AP Chemistry Topic 8.6
Authored by Charles Martinez
Chemistry
11th - 12th Grade
NGSS covered
Used 1+ times

AI Actions
Add similar questions
Adjust reading levels
Convert to real-world scenario
Translate activity
More...
Content View
Student View
15 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
15 mins • 1 pt

A buffer solution is prepared by mixing equal volumes of 0.50 M weak acid with 1.0 M of its conjugate base. Based on the data given in the table above, which of the following pairs of chemical solutions should be used to prepare the buffer solution so that the pH will be between 4 and 7?
CH3COOH and NH3
CH3COOH and CH3COONa
H2CO3 and NH3
H2CO3 and Na2CO3
2.
MULTIPLE CHOICE QUESTION
15 mins • 1 pt
Ascorbic acid, H2C6H6O6(s), is a diprotic acid with K1 = 7.9 × 10–5 and K2 = 1.6 × 10–12. In a 0.005 M aqueous solution of ascorbic acid, which of the following species is present in the lowest concentration?
H3O+(aq)
H2C6H6O6(aq)
HC6H6O6-(aq)
C6H6O62-(aq)
Tags
NGSS.HS-PS1-5
3.
MULTIPLE CHOICE QUESTION
15 mins • 1 pt
In the titration of a weak acid of unknown concentration with a standard solution of a strong base, a pH meter was used to follow the progress of the titration. Which of the following is true for this experiment?
The [H+] at the equivalence point equals the ionization constant of the acid.
The pH at the equivalence point depends on the indicator used.
The graph of pH versus volume of base added rises gradually at first and then much more rapidly.
The graph of pH versus volume of base added shows no sharp rise.
4.
MULTIPLE CHOICE QUESTION
15 mins • 1 pt
Which of the following is the correct equilibrium expression for the hydrolysis of CO3 2– ?
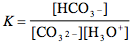
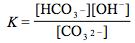
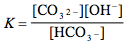
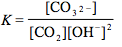
Tags
NGSS.HS-PS1-6
5.
MULTIPLE CHOICE QUESTION
15 mins • 1 pt

Which of the following best approximates the Ka value for this weak acid?
1 x 10–3
1 x 10–4
1 x 10–5
1 x 10–6
6.
MULTIPLE CHOICE QUESTION
15 mins • 1 pt

Which point on the titration curve corresponds to the point at which the moles of the added strong base are equal to the moles of the weak acid initially present?
Q
R
S
T
7.
MULTIPLE CHOICE QUESTION
15 mins • 1 pt

At point P in the titration, which of the following species has the highest concentration?
HA
A–
H3O+
OH–
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
Similar Resources on Wayground

15 questions
SEL VOLTA DAN SEL ELEKTROLISIS
Quiz
•
12th Grade

10 questions
4.4 Metallic bonding and Allotropes
Quiz
•
11th - 12th Grade

12 questions
Карбонові кислоти
Quiz
•
9th - 12th Grade

15 questions
Penilaian Harian II X IPA (Hukum-Hukum Dasar Kimia)
Quiz
•
10th - 12th Grade

14 questions
Алканы. Гомологический ряд
Quiz
•
10th Grade - University

10 questions
ÔN TẬP 10 CÂU CUỐI KỲ 2 LẦN 1 HÓA 12
Quiz
•
12th Grade

19 questions
Kwasy karboksylowe
Quiz
•
8th Grade - Professio...

18 questions
ANĐEHIT – AXIT CACBOXYLIC
Quiz
•
1st Grade - University
Popular Resources on Wayground

8 questions
Spartan Way - Classroom Responsible
Quiz
•
9th - 12th Grade

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

14 questions
Boundaries & Healthy Relationships
Lesson
•
6th - 8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

3 questions
Integrity and Your Health
Lesson
•
6th - 8th Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

9 questions
FOREST Perception
Lesson
•
KG

20 questions
Main Idea and Details
Quiz
•
5th Grade
Discover more resources for Chemistry

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
momentum and impulse
Quiz
•
9th - 12th Grade

40 questions
Unit 3 (Part 1) Chemical Equations & Reactions Review Game
Quiz
•
8th - 12th Grade

20 questions
Naming Covalent Compounds
Quiz
•
11th Grade

35 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

15 questions
Ionic Bonding
Quiz
•
10th - 11th Grade

12 questions
Gas Laws Practice
Quiz
•
10th - 12th Grade

10 questions
Exploring Solubility Curves
Interactive video
•
9th - 12th Grade