

Y10A P1.4 L1-Density
Presentation
•
Physics
•
9th - 12th Grade
•
Practice Problem
•
Hard
Standards-aligned
YCIS CQ
FREE Resource
27 Slides • 30 Questions
1

Y10 IGCSE Coordinated Sciences
PHYSICS
2

3
Open Ended
Why is understanding density important in everyday life?
4

5
Multiple Choice
What is the SI unit for measuring volume?
Cubic metre (m³)
Litre (l)
Cubic centimetre (cm³)
Millilitre (ml)
6

7
Multiple Choice
How is 1 cubic metre (m³) defined in terms of its dimensions?
1m x 1m x 1m
1m x 1m x 10m
10m x 10m x 10m
1m x 10m x 1m
8

9
Fill in the Blanks
10

11

12

13
Multiple Choice
If you have a container with a volume of 250 cm³, how many millilitres of water can it hold?
250 ml
25 ml
2.5 ml
1000 ml
14
Multiple Select
Which of the following statements are correct about the relationship between litres, cubic centimetres, and millilitres?
1 litre = 1000 cubic centimetres (cm³)
1 litre = 1000 millilitres (ml)
1 cubic centimetre (cm³) = 1 millilitre (ml)
1 litre = 100 cubic centimetres (cm³)
15

16
Multiple Choice
Which of the following statements about SI units of volume is correct?
1 cubic metre is equal to 1000 litres.
1 litre is equal to 100 cubic centimetres.
1 cubic centimetre is equal to 10 millilitres.
1 litre is equal to 100 cubic decimetres.
17

18
Fill in the Blanks
19

20
Multiple Select
Select all the substances from the table that have a density less than water.
Wood
Ice
Copper
Lead
21
Multiple Choice

Which of the following substances has the highest density according to the table?
Copper
Lead
Aluminium
Water
22
Match

Look at the picture and match the following:
Density 1.25g/ml
Density
0.89 g/ml
Density
1.05g/ml
C
A
B
C
A
B
23
Open Ended
Explain why lead is heavier than aluminium for the same volume, using the concept of density.
24

25

26
Multiple Choice
Which of the following formulas is used to calculate the density of an object?
density = mass / volume
density = volume / mass
density = mass x volume
density = mass + volume
27
Fill in the Blanks
28

29

30

31

32

33
Multiple Choice

Calculate the density of the rectangular prism
2 g/cm3
4 g/cm3
6 g/cm3
8 g/cm3
34
Multiple Choice
A mechanical pencil has the density of 3.000 g/cm3. The volume of the pencil is 15.8 cm3. What is the mass of the pencil?
35
Multiple Choice
Calculate the volume of a liquid with a density of 5.45 g/mL and a mass of 65 g.
11.9 mL
354 mL
0.0838 mL
59.6 mL
36
Multiple Choice
Pure gold has a density of 19.32 g/cm3. How large would a piece of gold be if it had a mass of 318.97 g?
6,163 cm3
16.51 cm3
0.6057 cm3
299.7 cm3
37
Multiple Choice

While digging in the backyard, you find an old coin. Its mass is 26.76 g and its volume is 3 cm3. What is the coin made of?
Marble
Quartz
Copper
Gold
Platinum
38

39

40
Calculating Volume of regular solids
For three-dimensional shapes like cubes and rectangular prisms, we can find the volume by measuring its dimensions; using a unit of length (e.g. meters or centimeters) and then applying the formula L x W x H to determine its volume. The unit of measurement would be the cubic value of the unit used for length.

41
Multiple Choice
Which formula would you use to calculate the volume of a rectangular block?
length x width x height
π x radius² x height
mass / volume
length + width + height
42
Multiple Choice
43
Calculating Volume of liquids
Measuring liquid volume requires a certain method. Vessels used to measure the volume of a liquid should always be placed on a flat surface.
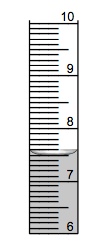
The surface of liquids have a tendency to form a curve (called the meniscus) which should be read at the lowest level of the surface of the liquid, making sure that the eye is level with the surface of the liquid.

44
Multiple Choice
What do you call the curve in the surface of a liquid in a graduated cylinder that we use to measure the volume of an irregular object?
a measurement curve
menicus
eclipsis
45
Multiple Choice

46
Multiple Choice
47
Calculating volume of irregular solids
We can find the volume of an irregular solid object using a method called water displacement method.
Step 1 is to fill the cylinder with enough water to immerse the object and measure the volume.
Next you would add the object to the water until it is totally submerged and measure the new volume of the water.
Subtract the first reading from the second to get the volume, in milliliters, of the object.

48

49
Multiple Select
Which of the following steps are necessary to determine the volume of an irregular solid using a measuring cylinder?
Measure the mass of the solid
Read the water level before and after adding the solid
Subtract the initial volume from the final volume
Multiply the mass by the volume
50

51

52
Multiple Choice

53
Multiple Choice
If you start with 150 mL in a graduated cylinder and add an irregular shaped object and the water level raises to 157.6 mL, what is the volume of your irregular shaped object?
7 mL
157.6 mL
7.6 mL
54
Fill in the Blanks

Type answer...
55
Multiple Choice
Which of the following best describes density?
The amount of space an object occupies
The amount of matter in an object
The mass per unit volume of a substance
The weight of an object in air
56
Open Ended
After learning about density, mass, and volume, what is one real-life situation where understanding these concepts could be useful?
57


Y10 IGCSE Coordinated Sciences
PHYSICS
Show answer
Auto Play
Slide 1 / 57
SLIDE
Similar Resources on Wayground

54 questions
Binary Numbers
Presentation
•
9th - 12th Grade

50 questions
Photosynthesis vs Cellular respiration (regular biology)
Presentation
•
9th - 12th Grade

52 questions
Lesson 5 - Using Communication Skills
Presentation
•
9th - 12th Grade

50 questions
Newton's Second Law of Motion PREVIEW
Presentation
•
9th - 12th Grade

50 questions
Thermodynamics
Presentation
•
9th - 12th Grade

53 questions
Wildlife Conservation
Presentation
•
9th - 12th Grade

52 questions
Subject Pronouns and Ser
Presentation
•
9th - 12th Grade

52 questions
Los Quehaceres (Pre-song story)
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Physics

11 questions
Electricity Explained
Interactive video
•
9th - 12th Grade

18 questions
Series and Parallel Circuits
Quiz
•
9th Grade

15 questions
waves and wave properties
Quiz
•
9th - 12th Grade

35 questions
Electricity and Magnetism SPS10
Quiz
•
8th - 10th Grade

10 questions
Exit Check 7.1 Intro to Waves
Quiz
•
9th Grade

10 questions
Exit Check 7.2: Electromagnetic Waves
Quiz
•
9th Grade

10 questions
Exit Check 7.3 - Analog and Digital
Quiz
•
9th Grade

18 questions
Unit 1 and 2 Concepts Review
Quiz
•
9th - 12th Grade