
- Resource Library
- Science
- Chemistry
- Counting Atoms In Compounds
- *structure Of Matter Atoms, Molecules And Compounds

*Structure of Matter - Atoms, Molecules and Compounds
Presentation
•
Science
•
6th - 8th Grade
•
Practice Problem
•
Easy
Standards-aligned
HUNTER BREKKE
Used 4+ times
FREE Resource
11 Slides • 8 Questions
1
Structure of Matter:
Atoms, Molecules, and Compounds
By Mr. Brekke
2
Learning target
By the end of this lesson, I will be able to:
Describe the difference between elements, molecules, and compounds.
Identify examples of simple molecules and compounds by reading a chemical formula.
Explain how atoms come together to make molecules and compounds.
3
Key Vocabulary
Element
An element is a pure substance made of only one kind of atom. Examples include Oxygen (O) and Hydrogen (H).
Molecule
A molecule forms when 2+ atoms join. They can be the same or different atom types.
Compound
A compound is a molecule with 2+ different elements bonded. Water (H2O) is an example.


4
Multiple Select
Which of the following are elements?
**Select multiple
Cl (chlorine)
NaCl
(sodium chloride)
He
(Helium)
CO2
(carbon dioxide)
5
Multiple Choice
What is the difference between H and H2O?
They are the same.
H is an element and H2 is a molecule.
They are made of different elements.
6
Review
Element
Substance that cannot be broken down into a simpler form.
Examples: Hydrogen, Helium, Oxygen, Gold, Magnesium.
Find them on the Periodic Table of Elements.
7
What are Molecules?
A group of two or more atoms held together by chemical bonds.
Can be the same or different atoms.

8
Match
Matching:
Which of the following chemical formulas are elements?
Which of the following chemical formulas are molecules?
H2O
O
O2
Co
CO2
Molecule
Element
Molecule
Element
Molecule
Molecule
Element
Molecule
Element
Molecule
9
2 or more different atoms (elements) bonded together.
Examples: H2O, CO2, or C6H12O6
Compound
2 or more atoms bonded together.
Can be the same or different atoms (H2 or H2O)
Molecule
Molecule vs compound
10
Compounds
Compounds are also Molecules.
Compounds can be separated by chemical means into their components. For example H2O can be broken into H2 and O through chemical reactions.
11
Multiple Choice
Can a compound also be a molecule?
Yes
No
12
O
O2
CO2
H
H2O
C6H12O6
Which of the following are Molecules?
**Think about the definition of a molecule.
13
Compounds
H2O
C6H12O6
CO2
Elements
H
O
Which of the following are Molecules? (Answers)
**Think about the definition of a molecule.
Molecules
H2O
C6H12O6
O2
CO2
14
Counting Atoms in chemical Formulas
Chemical formula represents the types (elements) and Number of atoms in a compound/molecule. Example = H2O or C6H12O6
Chemical Formula | First Atom | Second Atom | Third Atom |
|---|---|---|---|
O2 | Oxygen (O) = 2 | | |
H2O | Hydrogen (H) = 2 | Oxygen (O) = 1 | |
C6H12O6 | Carbon (C) = 6 | Hydrogen (H) = 12 | Oxygen (O) = 6 |
15
Exit Ticket - Quiz
16
Multiple Choice

hydrogen
boron
17
Multiple Choice

beryllium
sodium
18
Multiple Choice
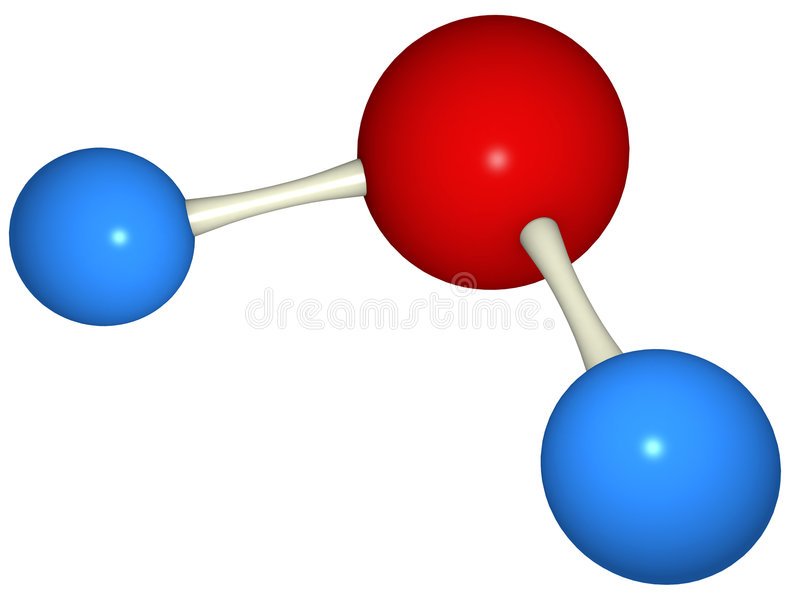
How many hydrogen atoms are there in a water molecule?
Red = Oxygen
Blue = Hydrogen
19
Multiple Choice

How many carbon atoms are there in a methane molecule?
Red = Carbon
Blue = Hydrogen
Structure of Matter:
Atoms, Molecules, and Compounds
By Mr. Brekke
Show answer
Auto Play
Slide 1 / 19
SLIDE
Similar Resources on Wayground

16 questions
APEMAN
Presentation
•
6th - 8th Grade

15 questions
LOO notes
Presentation
•
6th - 8th Grade

16 questions
HMH PS U3L4E2 analyzing the structure of matter
Presentation
•
6th - 8th Grade

14 questions
The Basics of Electricity
Presentation
•
6th - 8th Grade

14 questions
Models of Elements, Compounds, and Mixtures
Presentation
•
6th - 8th Grade

16 questions
Heat Transfer Evaluation Practice
Presentation
•
7th - 8th Grade

14 questions
Absolute Age Dating
Presentation
•
7th Grade

19 questions
Physical and Chemical Properties
Presentation
•
6th - 8th Grade
Popular Resources on Wayground

10 questions
5.P.1.3 Distance/Time Graphs
Quiz
•
5th Grade

10 questions
Fire Drill
Quiz
•
2nd - 5th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

22 questions
School Wide Vocab Group 1 Master
Quiz
•
6th - 8th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for Science

8 questions
Amoeba Sister Asexual vs Sexual Reproduction
Interactive video
•
8th Grade

19 questions
Introduction to Properties of Waves
Presentation
•
6th - 8th Grade

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

10 questions
Exploring the Layers of the Earth
Interactive video
•
6th - 10th Grade

16 questions
Photosynthesis and Cellular Respiration Review
Quiz
•
7th Grade

20 questions
Abiotic & Biotic Factors
Quiz
•
7th Grade

15 questions
Punnett Squares
Quiz
•
6th Grade

10 questions
Exploring the Evolution of Cell Theory
Interactive video
•
7th Grade