yes or no respond will do)
72
Open Ended
Is Sugar an element?
73
Sugar is not an element.
When heated, it breaks down into carbon and water vapour.
Some text here about the topic of discussion.
Is Sugar an element?


74
Two key questions to check whether a substance is an element.
Can the substance be broken down into two or more simpler substances?
Can it be found in the periodic table?
Subject | Subject
Some text here about the topic of discussion
75
Chemists use chemical symbols to represent elements.
Each symbol is unique, consisting of one or two letters.
Some text here about the topic of discussion.
Chemical symbols of elements

76

Elements can be classified into:
Metals
Metalloids
Non-metals
Some text here about the topic of discussion.
Classifying elements
77

Elements are classified based on:
Metallic and non-metallic properties
Physical states (at r.t.p)
Some text here about the topic of discussion.
Classifying elements
78

Some text here about the topic of discussion.
Classifying elements
79
Multiple Choice
Using the Periodic Table, find the symbols of Aluminium.
AL
al
Al
80
Multiple Choice
Using the Periodic Table, find the symbols of Magnesium.
MG
mg
Mg
81
Multiple Choice
Using the Periodic Table, find the symbols of Lead.
PB
pb
Pb
82
Multiple Choice
Using the Periodic Table, find the symbols of Copper.
Cu
CU
cu
83
Multiple Choice
Using the Periodic Table, find the symbols of Nitrogen.
N
n
84
Multiple Choice
Using the Periodic Table, find the symbols of Sulfur.
S
s
85
Elements exist in atoms and molecules.
Some text here about the topic of discussion.
Atoms and Molecules

86
Atoms are the smallest particles of an element that have the chemical properties of that element.
The atoms of an element are different from that of another element.
Some text here about the topic of discussion.
Atoms and Molecules

87

A molecule is a group of two or more atoms that are chemically combined (joined together).
Molecules exist in diatomic or polyatomic.
Some text here about the topic of discussion.
Atoms and Molecules
88
Draw
Circle all the molecules in the picture
89
What is a compound?
A compound is a pure substance that contains two or more elements that are chemically combined in a fixed ratio.
Some text here about the topic of discussion.
Compound


90
Ions
Molecules
What are compounds made of?
Some text here about the topic of discussion


91
A compound has different properties from the elements that form it.
Some text here about the topic of discussion.
Properties of Compounds
92
Names of Compounds
Each compound has a chemical name indicating the elements it contains.
Subject | Subject
Some text here about the topic of discussion

93
Chemical Formulas of Compounds
A compound can be represented by a chemical formula.
The chemical formula shows:
the types of elements present;
the ratio of the different atoms present.
Subject | Subject
Some text here about the topic of discussion

94
Composition of Compounds
Every compound has fixed compositions of the different elements it contains.
Chemical formula can be deduced by the ratio of atoms present in a compound.
Subject | Subject
Some text here about the topic of discussion

95
Decomposition of Compounds
A chemical reaction is necessary to separate the elements in a compound.
In thermal decomposition, heat is used to break down compounds into elements or simpler compounds.
Compounds can also be broken down by using electricity. This process is known as electrolysis.
Subject | Subject
Some text here about the topic of discussion

96
Open Ended
Is water an element? Explain your answer.
97
Multiple Choice
98
Learning goals
At the end of this section, you should be able to:
define a mixture;
differentiate between compounds and mixtures.
Some text here about the topic of discussion
99

Mixtures are made up of two or more substances that are not chemically combined.
Some text here about the topic of discussion.
What are mixtures?
100
There are a few scenarios on how substances are mixed together physically:
mixture of elements
mixture of compounds
mixture of compound and element
Type of mixtures

101
Multiple Select

Which of the following is a pure substance? (you can choose more than one answer)
A
B
C
D
102
Multiple Choice

Which substance is a solid?
A
B
C
D
103
Multiple Choice

Which diagram represent a solution?
A
B
None are solutions
104
Multiple Choice

Which of the following contain only elements?
A
B
C
D
105
Multiple Select

Which of the following is a mixture?
A
B
106
Multiple Select

Which of the following diagram represent a pure substance?
A
B
C
D
107
108
Multiple Choice
Using the Periodic Table, find the symbols of Magnesium.
MG
mg
Mg
109
Multiple Choice
Using the Periodic Table, find the symbols of Lead.
PB
pb
Pb
110
Multiple Choice
Using the Periodic Table, find the symbols of Copper.
Cu
CU
cu
111
Multiple Choice
Using the Periodic Table, find the symbols of Nitrogen.
N
n
Elements Compounds & Mixtures
By Hiral Mody
Show answer
Auto Play
Slide 1 / 111
SLIDE
Similar Resources on Wayground

110 questions
The Spread of Civilization
Presentation
•
6th - 7th Grade

109 questions
8.2.1-Gravity & Friction
Presentation
•
6th - 8th Grade

110 questions
Maps and Globes
Presentation
•
8th Grade

104 questions
Infinitives
Presentation
•
8th Grade

106 questions
AH Week 11
Presentation
•
8th Grade

106 questions
Ecology - Community Characteristics Lesson
Presentation
•
6th - 8th Grade

106 questions
High School Biology Review Lesson on Ecology
Presentation
•
6th - 8th Grade

106 questions
Ecology Review Lesson
Presentation
•
6th - 8th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Chemistry

15 questions
Fast food
Quiz
•
7th Grade

10 questions
Candy
Quiz
•
4th - 8th Grade

10 questions
Fact Check Ice Breaker: Two truths and a lie
Quiz
•
5th - 12th Grade

10 questions
Memorial Day History and Traditions
Interactive video
•
5th - 8th Grade

14 questions
Volume of rectangular prisms
Quiz
•
7th Grade

15 questions
Middle School Fun Trivia Questions
Quiz
•
7th - 8th Grade

22 questions
Simple Probability
Quiz
•
7th Grade

10 questions
Video Games
Quiz
•
6th - 12th Grade