

7.PS1.2-Mixtures, Reactions, and Changes in Matter-notes/slides
Presentation
•
Science
•
7th Grade
•
Hard
+14
Standards-aligned
Jessica Freeman
FREE Resource
69 Slides • 70 Questions
2

3
Mixtures
Mixtures are physical combinations of two or more substances.


Hetero = different
Homo = same
4

General properties of a mixture:
● The components of a mixture can be easily separated
● The components each keep their original properties
Characteristics of Mixtures
5
Multiple Select
Which of the following are examples of Mixtures?
The atmosphere
(N + O + Ar + H2O...)
Carbon Dioxide (CO2)
Oxygen
(O2)
Lemonade
(sugar, lemon juice, water)
Anything on the Periodic Table
6
Multiple Choice
Mixtures
are made of two different types of atoms chemically bonded together
Are only liquids
Cannot be separated
Can be separated using physical properties
7
Types of Mixtures
Heterogenous Mixture
- Heterogeneous mixtures have distinct components that can be easily seen and sorted.
- Heterogeneous mixtures can have uneven distribution of components.
Examples: Trail Mix, Salad, Spaghetti and Meatballs

8
Types of Mixtures
Homogenous Mixtures
- Homogeneous mixtures have a uniform appearance throughout the entire mixture, making it difficult to distinguish their different components.
Examples: saltwater, honey, and soy sauce

9
Multiple Choice
Lucky Charms
Heterogeneous
Homogeneous
10
Multiple Choice
Freshly brewed black coffee
Heterogeneous
Homogeneous
11
Multiple Choice
Dirt
Heterogeneous
Homogeneous
12
Multiple Choice
Air
Heterogeneous
Homogeneous
13
Multiple Choice
Trail Mix
Heterogeneous
Homogeneous
14

15
16
Multiple Choice

How many different types of particles are in this model?
Only 1 type of particle.
2 types of particles.
3 types of particles.
4 types of particles.
17
Multiple Choice

Is this a pure substance or a mixture?
pure substance
mixture
18
Multiple Choice

How many different types of particles are in this model?
Only 1 type of particle.
2 types of particles.
3 types of particles.
4 types of particles.
19
Multiple Choice

Is this a pure substance or a mixture?
pure substance
mixture
20
Multiple Choice

How many different types of particles are in this model?
Only 1 type of particle.
2 types of particles.
3 types of particles.
4 types of particles.
21
Multiple Choice

Is this a pure substance or a mixture?
pure substance
mixture
22
23
Multiple Choice
Which of the following is a mixture?
gold
cookie dough
water
salt
24
Fill in the Blanks
Type answer...
25
26
Multiple Choice
Imagine you mix sand and water in a beaker.
How could you separate the sand and water?
27
Multiple Choice
How could you separate a mixture of iron and sand?
28
29
Multiple Choice

Why is it possible to separate iron and sand after they have been mixed?
The properties of iron and sand become identical once they are mixed.
The properties of iron and sand change once they are mixed.
The properties of iron and sand do not change once they are mixed.
30
Multiple Choice

Imagine you have a mixture of sand and plastic beads you want to separate. You add water to the mixture. The beads float to the top and the sand sinks to the bottom.
Which of the following is true?
The beads and the sand kept their original densities after they mix.
The density of the beads and sand changed after they mix.
The density of the beads and sand become the same after they mix.
31
32
Multiple Choice
Which of the following is a mixture?
33
Multiple Choice
Which of the following is a mixture?
34
Multiple Choice
Which of the following is a mixture?
35

mixture
component
NOT
physical
properties
salt
water
36
37

In science, properties are the characteristics that enable us to differentiate one material from another.
A synonym is characteristic or trait.
What is a property?
38

39
Separation of Mixtures
How do we separate Homogeneous and Heterogeneous mixtures??

40
Multiple Choice
A combination that can be separated by physical processes is a .....
Pure Substance
Glucose
Element
Mixture
41

42

43

44
Chromatography
Chromatography SEPARATES substances as they are drawn across a barrier.
Chromatography happens when different colors from an ink pen spread out on a paper towel.

45
Distillation
Separates mixtures based on differences in substances' boiling points.
Distillation is used to process crude oil into its many different parts such as gasoline.

46

47

48
Multiple Choice
If a solid doesn't dissolve in a liquid we say that it is
Soluble
A solvent
Insoluble
Undissolveable
49
Multiple Choice
What happens when salty water is boiled?
The salt becomes a new compound and water evaporates as it has a low boiling point
The water evaporates and the salt remains in the beaker as it has a high melting point
The water becomes a new compound and evaporates while salt remains in the beaker
The water will disappear and the salt remains in the beaker as it is a solid
50
Multiple Choice
The process used to separate heterogeneous mixtures of solids and liquids is called
filtration
crystallization
distillation
chromatography
51
Multiple Choice

If you spilled nails in a sandbox, what separation tool/method could you use to clean out the sandbox so it would be safe for children to play in?
Filter
Screen
Magnet
Evaporation
52
Multiple Choice

This type of paper allows liquid to pass through it. What tool is it?
sieve
filter
53
Multiple Choice
Mixtures can be separated .......................
Physically
Chemically
54

55

56

57

58
Physical Properties of Matter
Think of the different ways you can describe an object....

59

60

61

62

63

64
Multiple Choice
Any measurable or observable attribute that describes matter
Physical Property
Chemical Property
65

66
Multiple Choice
The ability of a substance to be pressed or pounded into a thin sheet.
magnetism
conductivity
ductility
malleability
67
Multiple Choice
The ability of a substance to be pulled into a thin strand.
ductility
melting point
buoyancy
density
68

69

70

71
Multiple Choice

The ability of a substance to dissolve is called...
malleability
ductility
solubility
72
Multiple Choice
An attractive force for some metals.
Conductivity
Solubility
Magnetism
Mass
73
Multiple Choice
The ability of matter to conduct electric current or heat
Solubility
Mass
Weight
Conductivity
74
Multiple Choice

Which box has a higher density?
Box A
Box B
cannot be determined
they are the same
75
Multiple Choice
Properties that can be measured or observed without matter changing to an entirely different substance.
Chemical Properties of Matter
Physical Properties of Matter
Physical Change
Chemical Change
76
Multiple Choice
Density can be used as a physical property of identification of a substance because the density of a substance ___.
stays the same no matter the size of the sample
changes with each different amount of a substance
is determined by the substance's height
is also a chemical property
77
Multiple Choice
When a solid reaches the temperatures of its ___, it can become a liquid.
melting point
freezing point
boiling point
density
78
Multiple Choice
What is the state of matter that has a fixed volume but not a fixed shape?
Solid
Liquid
Gas
79
Multiple Choice
The temperature at which a substance changes from a liquid to a gas
Melting point
Boiling point
Freezing point
Condensation point
80

physical
property
color
mass
density
data
substances
81

82

83

84

85
Chemical Reactions

86

87
Multiple Choice
When a new substance forms as a result of a chemical reaction, what type of change has occurred?
Chemical Change
Physical Change
88

89
Multiple Choice
Which is NOT an indication of a chemical change?
Temperature Change
Shape Change
Formation of a precipitate
Production of light
90

91

92
Open Ended

Explain what is happening in the following image.
93

94

95
Open Ended
Explain how the properties of salt are different from the elements that form it.
96

97
How can you tell a chemical reaction is happening?
A change of color occurs during the reaction
A gas is produced during the reaction.
A solid product called a precipitate is produced during the reaction.
A transfer of energy occurs as a result of the reaction.
98
Example 1: Color Change
A solution of potassium permanganate is added to a solution of sodium hydroxide and sugar.
Notice how the colors change as the reaction continues
99
Poll
When food coloring is added to water, is a chemical reaction taking place?
Yes
No
100
A chemical reaction is NOT taking place? WHY???
Your reactants are food coloring and water and the product is food coloring and water.
There were no other signs of a chemical reaction taking place
The color change of water (went from clear to the color of the food coloring) was predictable. Often the color change in a chemical reaction are not necessarily predictable (as in the video)
101
Example 2: gas production
When zinc reacts with nitric acid, the reaction bubbles vigorously as Nitrogen gas is produced. The production of a gas is also an indication that a chemical reaction is occurring.
102
Example 3: precipitate formation
When a colorless solution of lead(II) nitrate is added to a colorless solution of potassium iodide, a yellow solid called a precipitate is instantly produced.
103
Transfer of Energy
All chemical changes involve a transfer of energy.
Energy/heat can be absorbed =Endothermic reaction.
Or
Energy/heat can be given off=Exothermic reaction
104
Example 4: Energy Transfer
Sodium and water produce an explosion. Energy/heat is being released. This is an exothermic reaction.
105
Open Ended
In two minutes,
write the four indicators of a chemical change.
106
Multiple Choice
Which of the following describes changes in energy during a chemical reaction?
Changes in energy do not occur during a chemical reaction.
Energy is always released during a reaction.
Energy may be either absorbed or released during a reaction.
Energy is always released during a reaction.
107
Multiple Select
Which of the following indicated a chemical change/chemical reaction?
Precipitate formation
Transfer of heat energy
108
Open Ended
Two reactants are mixed in a test tube, and the test tube becomes very warm. This is a sign of a transfer of ______ taking place.
109
Multiple Select
What state of matters, when produced, is a sign that a chemical reaction is occuring?
gas
liquid
solid
plasma
110
Multiple Choice
When a color change happens, a chemical reaction must have occurred.
false
true
111
Poll
Lesson Reflection: How confident do you feel in your ability to determine if a chemical change has occurred?
very confident
confident
partly confident
not confident at all
112

113

chemical
reaction
identity
different
gas formation
temperature change
color change
114

115

116
Drag and Drop

117
Hotspot
Which observations are evidence that a chemical reaction took place?
118
Drag and Drop

Experiment 2
Experiment 3
Experiment 4
did
119
Drag and Drop
120
Match
Match the observations with their evidence that a chemical change has occurred
a reaction goes from 40 degrees to 70 degrees
A baggie puffs up when 2 substances are mixed inside
rust on a nail
meat has gone bad
milk turns chunky after being left out for days
temperature change
gas production
color change
odor produced
formation of a precipitate
temperature change
gas production
color change
odor produced
formation of a precipitate
121
Dropdown
122
Dropdown
123
Multiple Choice
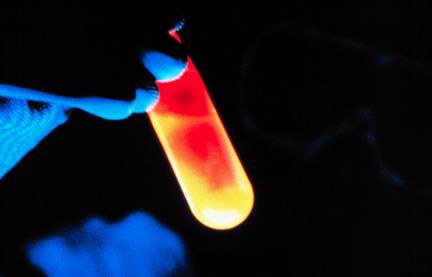
124

125

126
Multiple Choice

A student was doing an investigation. Below are the observations before and after the investigation.
Which observation provides evidence that a new substance formed in the beaker after the investigation?
Observation 1
Observation 1 & 4
Observation 2 & 3
A new substance was not formed
127
Multiple Choice

In the example provided, what evidence of a chemical change do you see?
Color change
Precipitate formation
Water production
Gas evolution
128
Multiple Choice

Based on their observations, can the students correctly conclude that a chemical reaction occurred?
No, because the metal strip was still visible
Yes, because a new material of a different color formed on part of the metal strip
No, because the solution stayed blue
Yes, because the volume of the blue solution stayed the same
129
Multiple Choice

Burning a candle is a chemical change. What is the primary evidence that burning the candle is a chemical reaction?
change in color
change in odor
formation of a solid
temperature change
130
Multiple Choice

What evidence of a chemical reaction do you see in the picture?
bubbling (bubbles)
color change
precipitate
heat
131
Multiple Choice

Production of Gas or Bubbles is evidence of a
Physical Change
Chemical Change
132
Multiple Choice

Question 7: Hoey
Dr. Hoey develops an experiment to investigate how different substances react when they are mixed with water. Before mixing the materials together, she measured the mass and temperature of the substances. During the experiment, Dr. Hoey mixes calcium and water in a glass with a plastic rod. Then, she mixes baking soda and water in a glass with a plastic rod. The results of the experiment are below.
Which of the following claims can Dr. Hoey make based on her investigation?
Solution A is a new substance because after the calcium and water mixed, the temperature increased and the color changed.
Solution B is a new substance because after the baking soda and water mixed, the solution was cloudy and the temperature remained the same.
Solution B is a new substance because after the baking soda and water mixed, the mass of the solution is equal to the mass of the baking soda and water before they were mixed.
Solution A and B are new substances because each solution stayed the same when they were mixed together.
133
Multiple Choice

A blue liquid was added to a clear liquid. Which evidence that a chemical reaction may have occurred?
the solution turned light blue
the temperature remained the same
solid particles formed
the volume increased
134
Multiple Choice

Students performed a lab investigation on chemical reactions. The
students put on goggles and plastic gloves and then followed these
steps. Which observation would indicate that a chemical reaction
occurred?
The vinegar took the shape of the container in step 1.
The steel wool sank into the vinegar in step 3.
The temperature of the vinegar increased during step 4.
The steel wool changed shape during step 6.
135

136

before
after
physical
evidence
chemical
137

138

139

Show answer
Auto Play
Slide 1 / 139
SLIDE
Similar Resources on Wayground

127 questions
ENERGIA
Presentation
•
8th Grade

137 questions
YLE Flyers Grammar & Vocabulary Review
Presentation
•
5th Grade

126 questions
Events Leading Up To Civil War
Presentation
•
8th Grade

138 questions
WH1 RC2: Classical Civilizations
Presentation
•
9th Grade

136 questions
8.ESS2.3-Powered from Below: How Earth’s Interior Moves the Plates-Lesson Slides/notes
Presentation
•
8th Grade

140 questions
CATEGORY 1 CIVICS EOC REVIEW
Presentation
•
7th - 8th Grade

127 questions
ipA KLS 7
Presentation
•
7th Grade

131 questions
UNIT 6: Expressions and Equations
Presentation
•
6th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

20 questions
genetics, punnett squares, heredity
Quiz
•
7th Grade

16 questions
Photosynthesis and Cellular Respiration Review
Quiz
•
7th Grade

20 questions
Food Webs + Energy Pyramids
Quiz
•
7th Grade

10 questions
Exploring the Rock Cycle
Interactive video
•
6th - 8th Grade

10 questions
Exploring Earth's Seasons and Their Causes
Interactive video
•
6th - 8th Grade

26 questions
Amplify Chemical Reactions
Quiz
•
6th - 8th Grade

19 questions
Natural and Artificial Selection
Quiz
•
7th Grade
