
Exploring Ionic and Covalent Bonds
Interactive Video
•
Science
•
6th - 10th Grade
•
Practice Problem
•
Hard
Standards-aligned
Lucas Foster
FREE Resource
Standards-aligned
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
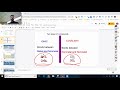
What is the main difference between ionic and covalent bonding?
Both ionic and covalent bonding involve transferring electrons.
Ionic bonding involves transferring electrons, while covalent bonding involves sharing electrons.
Both ionic and covalent bonding involve sharing electrons.
Ionic bonding involves sharing electrons, while covalent bonding involves transferring electrons.
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In ionic bonding, what happens to the electrons?
They are shared equally between atoms.
They are transferred from one atom to another.
They are lost completely.
They remain with the same atom.
Tags
NGSS.HS-PS1-7
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of elements typically form ionic bonds?
Metalloids
Two metals
Two non-metals
A metal and a non-metal
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In covalent bonding, what do atoms do with their electrons?
They lose them completely.
They share their valence electrons.
They gain extra electrons.
They transfer them to other atoms.
Tags
NGSS.HS-PS1-1
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following is an example of a covalent bond?
Carbon dioxide (CO2)
Calcium bromide (CaBr2)
Sodium chloride (NaCl)
Magnesium oxide (MgO)
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How do the melting and boiling points of ionic compounds compare to those of covalent compounds?
Ionic compounds have higher melting and boiling points.
Both have similar melting and boiling points.
Covalent compounds have higher melting and boiling points.
Ionic compounds have lower melting and boiling points.
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
At room temperature, ionic compounds are usually found in which state?
Solid
Liquid
Gas
Plasma
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
