
Rates of Reaction
Authored by Georgina Casey
Chemistry
11th Grade
Used 8+ times

AI Actions
Add similar questions
Adjust reading levels
Convert to real-world scenario
Translate activity
More...
Content View
Student View
23 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
Which factors increase the rate of a reaction.
2.
MULTIPLE CHOICE QUESTION
1 min • 1 pt
Increase in temperature of the reactants can do one of the following
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How does a catalyst work in speeding up a reaction?
By lowering the activation energy
by giving them more energy
by making them more available
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which graph shows the effect of increasing temperature on the rate of reaction of calcium carbonate with dilute hydrochloric acid?
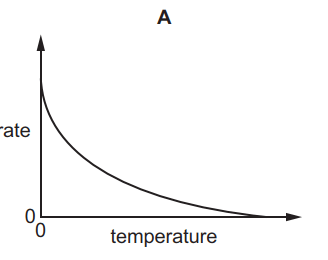
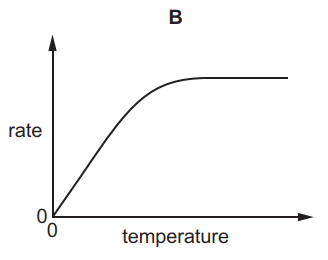
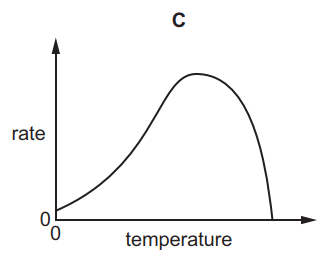
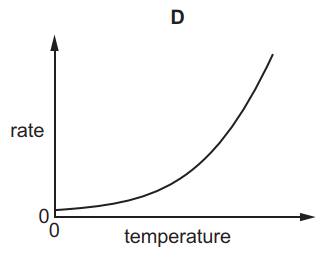
5.
MULTIPLE CHOICE QUESTION
15 mins • 1 pt

What type of reaction is this?
6.
MULTIPLE CHOICE QUESTION
1 min • 1 pt

The graph shows how the total volume of a gas given off from a reaction changes with time. In which time interval is least gas given off?
7.
MULTIPLE CHOICE QUESTION
1 min • 1 pt

In an experiment, a 2 g lump of zinc and 2 g of powdered zinc are added separately to equal volumes of dilute sulphuric acid. The solid line on the graph shows the volume of gas given off when the 2 g lump is used. Which dotted line is obtained when the zinc is powdered?
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?



