
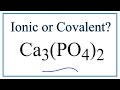
Phosphate Ion and Calcium Phosphate Concepts
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Lucas Foster
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of compound is calcium phosphate primarily considered?
Hydrogen-bonded
Covalent
Metallic
Ionic
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which elements in calcium phosphate are non-metals?
Calcium and phosphorus
Calcium and hydrogen
Phosphorus and oxygen
Calcium and oxygen
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the ionic charge of calcium in calcium phosphate?
2+
3+
4+
1+
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the charge on the phosphate ion in calcium phosphate?
2-
4-
1-
3-
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do the charges of calcium and phosphate ions balance in calcium phosphate?
They do not balance
Calcium's charge is greater
Phosphate's charge is greater
They balance to a net charge of zero
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of ion is the phosphate ion considered?
Triatomic ion
Diatomic ion
Monatomic ion
Polyatomic ion
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of bond exists between phosphorus and oxygen in the phosphate ion?
Ionic bond
Covalent bond
Metallic bond
Hydrogen bond
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
