

UNIT 6
Presentation
•
Science
•
11th Grade
•
Practice Problem
•
Medium
Standards-aligned
VCIS Educational Apps
Used 1+ times
FREE Resource
62 Slides • 23 Questions
1

2

3

4
Multiple Choice
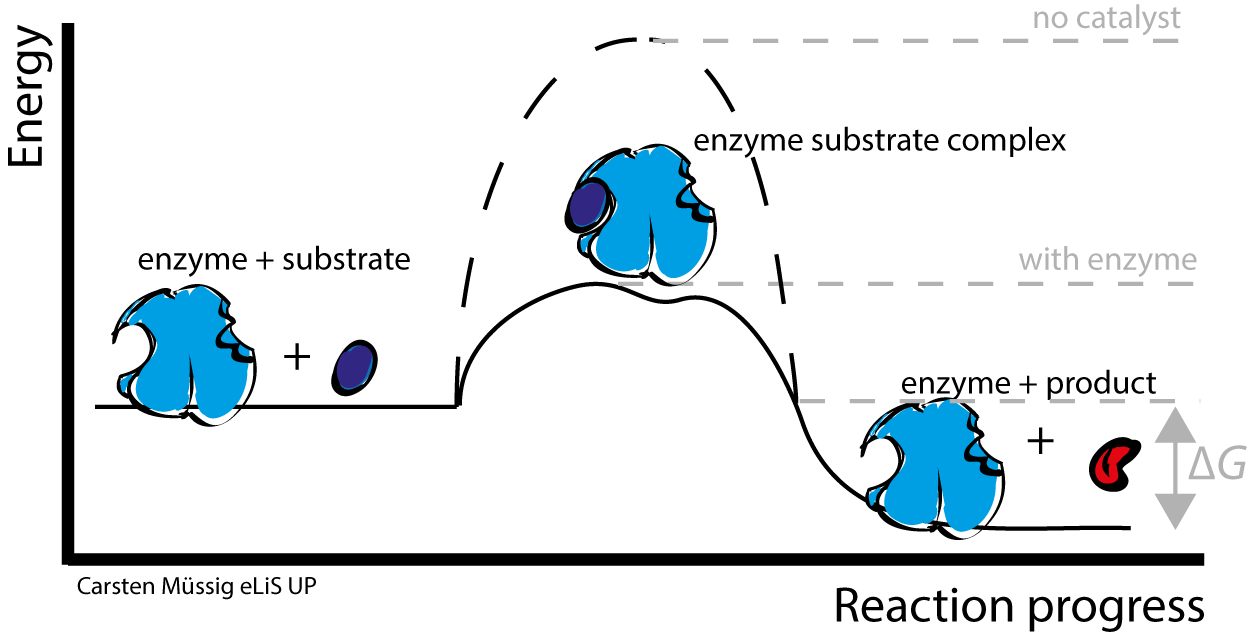
5

6

7

8
Multiple Choice
What is metabolism and why is it important for organisms?
It refers to the chemical reactions in organisms
It is the process of energy generation from food
It allows maintenance of normal functions and survival
All of the above
9

10
Fill in the Blanks
11

12
Multiple Choice
Which of the following processes could be described as catabolism?
The production of polypeptides from amino acids by the action of enzymes.
The synthesis of glycogen in the liver from glucose.
The digestion of starch into maltose in digestion.
The production of a large DNA molecule from nucleotide monomers.
13

14
Multiple Choice
What are the two main processes of metabolism illustrated in the image?
Catabolism
Anabolism
Photosynthesis
Respiration
15

16
Open Ended
What are oxidation/reduction reactions?
17
Multiple Choice
Which metal is most easily oxidized?
Co
Ag
Cu
Mg
18
Multiple Choice
Oxidation is the… (Higher Tier Question)
loss of electrons
gain of electrons
loss of protons
gain of protons
19

20
Multiple Choice
What is the difference between reduction and oxidation in terms of electron transfer?
Reduction involves losing electrons
Oxidation involves gaining electrons
Reduction involves gaining electrons
Oxidation involves losing protons
21

22
Multiple Choice
Redox reactions (reduction and oxidation reactions) pertain to the movement of what subatomic particle between atoms or molecules?
Electrons
Protons
Neutrons
Centrons
23

24

25

26
Open Ended
What are the real-life applications of redox reactions?
27

28
Multiple Choice

29

30
Multiple Choice
Which of the following is the correct sequence of stages in cellular respiration?
Glycolysis → Krebs cycle → Electron transport chain
Krebs cycle → Glycolysis → Electron transport chain
Electron transport chain → Glycolysis → Krebs cycle
Glycolysis → Electron transport chain → Krebs cycle
31

32
Multiple Choice
What are the products of glycolysis from one molecule of glucose?
2 ATP
2 NADH
2 pyruvate
2 ADP
33

34
Multiple Choice
What are the main products of the redox reactions in the Krebs cycle?
ATP
NADH
FADH2
CoA
35

36

37

38

39

40

41

42

43

44

45

46

47

48

49

50

51

52

53

54

55

56

57

58

59

60

61

62
Multiple Choice

This image shows oxidative phosphorylation, which is made of two steps. These steps are...
ETC and chemiosmosis
chemiosmosis and the Krebs Cycle
ATP Synthase and Glycolysis
Calvin Cycle and ETC
63

64

65

66

67

68

69
Multiple Choice

Examine the diagram of the electron transport chain below. Why do protons move through ATP synthase? Hint: how does diffusion work?
The protons move to an area of lower concentration.
The protons prefer to be in the cell's cytoplasm.
The protons need to enter the matrix to produce oxygen.
70

71
Multiple Choice
What are the two main processes shown in the equations for photosynthesis?
Oxidation of water and reduction of CO2
Oxidation of CO2 and reduction of water
Oxidation of glucose and reduction of oxygen
Oxidation of hydrogen and reduction of carbon
72

73
Multiple Choice
What are free radicals and why are they considered very reactive?
Atoms with paired electrons
Uncharged atoms or molecules with unpaired electrons
Charged molecules
Stable molecules
74

75
Multiple Choice
What are free radicals and how do they affect the body?
They are beneficial for health
They can cause damage
They are a type of antioxidant
They have no effect on the body
76

77
Multiple Choice
What role do antioxidants play in counteracting free radicals in the body?
They donate protons
They donate electrons
They absorb free radicals
They produce free radicals
78

79
Multiple Choice
What role do antioxidants play in relation to free radicals?
They donate protons to free radicals
They donate electrons to free radicals
They neutralize free radicals by absorbing them
They create more free radicals
80

81

82

83

84

85
Poll
How confident do you feel about this topic now?

Show answer
Auto Play
Slide 1 / 85
SLIDE
Similar Resources on Wayground

73 questions
Root, Stem, Leaf
Presentation
•
11th Grade

76 questions
Health Skills Grade 11 Term 2 Chapter 4
Presentation
•
11th Grade

85 questions
Public Opinion & Political Ideology
Presentation
•
11th - 12th Grade

81 questions
Measures of Central Tendency
Presentation
•
10th - 12th Grade

80 questions
The Periodic Table
Presentation
•
11th Grade

76 questions
Ch 5 notes - env science
Presentation
•
11th Grade

73 questions
Module No. 1: Universe, Solar System, and Earth System
Presentation
•
11th Grade

80 questions
2526 SESSION 3:Ang Mahiyaing Manok
Presentation
•
KG
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

40 questions
NCFE Earth and Environmental Science Released Test
Quiz
•
9th - 12th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade

20 questions
Frog Dissection Quiz
Quiz
•
11th Grade

20 questions
Final Review Thermochemistry
Quiz
•
9th - 12th Grade

11 questions
Explore Homeostasis and Feedback Mechanisms
Quiz
•
9th - 12th Grade

99 questions
PS Semester 2 Review
Quiz
•
9th - 12th Grade

49 questions
Florida Biology EOC Practice Test
Quiz
•
9th - 12th Grade

40 questions
Anatomy and Physiology Final Exam Review Part 1
Quiz
•
11th Grade