

Lição sem título
Presentation
•
Chemistry
•
University
•
Practice Problem
•
Hard
Standards-aligned
ELAINE ESPER
FREE Resource
51 Slides • 23 Questions
1

2
Multiple Choice

3

4
Multiple Choice
What are the key differences between the atomic models proposed by Dalton, Thomson, Rutherford, Bohr, Sommerfeld, and Schrödinger?
They differ in the number of particles
They differ in the arrangement of particles
They differ in the energy levels of electrons
They differ in the existence of the nucleus
5

6
Multiple Choice
What does the term "átomo" signify according to Leucippus and Democritus?
Indivisible particle
Divisible particle
Elemental substance
Compound substance
7

8
Multiple Choice
What are the main points of John Dalton's atomic theory as described in the image?
Atoms are indivisible and indestructible
Atoms of different substances are identical
Atoms can be created and destroyed
Atoms are not spherical
9

10
Multiple Choice
Dalton's model of the atom failed because it could not explain :
electric charges between matter
how atoms could form molecules
how particles could be so small
11

12
Multiple Choice

What scientist proposed this atomic model?
Dalton
Thomson
Rutherford
Bohr
13

14
Multiple Choice
What are the relative charges and masses of protons, neutrons, and electrons according to Thomson's atomic theory?
Proton: +1, Neutron: 0, Electron: -1
Proton: +1, Neutron: 1, Electron: 0
Proton: 0, Neutron: +1, Electron: -1
Proton: +1, Neutron: -1, Electron: 0
15

16
Multiple Choice
What is the conclusion drawn from the experiment involving cathode rays?
There are positively charged particles
There are negatively charged particles
There are no charged particles
There are only neutrons
17

18
Multiple Choice
What does the diagram illustrate about Thomson's atomic theory?
The structure of an atom
The behavior of electrons in a vacuum tube
The discovery of protons
The concept of atomic mass
19
Multiple Choice
Experiments with the use of cathode ray tubes led to the discovery of the
electron
protons
nucleus
orbital
20

21
Multiple Choice
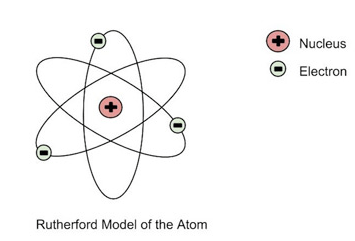
22

23
Multiple Choice
What did W. Röentgen discover about X-rays in 1895?
They can be deflected by magnetic fields
They can pass through opaque materials
They are visible to the naked eye
They are composed of charged particles
24

25

26

27
Multiple Choice
What did Rutherford interpret about the structure of the atom based on his experiments?
Most alpha particles passed through without deflection
The nucleus is negatively charged
Electrons are located in the nucleus
Alpha particles are positively charged
28

29
Multiple Choice
What are the main differences between the Thomson and Rutherford models of the atom?
Thomson model has a positive charge spread throughout the atom
Rutherford model has electrons orbiting a dense nucleus
Thomson model depicts electrons as negative particles
Rutherford model shows a solid sphere structure
30

31
Multiple Choice
What is the flaw in Rutherford's model as described by the Theory of Electromagnetism?
It does not account for electron emission
It suggests electrons are stationary
It ignores nuclear forces
It claims particles do not emit energy
32

33
Multiple Choice
What is the relationship between the colors observed in the flames and the atomic structure?
They indicate the temperature of the flame
They are related to the energy levels of electrons
They show the presence of different metals
They have no relation to atomic structure
34

35
Multiple Choice

In his model of the atom, Bohr proposed that electrons move at fixed distances from the nucleus and that the energy of the electron is related to its orbit. What discovery contributed to the development of this atomic model?
Electrons can flow freely through metals making metals good conductors.
Elements have characteristic wavelengths of light emission and absorption.
Noble gas elements are much less reactive than most other elements.
Electrons exhibit properties of both particles and waves.
36

37

38
Fill in the Blanks
Type answer...
39

40
Multiple Choice

Which scientist developed the model of the atom shown?
Chadwick
Thomson
Rutherford
Bohr
41

42

43

44
Multiple Choice
Qual é a correlação correta entre os cientistas e suas ideias sobre a composição do átomo?
apenas nos itens I e II
apenas no item II
apenas nos itens I e III
apenas nos itens II e III
apenas no item III
45

46
Multiple Choice

47

48
Multiple Choice

Which model of the atom describes the location of electrons as probable within a dense "cloud" surrounding the nucleus of an atom?
The Cannonball
Model
The Plum Pudding Model
The Planetary Model
The Quantum Mechanical Model
The Nuclear Model
49

50

51

52

53

54

55

56

57

58

59

60

61

62

63

64

65

66

67

68

69

70

71

72

73

74
Poll
Quão confiante você se sente sobre este assunto agora?

Show answer
Auto Play
Slide 1 / 74
SLIDE
Similar Resources on Wayground

69 questions
Regional Human Rights Protection
Presentation
•
University

67 questions
Quiz#1 Review (P1 of 3) - Ch1
Presentation
•
University

67 questions
SRM Lecture 3-1 STP and Branding
Presentation
•
University

68 questions
Chapter 2 Measurement and Problem Solving
Presentation
•
University

68 questions
CHEMICAL EQUATIONS AND REACTION TYPES
Presentation
•
University

70 questions
Bahasa Inggris 1 Week 10 and Week 11
Presentation
•
University

70 questions
Chemical Periodicity
Presentation
•
12th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Chemistry

20 questions
Guess The App
Quiz
•
KG - Professional Dev...

11 questions
NFL Football logos
Quiz
•
KG - Professional Dev...

19 questions
Minecraft
Quiz
•
6th Grade - Professio...

40 questions
8th Grade Math Review
Quiz
•
8th Grade - University

20 questions
Block Buster Movies
Quiz
•
10th Grade - Professi...

10 questions
Would you rather...
Quiz
•
KG - University

40 questions
Flags of the World
Quiz
•
KG - Professional Dev...

14 questions
Superhero
Quiz
•
1st Grade - University