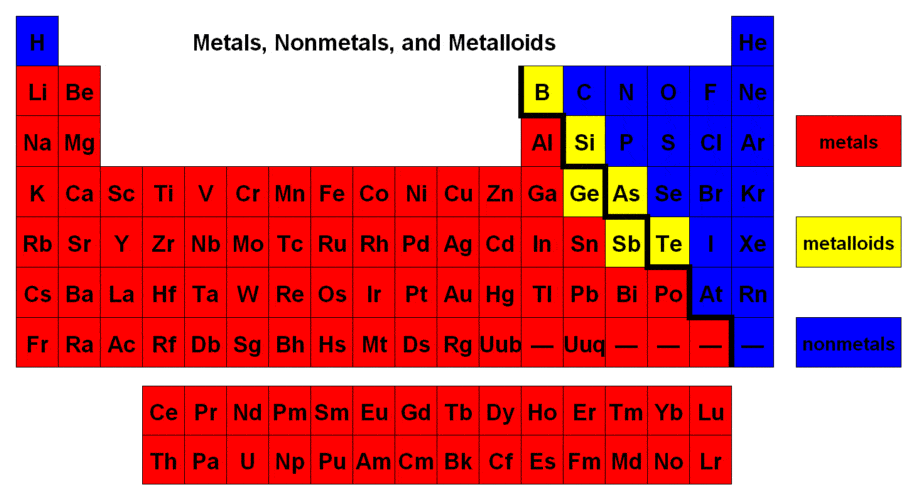
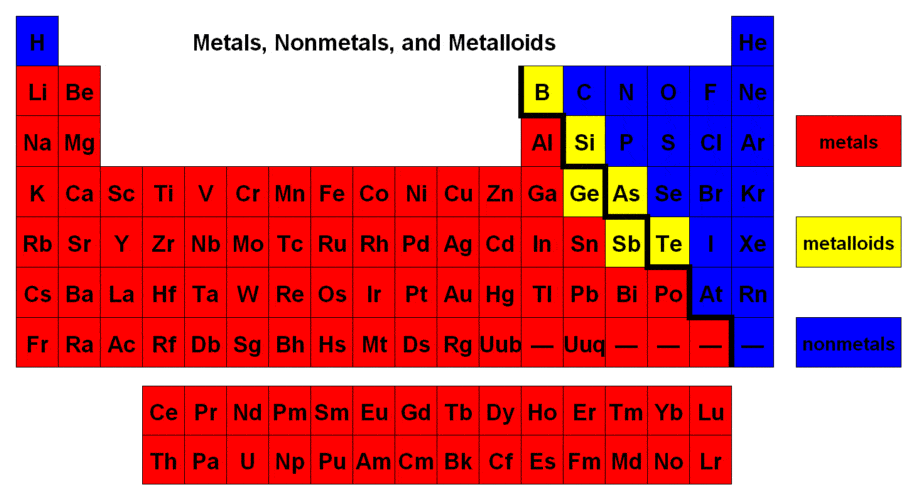
Module 7: Introduction to Chemistry
Presentation
•
Chemistry
•
9th - 12th Grade
•
Medium
+3
Standards-aligned
Used 1+ times
FREE Resource
49 Slides • 43 Questions
1
2
Multiple Choice
3
4
5
6
Multiple Choice
7
8
Categorize
9
10
Reorder
11
12
Multiple Choice
13
14
Multiple Choice
15
16
Multiple Choice
17
18
Multiple Choice
19
20
Multiple Choice
21
22
Fill in the Blank
23
24
Multiple Choice
25
Multiple Choice
26
Multiple Choice
27
Multiple Choice
28
29
Multiple Choice
30
31
Multiple Choice
32
33
34
Multiple Choice
35
36
37
Multiple Choice
38
39
Multiple Choice
40
Multiple Choice
41
Multiple Choice
42
43
Multiple Select
44
45
46
47
48
49
50
51
52
53
54
55
56
57
58
59
60
61
62
63
64
65
66
67
68
69
70
71
72
Multiple Choice
73
Multiple Choice
74
Multiple Choice
75
Multiple Choice
76
Multiple Choice
77
Multiple Choice
78
Multiple Choice
79
Multiple Choice
80
Multiple Choice
81
Multiple Choice
82
Multiple Choice
83
Multiple Choice
84
Multiple Choice
85
Multiple Choice
86
Multiple Choice
87
Multiple Choice
88
Multiple Choice
89
Multiple Choice
90
Multiple Choice
91
Fill in the Blank
92
Fill in the Blank
Show answer
Auto Play
Slide 1 / 92
SLIDE
Similar Resources on Wayground

88 questions
RL2 Part 2 Retell Stories GA Milestones Review
Lesson
•
KG

89 questions
AZ Soils Guide
Lesson
•
9th - 12th Grade

89 questions
WHI 11.01, 11.02. and 11.02 Empires of Asia
Lesson
•
9th - 12th Grade

86 questions
LEARNING GUIDE FAMILY
Lesson
•
KG - University

85 questions
GRAMMAR - REPORTED SPEECH
Lesson
•
9th Grade - University

92 questions
WH- Cold War
Lesson
•
9th - 12th Grade

91 questions
Unit 7 - age of revolutions
Lesson
•
9th Grade - University

86 questions
Lesson 8 - Photosynthesis and Cellular Respiration
Lesson
•
9th - 12th Grade
Popular Resources on Wayground

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

29 questions
Alg. 1 Section 5.1 Coordinate Plane
Quiz
•
9th Grade

22 questions
fractions
Quiz
•
3rd Grade

11 questions
FOREST Effective communication
Lesson
•
KG

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade
Discover more resources for Chemistry

22 questions
Unit 9 Gas Law Quiz
Quiz
•
10th Grade

10 questions
Exploring Types of Chemical Reactions
Interactive video
•
6th - 10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Acids and Bases
Quiz
•
10th Grade

30 questions
Energy Review
Quiz
•
9th Grade

7 questions
GCSE Chemistry - Balancing Chemical Equations #4
Interactive video
•
9th - 10th Grade

20 questions
Chemistry: Classification of Matter
Quiz
•
10th Grade

40 questions
Unit 3 (Part 1) Chemical Equations & Reactions Review Game
Quiz
•
8th - 12th Grade