

Redox review
Presentation
•
Chemistry
•
11th Grade
•
Medium
Standards-aligned
Subashini Onichandran
Used 2+ times
FREE Resource
33 Slides • 37 Questions
1

2
Open Ended
Do you know what redox means?
3

4
Multiple Choice
Oxidation is the _________ of electrons.
loss
gain
sharing
transfer
5
Multiple Choice
Which of the following statements is true about reduction?
It involves the gain of oxygen.
It involves the loss of electrons.
It involves the gain of electrons.
It involves the loss of hydrogen.
6
Multiple Choice
Which of the following best describes an oxidising agent?
A substance that gains oxygen or loses electrons
A substance that loses oxygen or gains electrons
A substance that oxidises another substance and itself gets reduced
A substance that reduces another substance and itself gets oxidised
7

8

9
Multiple Choice
In the reaction Zn + PbO → ZnO + Pb, which substance is oxidized and which is reduced?
Zn is oxidized, PbO is reduced
Zn is reduced, PbO is oxidized
ZnO is oxidized, Pb is reduced
Pb is oxidized, ZnO is reduced
10

11
Fill in the Blanks
Type answer...
12

13

14
Multiple Choice
Which of the following statements about the reaction Fe₂O₃ + 3CO → 2Fe + 3CO₂ is correct?
Fe₂O₃ is oxidized to Fe
CO is reduced to CO₂
Fe₂O₃ is reduced to Fe
CO₂ is oxidized to CO
15

16
Multiple Select
Select all reactions where a metal oxide is reduced to a metal.
Zn + PbO → ZnO + Pb
2CuO + C → 2Cu + CO₂
Fe₂O₃ + 3CO → 2Fe + 3CO₂
H₂O + Mg → MgO + H₂
17
Open Ended
Explain how the transfer of oxygen is used to identify oxidation and reduction in chemical reactions. Use examples from the images to support your answer.
18
Multiple Choice
Which of the following reactions demonstrates both oxidation and reduction?
Zn + PbO → ZnO + Pb
2CuO + C → 2Cu + CO₂
Fe₂O₃ + 3CO → 2Fe + 3CO₂
All of the above
19

20
Multiple Choice
Which process involves the gain of oxygen and which involves the loss of oxygen in the reaction H2O(g) + Mg → MgO + H2?
Oxidation involves gain of oxygen, reduction involves loss of oxygen
Oxidation involves loss of oxygen, reduction involves gain of oxygen
Both involve gain of oxygen
Both involve loss of oxygen
21

22

23
Multiple Select
Which of the following statements are correct about oxidation and reduction based on electron transfer?
Oxidation is the loss of electrons
Reduction is the gain of electrons
Oxidation is the gain of electrons
Reduction is the loss of electrons
24

25
Fill in the Blanks
Type answer...
26

27
Open Ended
Explain how the transfer of electrons between magnesium and oxygen leads to the formation of an ionic bond.
28

29
Open Ended
Describe the relationship between oxidation, reduction, and redox reactions using the equations provided in the electron transfer slides.
30
Multiple Choice
What is the overall equation for the reaction between magnesium and oxygen as shown in the electron transfer diagrams?
2Mg + O2 → 2MgO
Mg + O2 → MgO2
Mg + O → MgO
2Mg + 2O2 → 2MgO2
31

32
Multiple Choice
Which process involves the loss of electrons?
Oxidation
Reduction
Precipitation
Neutralization
33

34
Multiple Choice
In the reaction Cu2+ + Mg → Cu + Mg2+, which species is oxidized and which is reduced?
Cu2+ is reduced, Mg is oxidized
Cu2+ is oxidized, Mg is reduced
Both are oxidized
Both are reduced
35

36
Multiple Select
Select all correct statements about oxidation and reduction based on the diagrams shown.
Oxidation is the loss of electrons
Reduction is the gain of electrons
Oxidation state increases during oxidation
Reduction involves the loss of electrons
37
Open Ended
Explain how electron transfer occurs in the reaction Zn + 2H+ → Zn2+ + H2. What happens to zinc and hydrogen ions?
38

39

40

41
Multiple Choice
Which of the following elements has an oxidation state of 0 in its elemental form?
Mg
F2
O2
All of the above
42
Fill in the Blanks
Type answer...
43

44
Multiple Choice
Which of the following best describes the oxidation state in a compound?
It is always positive.
It corresponds to charges in a compound.
It is always negative.
It is the number of protons in an atom.
45

46
Multiple Choice
Based on the compounds MgCl2 and NH3, what is the total oxidation state of each compound?
+2 for both
0 for both
-2 for both
+1 for both
47

48

49
Fill in the Blanks
Type answer...
50

51
Multiple Choice
Which of the following statements is correct regarding oxidation and reduction?
A rise in oxidation number means reduction.
A fall in oxidation number means oxidation.
A rise in oxidation number means oxidation.
Oxidation and reduction do not involve changes in oxidation number.
52

53
Multiple Choice
In the redox equation shown, which element is reduced and which is oxidized?
Fe is reduced, C is oxidized
Fe is oxidized, C is reduced
Both Fe and C are oxidized
Both Fe and C are reduced
54

55
Multiple Choice
Mg+2 HCl→MgCl2+H2
Mg is
oxidized
reduced
a spectator ion
56
Multiple Choice
In the reaction 2K+Br2→2KBr , which element is reduced?
Potassium (K)
Bromine (Br_2)
Potassium bromide (KBr)
None of the above
57
Match
Determine the Oxidation State for each of the following
Mg2+
Cl
Cl in NaCl
S in LiHSO4
O in H2O
+2
0
-1
+6
-2
+2
0
-1
+6
-2
58
Multiple Choice
What is the oxidation number of Cr in Cr2O72- ?
+14
+6
-6
-14
59

60

61

62

63

64
Multiple Choice
What is the term used for a substance that donates electrons in a redox reaction?
Oxidising agent
Reducing agent
Catalyst
Inhibitor
65

66
Multiple Choice
Combustion and single-replacement reactions are __________ redox reactions.
always
never
sometimes
mostly
67
Dropdown
oxidation is defined as
while reduction is defined as
68
Multiple Choice
Which of the following best describes a reducing agent?
A substance that gains oxygen
A substance that loses electrons
A substance that reduces another substance and itself gets oxidised
A substance that oxidises another substance and itself gets reduced
69
Multiple Choice
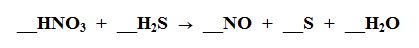
70
Multiple Choice
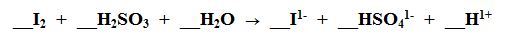

Show answer
Auto Play
Slide 1 / 70
SLIDE
Similar Resources on Wayground

69 questions
Part 3 EOC Review
Presentation
•
10th Grade

65 questions
Patterns and Properties of Written Texts
Presentation
•
11th Grade

61 questions
Federal Budget
Presentation
•
11th - 12th Grade

66 questions
Чумацький шлях. Будова галактики. Уроки 26-31
Presentation
•
11th Grade

66 questions
Industrialization and Gilded Age
Presentation
•
11th Grade

60 questions
Atomic Theory Notes
Presentation
•
11th Grade

69 questions
Equilibrium Calculations
Presentation
•
10th - 11th Grade

67 questions
CLAIMS
Presentation
•
11th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade