
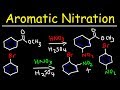
Nitration Mechanisms and Products
Interactive Video
•
Chemistry, Science
•
10th Grade - University
•
Practice Problem
•
Hard
Sophia Harris
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the major product when methyl benzoate is nitrated?
Dinitro methyl benzoate
Para-nitro methyl benzoate
Ortho-nitro methyl benzoate
Meta-nitro methyl benzoate
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why does the NO2 group attach to the meta position in methyl benzoate?
Because the ester group is electron-donating
Because the benzene ring is electron-rich
Because the carbonyl group is electron-withdrawing
Because the reaction is carried out at high temperature
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the role of sulfuric acid in the nitration of methyl benzoate?
It neutralizes the reaction
It acts as a catalyst
It provides the nitronium ion
It acts as a solvent
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which base can be used to remove the hydrogen in the nitration mechanism of methyl benzoate?
Water
Bisulfate ion
Any of the above
None of the above
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of director is bromine in the nitration of bromobenzene?
Activating director
Deactivating director
Meta director
Ortho-para director
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which product is formed when bromobenzene is nitrated at the ortho position?
Para-bromo nitrobenzene
Dinitro bromobenzene
Meta-bromo nitrobenzene
Ortho-bromo nitrobenzene
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the charge on the orthocarbon during the nitration of bromobenzene?
Negative
Double positive
Positive
Neutral
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
