

Common Assessment Unit 1 REVIEW
Presentation
•
Science
•
6th - 8th Grade
•
Medium
+14
Standards-aligned
Latasha S Tezeno
Used 2+ times
FREE Resource
0 Slides • 135 Questions
1
Dropdown

2
Multiple Choice
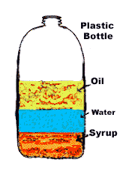
3
Multiple Choice
4
Multiple Choice
When the density of a substance is lower than the density of water the substance the substance does what in water?
sink
float
5
Categorize
Definite shape and definite volume
definite volume No definite shape
No definite volume and no definite shape
6
Match
Match the following
solid
liquid
gas
solid
liquid
gas
7
Match
Match the following to there state of matter
gas
solid
liquid
air
water
rock
air
water
rock
8
Multiple Choice

9
Multiple Choice

10
Multiple Choice

11
Multiple Choice

Which word is under the wrong heading?
12
Multiple Choice

What is an example of a liquid
The soda
The ice cubes
The bubbles
The glass
13
Multiple Choice

14
Multiple Choice

What is the new substance?
15
Multiple Choice

16
Multiple Choice

The picture below is a model of which physical state?
Solid
Liquid
Gas
Plasma
17
Multiple Choice
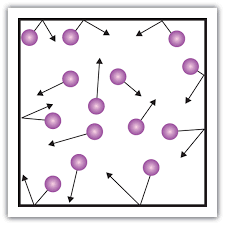
What state of matter?
Solid
Liquid
Gas
Plasma
18
Multiple Choice

Which of the choices provided correctly labels each image shown?
1. Heterogeneous mixture
2. Homogeneous mixture
3. Pure Substance (Compound)
4. Pure Substance (Element
1. Pure Substance (Element)
2. Pure Substance (Compound)
3. Homogeneous Mixture
4. Heterogeneous Mixture
1. Homogeneous mixture
2. Heterogeneous mixture
3. Pure Substance (Element)
4. Pure Substance (Compound)
1. Pure Substance (Compound).
2. Pure Substance (Element)
3. Heterogeneous Mixture
4. Homogeneous Mixture
19
Multiple Choice

What state of matter does this picture show?
gas
solid
liquid
ocean
20
Multiple Choice
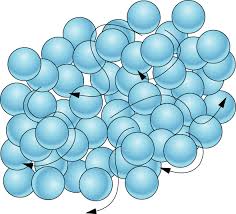
What state of matter?
Solid
Liquid
Gas
Plasma
21
Multiple Choice

List the states of matter in order.
Solid, liquid gas
Gas, solid, liquid
Liquid, solid, gas
Gas, gas, solid
22
Multiple Choice

Which form is this picture showing?
gas
solid
liquid
ocean
23
Multiple Choice

24
Multiple Choice

Look at the picture. Which is true?
Material A is a liquid
Material C is a solid
Material B is a gas
Material B is a liquid
25
Multiple Choice
26
Multiple Choice
27
Multiple Choice
One state of matter has no definite shape. Which one is it?
Solid
Gas
Liquid
Color
28
Multiple Choice
When mixing milk and vinegar, a white, chunky, solid substance is formed. What has occurred?
Physical change: state of matter
Chemical change: precipitate
Physical change: freezing
Chemical change: rust
29
Multiple Choice

Burning wood is a
chemical change
physical change
30
Multiple Choice
Which answer below makes a correct statement?
Physical change only changes the color.
Physical change only changes the size.
Physical change changes the matter into something totally different.
Physical change begins and ends with the same matter.
31
Multiple Choice
32
Multiple Choice
33
Multiple Choice

Change in color is a physical change.
True
False
34
Multiple Choice
35
Multiple Choice
A student mixes yellow powder with water. Which observation would prove a CHEMICAL change occurred?
The powder DISSOLVED in the water
The water turned the COLOR yellow
The bag's TEMPERATURE changed
The STATE OF MATTER changed
36
Multiple Choice

An ice pack getting cold after you put it in the freezer for an hour is a
chemical Change
physical Change
37
Multiple Choice

Salt dissolved in water is a
physical change
chemical change
38
Multiple Choice

Exploding fireworks is a
Physical Change
Chemical Change
Katy Perry Song
39
Multiple Choice
It a state of matter wherein it has a definite shape and volume.
A. liquid
B. gas
C. solid
D. all of the above
40
Multiple Choice
It is a mixture wherein it has a single phase or uniform substances.
A. homogenous mixture
B. heterogenous mixture
C. solute
D. solvent
41
Multiple Choice

COFFEE IS
GAS
ELEMENT
HOMOGENEOUS MIXTURE
HETEREGENEOUS MIXTURE
42
Multiple Choice

BURGER IS
LIQUID
ELEMENT
HETEROGENEOUS MIXTURE
HOMOGENEOUS MIXTURE
43
Multiple Choice

CHOCOLATE CHIPS IS
SOLUTION
NOT A MIXTURE
HOMOGENEOUS MIXTURE
HETEROGENEOUS MIXTURE
44
Multiple Choice

TOMATO KETCHUP IS
HETEROGENEOUS MIXTURE
ELEMENT
MOLECULE
HOMOGENEOUS MIXTURE
45
Multiple Choice
What is the definition of a homogeneous mixture?
A mixture where the components separate into distinct layers according to density.
A mixture where the components are distributed unevenly and the composition varies throughout.
A mixture where the components are distributed uniformly and the composition is the same throughout.
A mixture where the components are distributed randomly and the composition changes at different points.
46
Multiple Select
Identify which images are HETEROGENEOUS MIXTURES! (Check all that apply)
47
Multiple Choice
48
Multiple Choice
49
Multiple Choice
50
Multiple Choice
51
Multiple Choice
52
Multiple Choice
53
Multiple Choice
54
Multiple Choice
55
Multiple Choice
56
Multiple Choice

Elements in the shaded area can best be described as -
Malleable
Brittle
Dull
Insulators
57
Multiple Choice
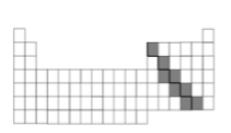
Elements in the shaded area can best be described as -
Shiny and brittle
Insulators
Good conductors
58
Multiple Choice

59
Multiple Choice

60
Multiple Choice
Fill in the blank: Nonmetals have a ___________ surface, and they do not conduct heat or electricity.
electricity
dull
changed
luster
metals
61
Multiple Choice
62
Multiple Choice
63
Multiple Choice
Metals are shiny, good conductors, malleable, and ductile.
True
False
64
Multiple Choice
65
Multiple Choice
66
Multiple Choice
67
Multiple Choice
68
Multiple Choice
69
Multiple Choice
ductile means...
can be made into wire
can be bent or hammered
can light up a light bulb
it's shiny
70
Multiple Choice

Which group of elements could have the characteristic of brittle?
Metal
Nonmetal
71
Multiple Choice
72
Multiple Choice
73
Multiple Choice

Students were given four unknown substances and asked to observe their characteristics. Their observations are shown in the table below. Which of the unknown substances is most likely metal?
1 and 4
2 and 5
1 and 3
2 and 4
74
Multiple Choice
Roger is examining an object in science class. It is dull, yellow, and a brittle solid. The sample is most likely a—
Metal
Nonmetal
Metalloid
Nobel gas
75
Multiple Choice

Elements in the shaded area can best be described as -
Malleable
Brittle
Dull
Insulators
76
Multiple Choice

77
Multiple Choice
Fill in the blank: Nonmetals have a ___________ surface, and they do not conduct heat or electricity.
electricity
dull
changed
luster
metals
78
Multiple Choice

Sulfur is considered:
malleable
brittle
ductile
a good conductor
79
Multiple Choice

Which elements are colored red?
nonmetals
metals
metalloids
80
Multiple Choice

81
Multiple Choice
82
Multiple Choice

83
Multiple Choice
84
Multiple Choice
Carbon is dull and brittle. Carbon is a
metal
nonmetal
metalloid
85
Multiple Choice

Elements in the shaded area can best be described as -
Shiny and brittle, semiconductors
Insulators
Good conductors
86
Multiple Choice
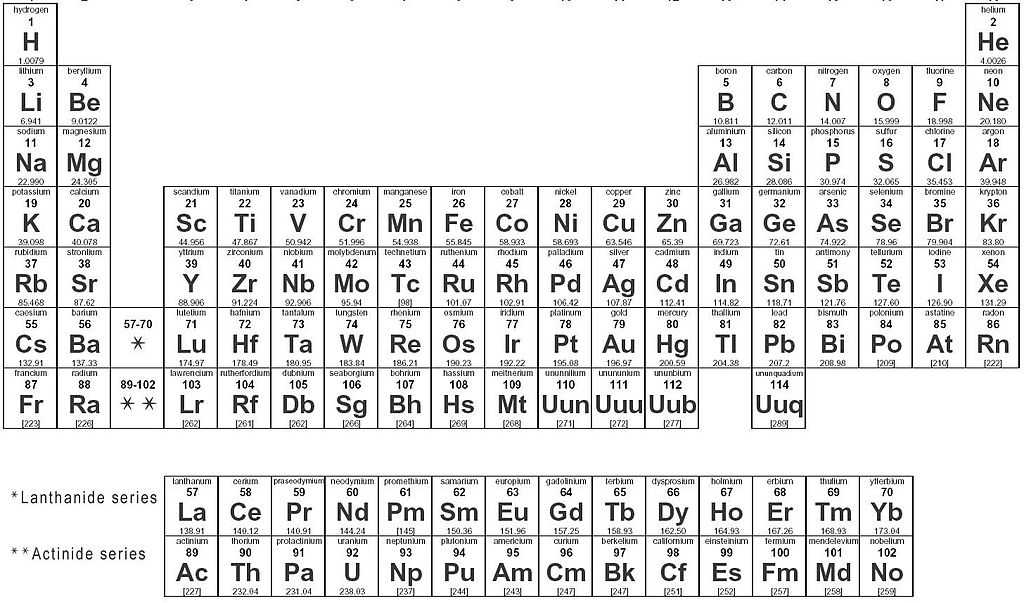
87
Categorize
Definite shape and definite volume
definite volume No definite shape
No definite volume and no definite shape
88
Multiple Choice

Identify the following
compound
homogeneous mixture
element
heterogeneous mixture
89
Multiple Choice

90
Multiple Choice
What causes particles to move around more?
Condensation
Melting
Liquid
Heat
91
Multiple Choice
Have a definite shape and a definite volume
Solid
Liquid
Gas
92
Multiple Choice
Have a definite volume but no definite shape
Solid
Liquid
Gas
93
Multiple Choice
Have no definite volume and no definite shape
Solid
Liquid
Gas
94
Multiple Choice

What does this image show?
Solid
Liquid
Gas
95
Multiple Choice

What does this image show?
Solid
Liquid
Gas
96
Multiple Choice

What does this image show?
Solid
Liquid
Gas
97
Multiple Choice
What causes a change in state?
Adding heat
Removing heat
Both adding and removing heat
98
Multiple Choice
When a solid changes into a liquid it is called
Melting
Sublimation
Condensation
Freezing
99
Multiple Choice
Freezing is when a liquid changes into a _____
Gas
Liquid
Solid
100
Multiple Choice
True or False: When you heat a solid or liquid its particles begin to move faster.
True
False
101
Multiple Choice
True or False: Temperature is the measure of the vibration speed of the atoms of a substance.
True
False
102
Multiple Choice
True or False: "Matter" is anything that has a mass and takes up space
True
False
103
Multiple Choice
True or False: A liquid has a indefinite shape, definite volume
True
False
104
Multiple Choice
Particles of a liquid
are tightly packed together and stayed fixed
have no viscosity
decrease in volume with increasing temperature
are free to move in a container but stay close together
105
Multiple Choice
In which state of matter do atoms spread apart and fill all the space available to them?
crystal
liquid
gas
solid
106
Multiple Choice
A group of two or more atoms held together by chemical bonds.
Atom
Elements
Compound
Molecule
107
Multiple Choice
A pure substance that cannot be decomposed into simpler substances.
Atom
Elements
Compound
Mixture
108
Multiple Choice
Beth left her crayons outside on a hot day. What will most likely happen to the crayons?
They will melt.
They will cook.
They will freeze.
109
Multiple Choice
Jared wants to make ice cubes from water. Which of these should he do?
put water in the freezer
boil water in a pan on the stove
melt water outside in the sunlight
110
Multiple Choice

Water from a faucet became ice cubes. What changed the water to ice?
heating the water
melting the water
freezing the water
111
Multiple Choice

The sun is changing the ice to water. What causes this change?
It is cooking.
It is melting.
It is freezing.
112
Multiple Choice
Which picture shows a gas?
113
Multiple Choice
Which object shows a liquid?
114
Multiple Choice
Which picture shows a liquid?
115
Multiple Choice

Steam is a.....
Solid
Liquid
Gas
Science
116
Multiple Choice

Rain is a.....
Solid
Liquid
Gas
Inconvenience
117
Multiple Choice

Ice is a....
Solid
Liquid
Gas
Water
118
Multiple Choice
Is air solid, liquid, or gas?
Solid
Liquid
Gas
119
Multiple Choice

The fish is swimming in....
Solid
Liquid
Gas
Water Vapor
120
Match
Match the following
another word for states of matter
has a definite shape and a definite volume
has a definite volume and no definite shape
has no definite volume and no definite shape
when particles in a solid or liquid move faster and further apart
phase
solid
liquid
gas
expansion
phase
solid
liquid
gas
expansion
121
Multiple Choice
How tightly particles are packed determines _______________.
the type of atoms present
what states the matter takes
the temperature of the matter
what the next phase will be
122
Multiple Choice
A student observes a substance in a beaker. The substance has a volume of 150mL. The student transfers the substance to a smaller flask. The volume did not change, but the substance has changed to match the shape of the flask. Which substance is the student observing.
a solid changing into a liquid
a liquid transferring to another container
a gas becoming a liquid
a gas becoming a solid
123
Multiple Choice
Vaporization is the term that describes __________________.
gas changing to a liquid
solid changing to a liquid
liquid changing to a gas
gas changing to a solid
124
Multiple Choice
Cooling causes particles _________________.
to move around more
to move around less
to evaporate
to bond better with each other
125
Match
Match the following
Gas
liquid
solid
Gas
liquid
solid
126
Multiple Choice
What do all chemical changes have in common?
Energy is absorbed and it feels cold.
Energy is released and it feels hot.
Gas bubbles are produced.
New substances form.
127
Multiple Choice
A teacher tells students to describe the physical properties of 10 mL of vinegar and 10 mL of milk. The teacher then pours the vinegar and milk into the same beaker. The students are surprised to see a white solid substance form within the beaker as the two substances are combined.
What occurs within the beaker as the teacher mixes the vinegar and milk?
A physical change that creates a new substance.
A physical combination that produces a solid.
A chemical change that produces a precipitate.
A corrosive reaction between two liquids.
128
Multiple Choice
3.Marie has a balloon with a small amount of baking soda in it. She fits the open end of the balloon over a graduated cylinder containing some vinegar. As she lifts the end of the balloon the baking soda pours into the vinegar causing bubbling and fizzing to occur. Marie observes the balloon began to "fill" up. Based on these observations, which of these can Marie determine about the new substance formed by mixing baking soda with vinegar?
only a physical change occurred.
a chemical reaction occurred producing a gas.
a chemical reaction occurred producing a solid.
no chemical reaction took place.
129
Multiple Choice
Before going to school, a student cuts a ripe apple into six slices and places them in a plastic bag. During lunch, the student observes that the apple slices now have brown spots. Which of the following indicates a chemical change in the apple?
cutting the apple into six slices.
placing the slices in a plastic bag.
unexpected color change of the apple.
removing the brown spots with a knife.
130
Multiple Choice

A teacher fills a bottle half-full of vinegar. He places baking soda in a paper napkin tied to a rubber stopper. When he shakes the bottle, the vinegar reacts with the baking soda producing bubbles. When the bubbles fill the bottle, the stopper is forced off the top of the bottle. Why does this happen?
The gas created in the reaction takes up more room than the space in the bottle.
The temperature of the bottle changes which makes the stopper become too loose.
The string, napkin, and stopper make the reaction happen too fast.
There is too much vinegar left in the bottle.
131
Multiple Choice

A student has two clear liquids, Liquid A and Liquid B. One beaker is filled with Liquid A and Liquid B is added drop by drop. The student recorded the data shown in the table below. What conclusion can be drawn from her observations?
A precipitate formed
Temperature increased
Color changed
Mass increased
132
Multiple Choice

At a cold football game, Robert uses an instant hand-warmer to keep his hands warm. He shakes the hand-warmer to mix the two chemicals inside the pouch which instantly releases heat. What evidence indicates that a chemical change has occurred?
The contents in the hand-warmer melted.
The contents in the hand-warmer dissolved.
The contents in the hand-warmer changed temperature.
The contents in the hand-warmer mixed.
133
Multiple Select

A student has two clear liquids, Liquid A and Liquid B. One beaker is filled with Liquid A and Liquid B
is added drop by drop. The student recorded data from her observations in the table below. What evidence that a chemical change has occurred can be found in her observations? Select TWO correct answers
A precipitate formed
Temperature increased
Color changed
Mass increased
134
Multiple Choice
A black iron chair is placed outdoors. Over time, the chair becomes coated with a red substance.
Which is the best explanation for the appearance of the red substance?
The red substance is a form of black iron that appears.
The red substance is a mixture of two substances in iron.
The red substance is the product of a chemical change.
The red substance formed when the black substance melted.
135
Multiple Choice

Three reactions are observed in a lab, as shown.
Reaction 1: a gas is produced after a solid and a liquid solution are combined.
Reaction 2: a solid precipitate appears after two liquid solutions are combined.
Reaction 3: a solid is dissolved after being placed in a beaker of water.
Reaction 4: a solid begins to burn after being exposed to air.
Which statement correctly compares two of the reactions?
Reaction 2 is a chemical change because a precipitate is formed. Reaction 4 is a physical change because the solid is burning.
Reaction 1 is a chemical change because a gas is produced. Reaction 3 is a physical change because dissolving does not make a new substance.
Reaction 3 is a chemical change because a solid is being dissolved. Reaction 1 is a physical change because a gas is produced.
Reaction 4 is a chemical change because a solid is burning. Reaction 1 is a physical change because a gas is produced.

Show answer
Auto Play
Slide 1 / 135
DROPDOWN
Similar Resources on Wayground

125 questions
Art chapter
Presentation
•
7th Grade

135 questions
Sound Waves AJH
Presentation
•
7th - 8th Grade

128 questions
sôn lình 12 v2
Presentation
•
KG

130 questions
EES Reviewer Q3
Presentation
•
7th Grade

128 questions
Numeracy
Presentation
•
6th - 8th Grade

128 questions
Numeracy
Presentation
•
6th - 8th Grade

127 questions
Unit 1 Review
Presentation
•
KG

120 questions
The Spread of Islam and Islamic Empires
Presentation
•
6th - 8th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

26 questions
Amplify Chemical Reactions
Quiz
•
6th - 8th Grade

20 questions
Rocks and The Rock Cycle
Quiz
•
6th Grade

16 questions
Photosynthesis and Cellular Respiration Review
Quiz
•
7th Grade

15 questions
Punnett Squares
Quiz
•
6th Grade

20 questions
genetics, punnett squares, heredity
Quiz
•
7th Grade

18 questions
Oceans, Atmosphere, Climate Vocabulary Amplify
Quiz
•
6th Grade

49 questions
NC Released EOG 8 Science (2024)
Quiz
•
8th Grade